Label: FELIMAZOLE- methimazole tablet, sugar coated
- NDC Code(s): 17033-225-10, 17033-250-10
- Packager: Dechra Veterinary Products
- Category: PRESCRIPTION ANIMAL DRUG LABEL
Drug Label Information
Updated December 22, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
DESCRIPTION:
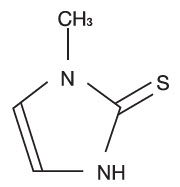
Methimazole is a thioureylene antithyroid drug, which inhibits the synthesis of thyroid hormones. Methimazole (1-methylimidazole-2-thiol) is a white, crystalline substance that is freely soluble in water. The chemical formula is C4H6N2S. Molecular weight is 114.16.
Methimazole Chemical Structure:
- VETERINARY INDICATIONS
-
DOSAGE & ADMINISTRATION
DOSAGE AND ADMINISTRATION:
The starting dose of Felimazole Coated Tablets is 2.5 mg administered every 12 hours. Following 3 weeks of treatment, the dose should be titrated to effect based on individual serum total T4 (TT4) levels and clinical response. Dose adjustments should be made in 2.5 mg increments. The maximum total dosage is 20 mg per day divided, not to exceed 10 mg as a single administration.
Hematology, biochemistry, and TT4 should be evaluated prior to initiating treatment and monitored after 3 weeks and 6 weeks of treatment. Thereafter, bloodwork should be monitored every 3 months and the dose adjusted as necessary. Cats receiving doses greater than 10 mg per day should be monitored more frequently.
-
CONTRAINDICATIONS
CONTRAINDICATIONS:
Do not use in cats with hypersensitivity to methimazole, carbimazole or the excipient, polyethylene glycol.
Do not use in cats with primary liver disease or renal failure.
Do not use in cats with autoimmune disease. See ADVERSE REACTIONS.
Do not use in cats with hematological disorders (such as anemia, neutropenia, lymphopenia, or thrombocytopenia) or coagulopathies. See ADVERSE REACTIONS.
Do not use in pregnant or lactating queens. Laboratory studies in rats and mice have shown evidence of teratogenic and embryotoxic effects of methimazole.
-
WARNINGS
WARNINGS:
Methimazole has anti-vitamin K activity and may induce bleeding diathesis without evidence of thrombocytopenia. See ADVERSE REACTIONS.
-
SAFE HANDLING WARNING
HUMAN WARNINGS:
Not for use in humans. Keep out of reach of children. For use in cats only. Wash hands with soap and water after administration to avoid exposure to drug. Do not break or crush tablets. Wear protective gloves to prevent direct contact with litter, feces, urine, or vomit of treated cats, and broken or moistened tablets. Wash hands after contact with the litter of treated cats.
Methimazole is a human teratogen and crosses the placenta concentrating in the fetal thyroid gland. There is also a high rate of transfer into breast milk. Pregnant women or women who may become pregnant, and nursing mothers should wear gloves when handling tablets, litter or bodily fluids of treated cats.
Methimazole may cause vomiting, gastric distress, headache, fever, arthralgia, pruritus, and pancytopenia. In the event of accidental ingestion/overdose, seek medical advice immediately and show the product label to the physician.
-
PRECAUTIONS
PRECAUTIONS:
Use of Felimazole Coated Tablets in cats with renal dysfunction should be carefully evaluated. Reversal of hyperthyroidism may be associated with decreased glomerular filtration rate and a decline in renal function, unmasking the presence of underlying renal disease. Due to potentially serious adverse reactions such as hepatopathy, immune-mediated anemia, thrombocytopenia, and agranulocytosis, cats on methimazole therapy should be monitored closely for any sign of illness including anorexia, vomiting, head/facial pruritus or edema, depression/lethargy, weight loss, anemia, skin lesions, diarrhea, fever, or lymphadenopathy. If a cat becomes ill while on Felimazole Coated Tablets, the drug should be stopped and appropriate hematological and biochemical testing should be done (see ANIMAL SAFETY and POST APPROVAL EXPERIENCE).
Anticoagulants may be potentiated by the anti-vitamin K activity of Felimazole Coated Tablets. Concurrent use of phenobarbital may reduce the clinical effectiveness of Felimazole Coated Tablets.
A reduction in dose of certain drugs (β-adrenergic blocking agents, digitalis glycosides, and theophylline) may be needed when the patient becomes euthyroid.
Methimazole is known to reduce the hepatic oxidation of benzimidazole anthelmintics (e.g. fenbendazole), leading to increased plasma concentration of these anthelmintics when administered concurrently.
Felimazole Coated Tablets caused delayed maturation of the testes in young male cats in the 12-week safety study. See ANIMAL SAFETY. The safety of Felimazole Coated Tablets has not been evaluated in male cats intended for breeding.
