Label: SABIWAY, HAIR RE-GROWTH (rosemary- rosmarinus officinalis leaf extract shampoo
-
Contains inactivated NDC Code(s)
NDC Code(s): 70527-001-01 - Packager: Navarrete Estacio Fernando
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 9, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
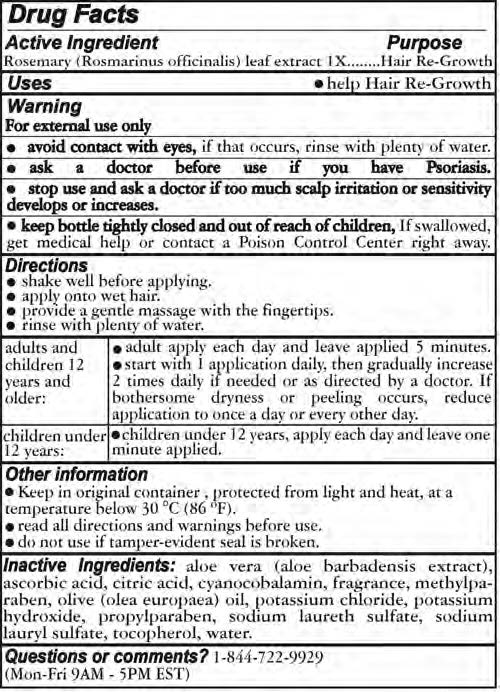
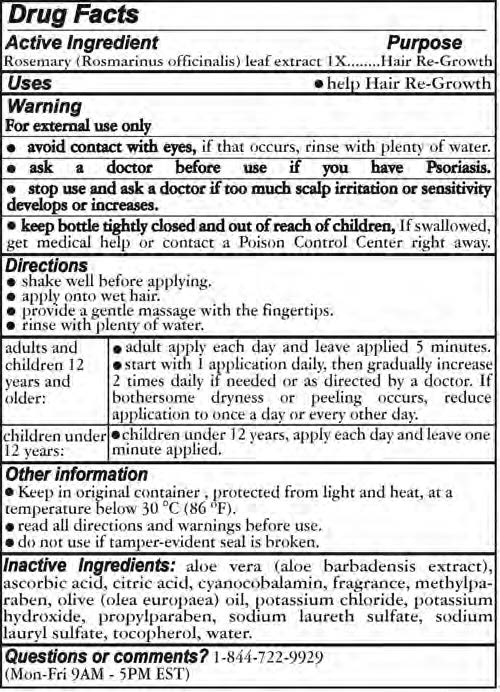
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
-
DOSAGE & ADMINISTRATION
Directions
- Shake well before applying
- apply onto wet hair
- provide a gentle massage with teh fingertips
- rinse with plenty of water
Adults and Childrebn 12 years and older
- adult apply each day and leave applied 5 minutes
- start with 1 application daily, then gradually increase 2 times daily if needed or as directed by a doctor. If bothersome dryness or peeling occurs, reduce application to once a day or every other day
Children under 12 years
- Children under 12 years, apply each day and leave one minute applied
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SABIWAY, HAIR RE-GROWTH
rosemary (rosmarinus officinalis) leaf extract shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70527-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ROSEMARY (UNII: IJ67X351P9) (ROSEMARY - UNII:IJ67X351P9) ROSEMARY 1 [hp_X] in 10 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) TOCOPHEROL (UNII: R0ZB2556P8) METHYLPARABEN (UNII: A2I8C7HI9T) OLIVE OIL (UNII: 6UYK2W1W1E) POTASSIUM CHLORIDE (UNII: 660YQ98I10) ASCORBIC ACID (UNII: PQ6CK8PD0R) ALOE VERA LEAF (UNII: ZY81Z83H0X) CYANOCOBALAMIN (UNII: P6YC3EG204) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) PROPYLPARABEN (UNII: Z8IX2SC1OH) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM LAURETH SULFATE (UNII: BPV390UAP0) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70527-001-01 500 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/09/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/09/2016 Labeler - Navarrete Estacio Fernando (880278943) Establishment Name Address ID/FEI Business Operations Navarrete Estacio Fernando 880278943 manufacture(70527-001)