Label: TRIDERM- triamcinolone acetonide cream
-
NDC Code(s):
0316-0165-15,
0316-0165-16,
0316-0165-85,
0316-0170-01, view more0316-0170-02, 0316-0170-03, 0316-0175-15, 0316-0175-16
- Packager: Crown Laboratories
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 24, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
The topical corticosteroids constitute a class of primarily synthetic steroids used as anti-inflammatory and antipruritic agents. Triamcinolone acetonide is a member of this class. Triamcinolone acetonide is chemically pregna-1,4-diene-3,20-dione, 9-fluoro-11,21-dihydroxy-16,17-[(1-methylethylidene)bis-(oxy)]-(11β, 16α)- with the empirical formula C 24H 31FO 6 and molecular weight 434.50. Its structural formula is:
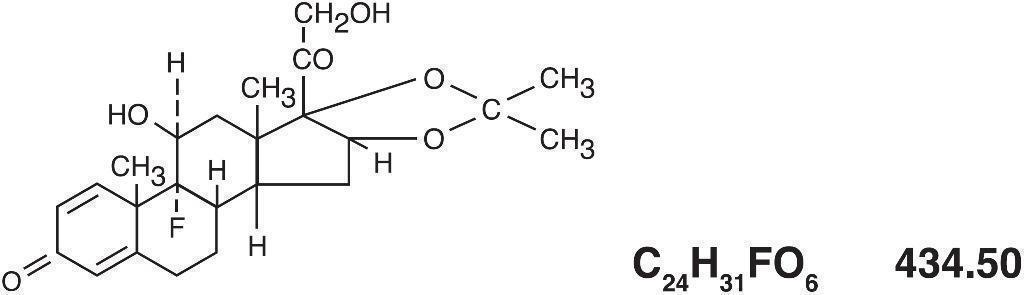
Each gram of Triderm (Triamcinolone Acetonide Cream USP), 0.025% contains 0.25 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
Each gram of Triderm (Triamcinolone Acetonide Cream USP), 0.1% contains 1 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
Each gram of Triderm (Triamcinolone Acetonide Cream USP), 0.5% contains 5 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate. -
CLINICAL PHARMACOLOGY
Topical corticosteroids share anti-inflammatory, antipruritic and vasoconstrictive actions.
The mechanism of anti-inflammatory activity of the topical corticosteroids is unclear. Various laboratory methods, including vasoconstrictor assays, are used to compare and predict potencies and/or clinical efficacies of the topical corticosteroids. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man.
Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings.
Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids. Thus, occlusive dressings may be a valuable therapeutic adjunct for treatment of resistant dermatoses. (See DOSAGE AND ADMINISTRATION).
Once absorbed through the skin, topical corticosteroids are handled through pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees. Corticosteroids are metabolized primarily in the liver and are then excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile. - INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
PRECAUTIONS
General
Systemic absorption of topical corticosteroids has produced reversible hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome, hyperglycemia, and glucosuria in some patients.
Conditions which augment systemic absorption include the application of the more potent steroids, use over large surface areas, prolonged use, and the addition of occlusive dressings.
Therefore, patients receiving a large dose of a potent topical steroid applied to a large surface area or under an occlusive dressing should be evaluated periodically for evidence of HPA axis suppression by using the urinary free cortisol and ACTH stimulation tests. If HPA axis suppression is noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid.
Recovery of HPA axis function is generally prompt and complete upon discontinuation of the drug. Infrequently, signs and symptoms of steroid withdrawal may occur, requiring supplemental systemic corticosteroids. Pediatric patients may absorb proportionally larger amounts of topical corticosteroids and thus be more susceptible to systemic toxicity (See PRECAUTIONS-Pediatric use).
If irritation develops, topical corticosteroids should be discontinued and appropriate therapy instituted.
In the presence of dermatological infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, the corticosteroid should be discontinued until the infection has been adequately controlled. -
Information for the Patient
Patients using topical corticosteroids should receive the following information and instructions:
1. This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes.
2. Patients should be advised not to use this medication for any disorder other than for which it was prescribed.
3. The treated skin area should not be bandaged or otherwise covered or wrapped as to be occlusive unless directed by the physician.
