Label: CONVENIA- cefovecin sodium injection, powder, lyophilized, for solution
- NDC Code(s): 54771-1520-1
- Packager: Zoetis Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated December 8, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- CAUTION
-
DESCRIPTION
Cefovecin sodium is a semi-synthetic broad-spectrum antibacterial agent from the cephalosporin class of chemotherapeutic agents. Cefovecin is the non-proprietary designation for (6R,7R)-7-[[(2Z)-(2-amino-4-thiazolyl)(methoxyimino)acetyl]amino]-8-oxo-3-[(2S)-tetrahydro-2-furanyl]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, monosodium salt.
Figure 1: Chemical structure of cefovecin sodium.
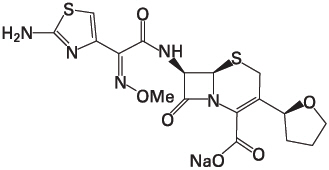
Each mL of CONVENIA reconstituted lyophile contains cefovecin sodium equivalent to 80.0 mg cefovecin, methylparaben 1.8 mg (preservative), propylparaben 0.2 mg (preservative), sodium citrate dihydrate 5.8 mg and citric acid monohydrate 0.1 mg, sodium hydroxide or hydrochloric acid as required to adjust pH.
- INDICATIONS
-
DOSAGE AND ADMINISTRATION
Dogs
CONVENIA should be administered as a single subcutaneous injection of 3.6 mg/lb (8 mg/kg) body weight. A second subcutaneous injection of 3.6 mg/lb (8 mg/kg) may be administered if response to therapy is not complete. The decision for a second injection for any individual dog should take into consideration such factors as progress toward clinical resolution, the susceptibility of the causative organisms, and the integrity of the dog's host-defense mechanisms. Therapeutic drug concentrations after the first injection are maintained for 7 days for S. intermedius infections and for 14 days for S. canis (Group G) infections. Maximum treatment should not exceed 2 injections.
Cats
CONVENIA should be administered as a single, one-time subcutaneous injection at a dose of 3.6 mg/lb (8 mg/kg) body weight. After an injection of CONVENIA, therapeutic concentrations are maintained for approximately 7 days for Pasteurella multocida infections.
General Dosing Information
A sample of the lesion should be obtained for culture and susceptibility testing prior to beginning antimicrobial therapy. Once results become available, continue with appropriate therapy. If acceptable response to treatment is not observed, or if no improvement is seen within 3 to 4 days, then the diagnosis should be re-evaluated and appropriate alternative therapy considered.
CONVENIA may persist in the body for up to 65 days. The effect of remaining concentrations of cefovecin on any subsequent antimicrobial therapies has not been determined. Fluoroquinolone and aminoglycoside antimicrobials have been reported to be compatible with cephalosporin antimicrobial agents.1,2,3
Table 1: Dose Table for CONVENIA at 8 mg/kg Body Weight Weight of Animal Volume of CONVENIA
(3.6 mg/lb or 0.045 mL/lb)5 lb 0.23 mL 10 lb 0.45 mL 15 lb 0.67 mL 20 lb 0.90 mL 40 lb 1.8 mL 80 lb 3.6 mL PREPARATION OF SOLUTION FOR INJECTION: To deliver the appropriate dose, aseptically reconstitute CONVENIA with 10 mL Sterile Water for Injection. Shake and allow vial to sit until all material is visually dissolved. The resulting solution contains cefovecin sodium equivalent to 80 mg/mL cefovecin. CONVENIA is light sensitive. The vial should be stored in the original carton and refrigerated when not in use. Use the entire contents of the vial within 56 days of reconstitution.
-
CONTRAINDICATIONS
CONVENIA is contraindicated in dogs and cats with known allergy to cefovecin or to β-lactam (penicillins and cephalosporins) group antimicrobials. Anaphylaxis has been reported with the use of this product in foreign market experience. If an allergic reaction or anaphylaxis occurs, CONVENIA should not be administered again and appropriate therapy should be instituted. Anaphylaxis may require treatment with epinephrine and other emergency measures, including oxygen, intravenous fluids, intravenous antihistamine, corticosteroids, and airway management, as clinically indicated. Adverse reactions may require prolonged treatment due to the prolonged systemic drug clearance (65 days).
