Label: ONEVITE tablet
- NHRIC Code(s): 70898-198-09
- Packager: NEOMED PHARMACEUTICAL
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated June 14, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- STATEMENT OF IDENTITY
-
WARNINGS
INDICATIONS AND USAGE
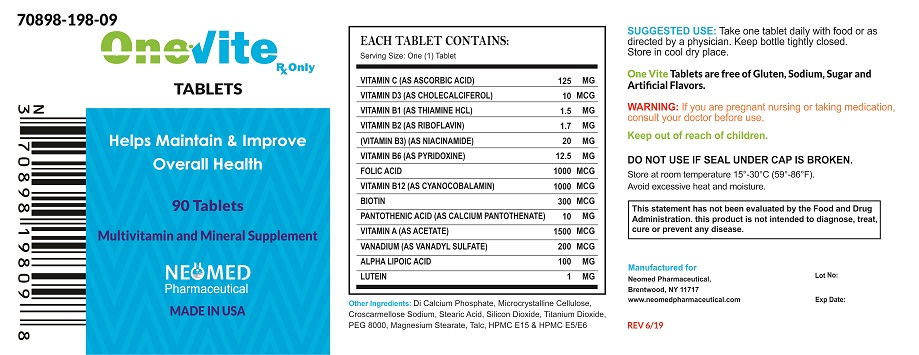
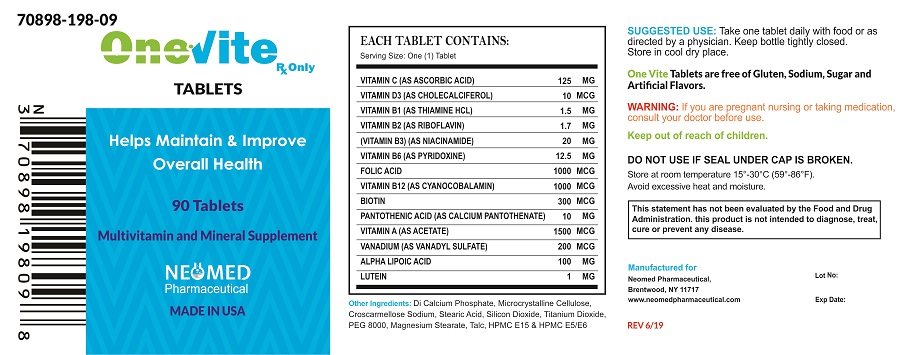
ONEvite Tablets is an orally administered prescription vitamin supplement specifically formulated for the clinical dietary management of patients with unique nutritional needs requiring, increased folate levels and nutritional supplementation in physiologically stressful conditions. As well as for maintenance of good health and beneficial in maintaining overall skin health, also help maintain healthy blood sugar levels and help promote nerve health and function including normal vision.
CONTRAINDICATIONS
ONEvite Tablets is contraindicated in patients with hypersensitivity to any of its components. Folic Acid is contraindicated in patients with untreated and uncomplicated pernicious anemia, and in those with anaphylactic sensitivity to folic acid.
Cyanocobalamin is contraindicated in patients with sensitivity to cobalt or to cyanocobalamin (Vitamin B12).
WARNING/PRECAUTIONS
Vitamin D supplementation should be used with caution in those with hypercalcemia or conditions that may lead to hypercalcemia such as hyperparathyroidism and those who form calcium-containing kidney stones. High doses of vitamin D can lead to elevated levels of calcium that reside in the blood and soft tissues. Bone pain, high blood pressure, formation of kidney stones, renal failure, and increased risk of heart disease can occur.
Folic acid, especially in doses above 0.1 mg daily, may obscure pernicious anemia, in that hematologic remission may occur while neurological manifestations remain progressive.
The use of folic acid doses above 1 mg daily may precipitate or exacerbate the neurological damage of vitamin B12 deficiency.
Avoid Over dosage. Keep out of the reach of children.
Drug Interactions
High doses of folic acid may result in decreased serum levels of anticonvulsant drugs.
Vitamin D supplementation should not be given with large amounts of calcium in those with hypercalcemia or conditions that may lead to hypercalcemia such as hyperparathyroidism and those who form calcium-containing kidney stones.
