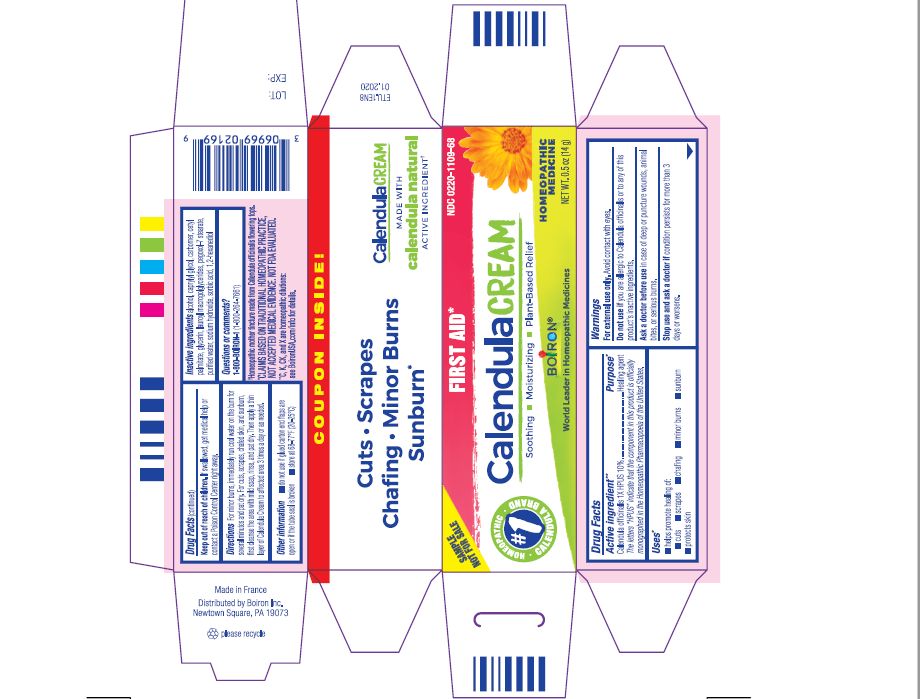
Label: CALENDULA- calendula officinalis flowering top cream
- NDC Code(s): 0220-1109-63, 0220-1109-68, 0220-1109-93
- Packager: Laboratoires Boiron
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated January 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- ASK DOCTOR
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
For minor burns, immediately run cool water on the burn for several minutes and pat dry. Then apply a thin layer of Calendula Cream to affected area 3 times a day or as needed.
For cuts, scrapes, chafed skin, and sunburn, first cleanse the area with mild soap, rinse, and pat dry. Then apply a thin layer of Calendula Cream to affected area 3 times a day or as needed.
-
INACTIVE INGREDIENT
Calendula Cream:
alcohol, caprylyl glycol, carbomer, cetyl palmitate, EDTA disodium, glycerin, lauroyl macrogolglycerides, pegoxol-7 stearate, purified water, sodium hydroxide, sorbic acid, 1,2-hexanediol
Calendula Cream (without disodium EDTA)
alcohol, caprylyl glycol, carbomer, cetyl palmitate, glycerin, lauroyl macrogolglycerides, pegoxol-7 stearate, purified water, sodium hydroxide, sorbic acid, 1,2-hexanediol
- QUESTIONS
-
SPL UNCLASSIFIED SECTION
do not use if pouch is torn or open
dispose of 1 day after opening (0.18 oz / 5g)
do not use if glued carton end flaps are open or if the tube seal is broken
store at 68-77° F (20-25° C)
Pouch 0.18 OZ (5g)
Tube 2.6 OZ (70 G)
Tube 0.5 OZ (14 G)
Plant-Powered Relief
Soothes, Moisturizes, Protects & Heals
Homeopathic mother tincture made from Calendula officinalis flowering tops.
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
**C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details. - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CALENDULA
calendula officinalis flowering top creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0220-1109 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALENDULA OFFICINALIS FLOWERING TOP (UNII: 18E7415PXQ) (CALENDULA OFFICINALIS FLOWERING TOP - UNII:18E7415PXQ) CALENDULA OFFICINALIS FLOWERING TOP 1 [hp_X] in 1 g Inactive Ingredients Ingredient Name Strength HYDROGENATED PALM/PALM KERNEL OIL PEG-6 ESTERS (UNII: 8EPU9MJ01K) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CARBOMER HOMOPOLYMER TYPE C (UNII: 4Q93RCW27E) CETYL PALMITATE (UNII: 5ZA2S6B08X) EDETATE DISODIUM (UNII: 7FLD91C86K) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBIC ACID (UNII: X045WJ989B) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) GLYCERIN (UNII: PDC6A3C0OX) PEGOXOL 7 STEARATE (UNII: 3EW5AXE5X5) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0220-1109-63 70 g in 1 TUBE; Type 0: Not a Combination Product 03/21/2006 2 NDC:0220-1109-68 1 in 1 BOX 02/29/2016 2 14 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:0220-1109-93 5 g in 1 POUCH; Type 0: Not a Combination Product 02/17/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/21/2006 Labeler - Laboratoires Boiron (282560473) Registrant - Boiron Inc. (014892269) Establishment Name Address ID/FEI Business Operations Boiron 282560473 manufacture(0220-1109)