-
WARNINGS
Not for use in humans. Keep this and all drugs out of reach of children. Consult a physician in case of accidental human exposure. For subcutaneous use in dogs and cats only. Antimicrobial drugs, including penicillins and cephalosporins, can cause allergic reactions in sensitized individuals. To minimize the possibility of allergic reactions, those handling such antimicrobials, including cefovecin, are advised to avoid direct contact of the product with the skin and mucous membranes.
-
PRECAUTIONS
Prescribing antibacterial drugs in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to treated animals and may increase the risk of the development of drug-resistant animal pathogens.
The safe use of CONVENIA in dogs or cats less than 4 months of age (see Animal Safety) and in breeding or lactating animals has not been determined. Safety has not been established for IM or IV administration. The long-term effects on injection sites have not been determined. CONVENIA is slowly eliminated from the body, approximately 65 days is needed to eliminate 97% of the administered dose from the body. Animals experiencing an adverse reaction may need to be monitored for this duration.
CONVENIA has been shown in an experimental in vitro system to result in an increase in free concentrations of carprofen, furosemide, doxycycline and ketoconazole. Concurrent use of these or other drugs that have a high degree of protein-binding (e.g. NSAIDs, propofol, cardiac, anticonvulsant, and behavioral medications) may compete with cefovecin binding and cause adverse reactions.
Positive direct Coombs' test results and false positive reactions for glucose in the urine have been reported during treatment with some cephalosporin antimicrobials. Cephalosporin antimicrobials may also cause falsely elevated urine protein determinations. Some antimicrobials, including cephalosporins, can cause lowered albumin values due to interference with certain testing methods.
Occasionally, cephalosporins and NSAIDs have been associated with myelotoxicity, thereby creating a toxic neutropenia4. Other hematological reactions seen with cephalosporins include neutropenia, anemia, hypoprothrombinemia, thrombocytopenia, prolonged prothrombin time (PT) and partial thromboplastin time (PTT), platelet dysfunction and transient increases in serum aminotransferases.
-
ADVERSE REACTIONS
Dogs
A total of 320 dogs, ranging in age from 8 weeks to 19 years, were included in a field study safety analysis. Adverse reactions reported in dogs treated with CONVENIA and the active control are summarized in Table 2.
Table 2: Number of Dogs* with Adverse Reactions Reported During the Field Study with CONVENIA Adverse Reaction CONVENIA
(n=157)Active Control
(n=163)- *
- Some dogs may have experienced more than one adverse reaction or more than one occurrence of the same adverse reaction during the study.
Lethargy 2 7 Anorexia/Decreased Appetite 5 8 Vomiting 6 12 Diarrhea 6 7 Blood in Feces 1 2 Dehydration 0 1 Flatulence 1 0 Increased Borborygmi 1 0 Mild to moderate elevations in serum gamma glutamyl transferase or serum alanine aminotransferase were noted post-treatment in several of the CONVENIA-treated dogs. No clinical abnormalities were noted with these findings.
One CONVENIA-treated dog in a separate field study experienced diarrhea post-treatment lasting four weeks. The diarrhea resolved.
Cats
A total of 291 cats, ranging in age from 2.4 months (one cat) to 21 years, were included in the field study safety analysis. Adverse reactions reported in cats treated with CONVENIA and the active control are summarized in Table 3.
Table 3: Number of Cats* with Adverse Reactions Reported During the Field Study with CONVENIA. Adverse Reaction CONVENIA
(n=147)Active Control
(n=144)- *
- Some cats may have experienced more than one adverse reaction or more than one occurrence of the same adverse reaction during the study.
Vomiting 10 14 Diarrhea 7 26 Anorexia/Decreased Appetite 6 6 Lethargy 6 6 Hyper/Acting Strange 1 1 Inappropriate Urination 1 0 Four CONVENIA cases had mildly elevated post-study ALT (one case was elevated pre-study). No clinical abnormalities were noted with these findings.