-
ADVERSE REACTIONS
ADVERSE REACTIONS:
In a US field study with 113 cats, the most common adverse reactions included change in food consumption (increase or decrease), lethargy, vomiting, diarrhea/loose stool, skin lesions, and abnormal vocalization. Three cats were withdrawn early from the study, one due to unmasking of latent renal disease and two due to the development of skin lesions. Over the course of the study, there was a decreasing trend in the mean counts of red blood cells, lymphocytes, neutrophils and monocytes; however, means remained within or near normal ranges for the testing laboratory.
In the extended use phase of the US field study with 101 cats, the most common adverse reactions reported in the study above (lethargy, anorexia) were also observed. Additional signs occurring more frequently in the long-term study were: depression/withdrawn behavior, weight loss, haircoat abnormalities, increased blood urea nitrogen (BUN), weakness, agitation and diarrhea. Most of the adverse reactions reported were mild and transient.
Serum chemistry and hematology results in the extended use study were consistent with the trends noted during the field study. The mean alanine transaminase (ALT) was above the reference range at the first two quarterly visits, but within the normal reference range (10-100 U/L) through the next two quarterly visits. Mean lymphocyte counts decreased consistently during the study period, to slightly below the reference range (1200-8000 cells/mcL) at the fourth quarterly visit.
Sixteen cats experienced elevated antinuclear antibody (ANA) titers at one or more points during long-term therapy with Felimazole Coated Tablets but the significance was not determined. Eighteen cats died or were euthanized during the extended use study, four of which may have been related to Felimazole Coated Tablets due to the unmasking/acceleration of chronic renal failure. See PRECAUTIONS.
In a foreign field study with 26 cats using a starting dose of 5 mg twice daily (twice the recommended starting dose), one cat was withdrawn due to lethargy, vomiting and facial excoriations. Marked thrombocytopenia was reported in two cats; the platelet count returned to normal in one cat when Felimazole Coated Tablets® (methimazole tablets) were discontinued. Two cats collapsed and died within 12 days of starting Felimazole Coated Tablets at a dose of 5 mg twice daily. Both cats were reported with lethargy, vomiting, anorexia, and bloody diarrhea; one cat also had pallor.
In a second foreign field study with 78 cats using a starting dose of 2.5 mg twice daily, 4 cats were withdrawn due to suspected adverse reactions to Felimazole Coated Tablets including anemia, cholangiohepatitis, excoriations, vomiting, lethargy, jaundice, and anorexia. One cat receiving 2.5 mg three times daily collapsed and died after 8 weeks of treatment. Adverse reactions included pallor, anorexia, dehydration, jaundice, bleeding diathesis, and anemia. The most frequently reported adverse reactions included mild, transient, self-limiting vomiting, lethargy, and anorexia.
If overdosage occurs, stop treatment and give symptomatic and supportive care.
Foreign Market Experience: The following events were reported voluntarily during post-approval use of Felimazole Coated Tablets in foreign markets: facial pruritus, self-induced excoriations of the head and neck, generalized lymphadenopathy, thrombocytopenia, hematemesis, epistaxis, and elevation of serum liver enzymes and bilirubin.
POST APPROVAL EXPERIENCE (revised 2015):
The following adverse events are based on voluntary, post approval reporting. Not all adverse events are reported to FDA/CVM. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using these data. The following adverse events are listed in decreasing order of reporting frequency: Anorexia, vomiting, head/facial pruritus or edema, depression/lethargy, weight loss, anemia, elevated liver enzymes, skin lesions, elevated BUN, diarrhea, and thrombocytopenia. In some reported cases, the patients recovered after adverse signs were recognized, the drug was withdrawn, and veterinary care was applied. In some cases, death (or euthanasia) has been reported as an outcome of the adverse reactions listed above.
CONTACT INFORMATION:
To report suspected adverse events, for technical assistance or to obtain a copy of the Safety Data Sheet (SDS), contact Dechra at (866) 933-2472.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS, or online at http://www.fda.gov/reportanimalae
-
CLINICAL PHARMACOLOGY
CLINICAL PHARMACOLOGY:
Methimazole is an antithyroid drug that acts by blocking the biosynthesis of thyroid hormone in vivo. The primary action is to inhibit binding of iodide to the enzyme thyroid peroxidase, thereby preventing the catalyzed iodination of thyroglobulin and T3 and T4 synthesis.
Felimazole Coated Tablets are well absorbed following oral administration. Maximum plasma concentrations are achieved within 1-1½ hours after dosing and methimazole is rapidly eliminated from the blood (T½ is approximately 3 hours). Administration of Felimazole Coated Tablets in a fasted state enhances absorption.