Consult appropriate references for additional specific vitamin-drug interactions.
Information for Patients
Patients should be counseled to disclose all medical conditions, including use of all medications, vitamins and supplements, pregnancy, and breastfeeding.
Pediatric Use
Not recommended for pediatric use.
ADVERSE REACTIONS
Adverse reactions have been reported with specific vitamins and minerals, but generally at levels substantially higher than those in ONEvite Tablets
DOSAGE AND ADMINISTRATION
ONEvite Tablets is a white film coating with caplet shape.
One tablet daily or as directed by a physician.
-
HEALTH CLAIM
HOW SUPPLIED
ONEvite Tablets available 90 tablets per bottle,
70898-198-09
Store at room temperature 15°-30°C (59°-86°F). Avoid excessive heat and moisture.
To report a serious adverse event or obtain product information, call 631-524-5758
Manufactured for:
Neomed Pharmaceutical
Brentwood, NY 11717
www.neomedpharmaceutical.com
MADE IN USA
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ONEVITE
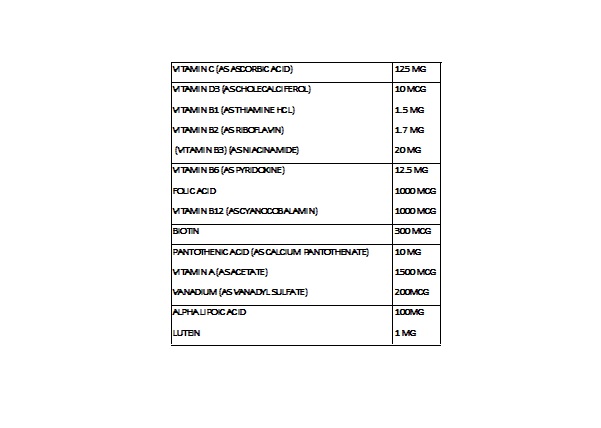
onevite tabletProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:70898-198 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 125 mg CHOLECALCIFEROL (UNII: 1C6V77QF41) (CHOLECALCIFEROL - UNII:1C6V77QF41) CHOLECALCIFEROL 10 ug THIAMINE HYDROCHLORIDE (UNII: M572600E5P) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE HYDROCHLORIDE 1.5 mg RIBOFLAVIN (UNII: TLM2976OFR) (RIBOFLAVIN - UNII:TLM2976OFR) RIBOFLAVIN 1.7 mg NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 20 mg PYRIDOXINE (UNII: KV2JZ1BI6Z) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE 12.5 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 1000 ug CYANOCOBALAMIN (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) CYANOCOBALAMIN 1000 ug BIOTIN (UNII: 6SO6U10H04) (BIOTIN - UNII:6SO6U10H04) BIOTIN 300 ug CALCIUM PANTOTHENATE (UNII: 568ET80C3D) (PANTOTHENIC ACID - UNII:19F5HK2737) PANTOTHENIC ACID 10 mg VITAMIN A ACETATE (UNII: 3LE3D9D6OY) (VITAMIN A - UNII:81G40H8B0T) VITAMIN A 1500 ug VANADYL SULFATE (UNII: 6DU9Y533FA) (VANADIUM - UNII:00J9J9XKDE) VANADYL SULFATE 200 ug .ALPHA.-LIPOIC ACID (UNII: 73Y7P0K73Y) (.ALPHA.-LIPOIC ACID - UNII:73Y7P0K73Y) .ALPHA.-LIPOIC ACID 100 mg LUTEIN (UNII: X72A60C9MT) (LUTEIN - UNII:X72A60C9MT) LUTEIN 1 mg Inactive Ingredients Ingredient Name Strength CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: L11K75P92J) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) STEARIC ACID (UNII: 4ELV7Z65AP) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) MAGNESIUM STEARATE (UNII: 70097M6I30) TALC (UNII: 7SEV7J4R1U) HYPROMELLOSES (UNII: 3NXW29V3WO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:70898-198-09 1 in 1 PACKAGE 1 90 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 12/01/2018 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color scoring 1 shape size (solid drugs) 4 mm Labeler - NEOMED PHARMACEUTICAL (048388130)