Twenty-four CONVENIA cases had normal pre-study BUN values and elevated poststudy BUN values (37 – 39 mg/dL post-study). There were 6 CONVENIA cases with normal pre- and mildly to moderately elevated post-study creatinine values. Two of these cases also had an elevated post-study BUN. No clinical abnormalities were noted with these findings.
One CONVENIA-treated cat in a separate field study experienced diarrhea post-treatment lasting 42 days. The diarrhea resolved.
FOREIGN MARKET EXPERIENCE: The following adverse events were reported voluntarily during post-approval use of the product in dogs and cats in foreign markets: death, tremors/ataxia, seizures, anaphylaxis, acute pulmonary edema, facial edema, injection site reactions (alopecia, scabs, necrosis, and erythema), hemolytic anemia, salivation, pruritus, lethargy, vomiting, diarrhea, and inappetance.
- CONTACT INFORMATION
-
CLINICAL PHARMACOLOGY
Pharmacokinetics
Cefovecin is rapidly and completely absorbed following subcutaneous administration. Non-linear kinetics is exhibited (plasma concentrations do not increase proportionally with dose). Cefovecin does not undergo hepatic metabolism and the majority of a dose is excreted unchanged in the urine. Elimination also occurs from excretion of unchanged drug in the bile. Cefovecin is a highly protein bound molecule in dog plasma (98.5%) and cat plasma (99.8%) and may compete with other highly protein bound drugs for plasma protein binding sites that could result in transient, higher free drug concentrations of either compound. Pharmacokinetic parameters following subcutaneous dosing at 8 mg/kg in the dog and cat are summarized in Table 4.
Table 4: Pharmacokinetic Parameters Reflecting Total Drug Concentrations in Plasma (mean ± standard deviation or range) Following an 8 mg/kg Intravenous or Subcutaneous Dose of Cefovecin in Dogs and Cats PARAMETER MEAN ± SD* or (Range) Dogs Cats † Terminal plasma elimination half-life, T1/2 (h)‡§ 133 ± 16 166 ± 18 AUC0-inf (μg∙h/mL)‡¶ 10400 ± 1900 † 22700 ± 3450 Time of maximum concentration, Tmax (h) ‡§ 6.2 (0.5-12.0) 2.0 (0.5-6.0) Maximum concentration, Cmax (μg/mL) ‡# 121 ± 51 141 ± 12 Vdss (L/kg) Þ¶ 0.122 ± 0.011 0.090 ± 0.010 CLtotal (mL/h/kg)Þ¶ 0.76 ± 0.13 † 0.350 ± 0.40 Population Pharmacokinetics
Dogs
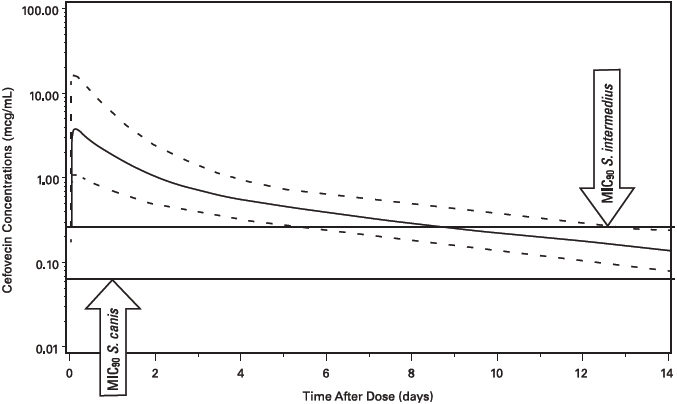
Cefovecin plasma concentrations in the dog have been characterized by the use of population pharmacokinetic (PPK) data. Plasma cefovecin concentration data were pooled from seven laboratory pharmacokinetic studies, each involving young, normal healthy Beagle dogs. The final dataset contained 591 concentration records from 39 dogs. The simulations from the model provide the mean population estimate and the 5th and 95th percentile of the population estimates of total and free cefovecin concentrations over time. Figure 2 shows the predicted free plasma concentrations following administration of 8 mg/kg body weight to dogs. Based upon these predicted concentrations, 95% of the canine population will have active (free) drug concentrations > the MIC90 of S. canis (0.06 µg/mL) for approximately 14 days and free concentrations > the MIC90 for S. intermedius (0.25 μg/mL) for approximately 7 days following a single 8 mg/kg subcutaneous injection of cefovecin. (See MICROBIOLOGY.)