-
SPL UNCLASSIFIED SECTION
EFFECTIVENESS:
In a US effectiveness field study with 113 cats, the product was considered effective if both the TT4 concentration was ≤ 4.0 mcg/dL, and the Investigator's clinical assessment documented clinical improvement. Of the 105 evaluable cases, 64 (61%) were considered treatment successes. The decrease in TT4 concentration was significant from the pre-enrollment visit to the Day 42 visit. A TT4 of ≤ 4.0 mcg/dL occurred in 59.1% and 61.9% of cats on Day 21 and Day 42, respectively. Investigators assessed 91.8% and 87.6% of cats as clinically improved on Days 21 and 42, respectively.
In the extended use phase of the US effectiveness field study with 101 cats, effectiveness was based on a combination of Investigator's clinical assessment, maintenance of TT4 concentrations at or near the laboratory reference range of 0.8-4.0 mcg/dL, and the presence or absence of adverse reactions. Mean TT4 concentrations were within or near the laboratory reference range during the first four quarterly visits. At the first quarterly visit, Investigators categorized 80.9% of cats as stable or improved relative to their baseline assessment. By the fourth quarterly visit, 75.8% were deemed to be stable or improved.
The average maintenance dose required in the extended use phase was 2.5 mg twice daily, with a minimum of 2.5 mg per cat and a maximum of 15 mg per cat on a daily basis.
-
SPL UNCLASSIFIED SECTION
ANIMAL SAFETY:
ANIMAL SAFETY STUDIES: In a 12-week safety study, healthy young cats were dosed with 0, 10, 20, and 30 mg Felimazole Coated Tablets per day, divided into two doses. Cats in all treated groups experienced anorexia, vomiting, loose stool and lethargy. Cats in the 20 and 30 mg/day groups also had facial excoriations, pruritus, and lymphadenopathy. The following hematological changes were seen: neutropenia, lymphopenia, anemia, and thrombocytopenia. The following biochemical changes were seen: increased globulin, increased magnesium, increased blood urea nitrogen, increased creatinine and decreased phosphorus. There was a dose-dependent occurrence of antinuclear antibodies. Most of the clinical pathology changes were mild in nature. One cat dosed with 20 mg/day experienced a six-fold increase in ALT during the study. This cat had loose stool, but was otherwise healthy throughout the study. Hepatomegaly was seen in this cat at necropsy and the histopathological examination was comparable to other treated cats with hepatomegaly and normal ALT.
Gross necropsy findings in all treated groups included hepatomegaly, thymus atrophy and thyroid hyperplasia and darkening. Some treated males had delayed maturation of the testes.
The 30 mg/day dose was poorly tolerated and resulted in the clinical deterioration and euthanasia of four of the six cats in that group. Two of the cats showed signs of immune-mediated hemolytic anemia, thrombocytopenia and severe clinical deterioration. One had been on the drug for 34 days, the other for 9 weeks. The drug was discontinued in a third cat treated with 30 mg/day while it received supportive care.
It was euthanized on day 55 after becoming anorexic. This cat had anemia (HCT 21.6%) and red blood cell agglutination. Necropsy showed inflammation of the muscular layer of the stomach and a small erosion in the stomach. A fourth cat treated with 30 mg/day was euthanized after several days of anorexia when the decision was made to discontinue dosing in this group. All 30 mg/day cats that died had generalized lymphadenopathy. Necropsies revealed reactive lymph nodes and varying degrees of inflammation throughout the body. The remaining 2 cats in the 30 mg/day group were taken off Felimazole Coated Tablets at week 9 and fully recovered.
- STORAGE AND HANDLING
- HOW SUPPLIED
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 2.5 mg Tablet Bottle Carton
- PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Carton
-
INGREDIENTS AND APPEARANCE
FELIMAZOLE
methimazole tablet, sugar coatedProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:17033-225 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength methimazole (UNII: 554Z48XN5E) (methimazole - UNII:554Z48XN5E) methimazole 2.5 mg Product Characteristics Color PINK Score no score Shape ROUND Size 6mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17033-225-10 1 in 1 CARTON 1 100 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141292 06/15/2009 FELIMAZOLE
methimazole tablet, sugar coatedProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:17033-250 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength methimazole (UNII: 554Z48XN5E) (methimazole - UNII:554Z48XN5E) methimazole 5 mg Product Characteristics Color ORANGE Score no score Shape ROUND Size 6mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17033-250-10 1 in 1 CARTON 1 100 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141292 06/15/2009 Labeler - Dechra Veterinary Products (362142734) Registrant - Dechra Ltd (641097493)