Figure 2: Population Predicted Free Concentration of Cefovecin in Plasma Following a Single Subcutaneous Injection of 8 mg/kg Body Weight in Dogs (solid line is population prediction, dotted lines are the 5th and 95th percentiles for the population prediction).
Cats
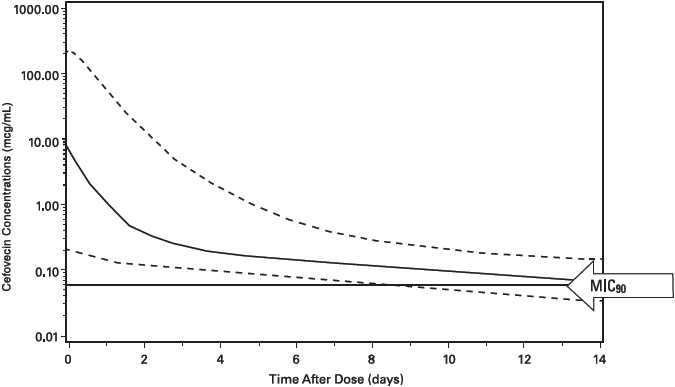
Cefovecin plasma concentrations in the cat have been characterized by the use of PPK data. Plasma cefovecin concentration data were pooled from four laboratory pharmacokinetic studies. The final dataset contained 338 concentration records from 22 cats. The simulations from the model provide the mean population estimate as well as the 5th and 95th percentile of the population estimates of total and free cefovecin concentrations over time. Figure 3 displays the predicted free plasma concentrations following administration of 8 mg/kg body weight to cats. Based upon these predicted concentrations, 95% of the feline population will have active (free) drug concentrations > the MIC90 of Pasteurella multocida (0.06 µg/mL) for approximately 7 days when administered a single 8 mg/kg subcutaneous injection of cefovecin. (See MICROBIOLOGY.)
Figure 3: Population Predicted Free Concentration of Cefovecin in Plasma Following a Single Subcutaneous Injection of 8 mg/kg Body Weight in Cats (solid line is population prediction, dotted lines are the 5th and 95th percentiles for the population prediction).
MICROBIOLOGY: CONVENIA is a cephalosporin antibiotic. Like other β-lactam antimicrobials, CONVENIA exerts its inhibitory effect by interfering with bacterial cell wall synthesis. This interference is primarily due to its covalent binding to the penicillin-binding proteins (PBPs) (i.e., transpeptidase and carboxypeptidase), which are essential for synthesis of the bacterial cell wall. For E. coli, the in vitro activity of CONVENIA is comparable to other cephalosporins, but due to the high-affinity protein-binding, the in vivo free concentration of cefovecin does not reach the MIC90 for E. coli (1.0 μg/mL). CONVENIA is not active against Pseudomonas spp. or enterococci.
Dogs
The minimum inhibitory concentration (MIC) values for cefovecin against label-claim pathogens isolated from skin infections in dogs enrolled in a 2001-2003 field effectiveness study are presented in Table 5. All MICs were determined in accordance with the Clinical and Laboratory Standards Institute (CLSI) standards.
Table 5. Activity of CONVENIA Against Pathogens Isolated from Dogs Treated With CONVENIA in Field Studies in the U.S. During 2001-2003. Disease Pathogen Microbiological Treatment Outcome Number of Isolates Sample Collection (Time Relative to Treatment) MIC50 µg/mL MIC90 µg/mL MIC Range µg/mL Skin infections Staphylococcus intermedius Success 44 Pre-Treatment 0.12 0.25 ≤ 0.06 - 2 Failure 4 Pre-Treatment 0.12 - 2 Streptococcus canis (Group G) Success 16 Pre-Treatment ≤ 0.06 ≤ 0.06 ≤ 0.06 Failure 2 Pre-Treatment ≤ 0.06 Cats
The MIC values for cefovecin against Pasteurella multocida isolated from skin infections (wounds and abscesses) in cats enrolled in a 2001-2003 field effectiveness study are presented in Table 6. All MICs were determined in accordance with the CLSI standards.
Table 6. Activity of CONVENIA Against Pathogens Isolated from Cats Treated With CONVENIA in Field Studies in the U.S. During 2001-2003. Disease Pathogen Microbiological Treatment Outcome Number of Isolates Sample Collection (Time Relative to Treatment) MIC50 µg/mL MIC90 µg/mL MIC Range µg/mL Skin infections Pasteurella multocida Success 57 Pre-Treatment ≤ 0.06 ≤ 0.06 ≤ 0.06 -0.12 Failure 1 Pre-Treatment ≤ 0.06 -
EFFECTIVENESS
Dogs
In a double-masked, 1:1 randomized canine field study conducted in the United States, the effectiveness of CONVENIA was compared to a cephalosporin active control. In this study, 320 dogs with superficial secondary pyoderma, abscesses, or infected wounds were treated with either a single injection of CONVENIA (n = 157) at 3.6 mg/lb (8 mg/kg) body weight or with an oral active control antibiotic (n = 163), administered twice daily for 14 days. In this study, dogs could receive a second course of therapy 14 days after the initial treatment. Of the 320 enrolled dogs, 22 of 157 dogs received two treatments of CONVENIA and 35 of 163 dogs received two courses of treatment with the active control. In the study, 118 of the 157 enrolled cases were evaluable for effectiveness for CONVENIA, and 117 of the 163 enrolled cases were evaluable for effectiveness of the active control antibiotic. CONVENIA was non-inferior to the active control. Table 7 summarizes the clinical success rates obtained 28 days after the initiation of the final course of therapy.
Table 7: Clinical Success Rates by Treatment Group 28 Days after the Initiation of the Final Course of Therapy. Type of Infection Dogs CONVENIA
(n=118)Active Control
(n=117)Skin (secondary superficial pyoderma, abscesses, and infected wounds) 109 (92.4%) 108 (92.3%) CONVENIA was administered concomitantly with other commonly used veterinary products such as heartworm preventatives, flea control products, sedatives/tranquilizers, anesthetic agents, routine immunizations, antihistamines, thyroid hormone supplementation, and non-steroidal anti-inflammatory drugs during the field study.
Cats
In a double-masked, 1:1 randomized cat field study conducted in the United States, the effectiveness of CONVENIA was compared to an active control. In this study, 291 cats with infected wounds or abscesses were treated with either a single injection of CONVENIA (n = 147) at 3.6 mg/lb (8 mg/kg) body weight or with an oral active control antibiotic (n = 144), administered once daily for 14 days. CONVENIA was non-inferior to the active control. The clinical success rates were obtained 28 days after the initiation of therapy and are presented in Table 8.
Table 8: Clinical Success Rates by Treatment Group 28 Days after the Initiation of Therapy. Type of Infection Cats CONVENIA
(n=89)Active Control
(n=88)Skin (wounds and abscesses) 86 (96.6%) 80 (90.9%) CONVENIA was used concomitantly with other commonly used veterinary products such as heartworm preventatives, flea control products, sedatives/tranquilizers, anesthetic agents, and vaccines during the field study.
-
ANIMAL SAFETY
Dogs
CONVENIA administered to healthy four month old dogs at doses of 12 mg/kg (1.5 ×), 36 mg/kg (4.5 ×), and 60 mg/kg (7.5 ×) every seven days by dorsoscapular subcutaneous injections was well-tolerated for a total of 5 doses. Vomiting and diarrhea were seen in all treatment groups, with the incidence of vomiting and the incidence and duration of diarrhea increasing in a dose-related manner. Injection site irritation and transient edema occurred with increasing frequency in a dose-related manner and with repeat injections. Two injection site reactions included a seroma over the shoulder and swelling lasting > 30 days. Dogs dosed at 36 mg/kg had a significant (p = 0.0088) increase in BUN (all means remained within the normal range) compared to the controls. One dog dosed at 60 mg/kg exhibited a glomerulopathy on histopathology, and one dog in this same group had minimal peliosis hepatis.
At an exaggerated dose of 180 mg/kg (22.5×) in dogs, CONVENIA caused some injection site irritation, vocalization and edema. Edema resolved within 8-24 hours.
Cats
CONVENIA administered to healthy four month old cats at doses of 12 mg/kg (1.5 ×), 36 mg/kg (4.5 ×), and 60 mg/kg (7.5 ×) every seven days by dorsoscapular subcutaneous injections was well tolerated for a total of 5 doses. Vomiting and diarrhea were observed in cats, with the incidence of vomiting, and the incidence and duration of diarrhea increasing in a dose-related manner. The mean albumin values for all the CONVENIA-treated cats were significantly lower (p ≤ 0.05) than the control values (all means remained within the normal range) for all time periods. The mean alkaline phosphatase values in the 60 mg/kg group were significantly higher (p ≤ 0.0291) than the control values for all time periods. Injection-site irritation and transient edema occurred with increasing frequency in a dose-related manner and with repeat injections. One cat in the 12 mg/kg group had a mild renal tubular and interstitial fibrosis, and one cat in the 12 mg/kg group had mild glomerulosclerosis on histopathology.
At an exaggerated dose of 180 mg/kg (22.5×), CONVENIA was associated with injection site irritation, vocalization and edema. Edema resolved within 8-24 hours. On Day 10, cats had lower mean white blood cell counts compared to the controls. One cat had a small amount of bilirubinuria on Day 10.
-
STORAGE INFORMATION
Store the powder and the reconstituted product in the original carton, refrigerated at 2° to 8° C (36° to 46° F). Use the entire contents of the vial within 56 days of reconstitution. PROTECT FROM LIGHT. After each use it is important to return the unused portion back to the refrigerator in the original carton. As with other cephalosporins, the color of the solution may vary from clear to amber at reconstitution and may darken over time. If stored as recommended, solution color does not adversely affect potency.
- HOW SUPPLIED
-
REFERENCES
1Pillai SK, Moellering RC, and Eliopoulos GM. 2005. Antimicrobial combinations, pp 365-440. In V. Lorian (ed.) Antibiotics in Laboratory Medicine, 5th ed., Lippincott, Williams, and Wilkins, Philadelphia, PA.
2Fish DN, Choi MK, and Jung R: Synergic activity of cephalosporins plus fluoroquinolones against Pseudomonas aeruginosa with resistance to one or both drugs. Journal of Antimicrobial Chemotherapy (2002) 50, 1045–1049.
3Mayer I and Nagy E: Investigation of the synergic effects of aminoglycoside-fluoroquinolone and third-generation cephalosporin combinations against clinical isolates of Pseudomonas spp. Journal of Antimicrobial Chemotherapy (1999) 43, 651–657.
4Birchard SJ and Sherding RG. Saunders Manual of Small Animal Practice, 2nd edition. W.B. Saunders Co. 2000: p. 166.
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton
-
INGREDIENTS AND APPEARANCE
CONVENIA
cefovecin sodium injection, powder, lyophilized, for solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:54771-1520 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CEFOVECIN SODIUM (UNII: DL8Q24959P) (CEFOVECIN - UNII:0D1OL46ZIE) CEFOVECIN 80 mg in 1 mL Inactive Ingredients Ingredient Name Strength METHYLPARABEN (UNII: A2I8C7HI9T) 1.8 mg in 1 mL PROPYLPARABEN (UNII: Z8IX2SC1OH) 0.2 mg in 1 mL SODIUM CITRATE (UNII: 1Q73Q2JULR) 5.8 mg in 1 mL CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) 0.1 mg in 1 mL SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54771-1520-1 10 mL in 1 VIAL, GLASS Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141285 04/25/2008 Labeler - Zoetis Inc. (828851555)