4. Patients should report any signs of local adverse reactions especially under occlusive dressing.
5. Parents of pediatric patients should be advised not to use tight-fitting diapers or plastic pants on a child being treated in the diaper area, as these garments may constitute occlusive dressings. - Laboratory Tests
- Carcinogenesis, Mutagenesis, and Impairment of Fertility
-
Pregnancy Category C
Corticosteroids are generally teratogenic in laboratory animals when administered systemically at relatively low dosage levels. The more potent corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. There are no adequate and well-controlled studies in pregnant women on teratogenic effects from topically applied corticosteroids. Therefore, topical corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Drugs of this class should not be used extensively on pregnant patients, in large amounts, or for prolonged periods of time.
-
Nursing Mothers
It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in breast milk. Systemically administered corticosteroids are secreted into breast milk in quantities not likely to have a deleterious effect on the infant. Nevertheless, caution should be exercised when topical corticosteroids are administered to a nursing woman.
-
Pediatric Use
Pediatric patients may demonstrate greater susceptibility to topical corticosteroid-induced HPA axis suppression and Cushing’s syndrome than mature patients because of a larger skin surface area to body weight ratio.
Hypothalamic-pituitary-adrenaI (HPA) axis suppression, Cushing’s syndrome, and intracranial hypertension have been reported in pediatric patients receiving topical corticosteroids. Manifestations of adrenal suppression in pediatric patients include linear growth retardation, delayed weight gain, low plasma cortisol levels, and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
Administration of topical corticosteroids to pediatric patients should be limited to the least amount compatible with an effective therapeutic regimen. Chronic corticosteroid therapy may interfere with the growth and development of pediatric patients. -
ADVERSE REACTIONS
The following local adverse reactions are reported infrequently with topical corticosteroids, but may occur more frequently with the use of occlusive dressings. These reactions are listed in an approximate decreasing order of occurrence: burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infection, skin atrophy, striae, miliaria.
-
OVERDOSAGE
Topically applied corticosteroids can be absorbed in sufficient amounts to produce systemic effects. (See PRECAUTIONS)
-
DOSAGE AND ADMINISTRATION
Apply to the affected area as a thin film from two to four times daily for the 0.025% strength and two or three times daily for the 0.1% and 0.5% strength depending on the severity of the condition.
Occlusive dressings may be used for the management of psoriasis or recalcitrant conditions.
If an infection develops, the use of occlusive dressings should be discontinued and appropriate antimicrobial therapy instituted. -
HOW SUPPLIED
Triderm (Triamcinolone Acetonide Cream USP), 0.025% is supplied in:
15 grams tube NDC 0316-0165-15
85.2 grams tube NDC 0316-0165-85
454 grams jar NDC 0316-0165-16
Triderm (Triamcinolone Acetonide Cream USP), 0.1% is supplied in:
28.4 grams tube NDC 0316-0170-0185.2 grams tube NDC 0316-0170-03
Triderm (Triamcinolone Acetonide Cream USP), 0.5% is supplied in:
15 grams tube NDC 0316-0175-15
454 grams jar NDC 0316-0175-16
- STORAGE
- SPL UNCLASSIFIED SECTION
-
Triderm (Triamcinolone Acetonide Cream USP), 0.1% tube label
NDC 0316-0170-01
Rx Only
TRIDERM TM
Triamcinolone Acetonide Cream USP, 0.1%
1 oz (28.4 grams)
WARNING: Keep Out Of Reach Of Children.
For external use only.
Not for ophthalmic use.
.
Each gram contains: 1 mg Triamcinolone Acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
Usual Dosage: 2 to 3 applications daily. See package insert for full prescribing information.
TO OPEN: Use cap to puncture seal.
IMPORTANT: Do not use if seal has been punctured or is not visible.
Store at 20 o-25 oC (68 o-77 oF) [see USP Controlled Room Temperature]. Avoid excessive heat. Protect from freezing.
See Crimp of tube for Expiration Date and Batch Number.
Manufactured and Distributed by:
Crown Laboratories, Inc., Johnson City, TN 37604

-
Triderm (Triamcinolone Acetonide Cream USP), 0.1% carton label
NDC 0316-0170-01
Rx Only
TRIDERM TM
Triamcinolone Acetonide Cream USP, 0.1%1 oz (28.4 grams)
WARNING: Keep Out Of Reach Of Children.
For External Use Only.
Not for Ophthalmic Use.Each gram contains: 1 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
Directions for puncturing tube seal: Remove cap. Turn cap upside down and place puncture tip onto tube seal: push down until seal is punctured. Screw cap back on to close.
IMPORTANT: Do not use if seal has been punctured or is not visible.
Store at 20 o-25 oC (68 o-77 oF) [see USP Controlled Room Temperature].
Avoid excessive heat. Protect from freezing.
Manufactured and Distributed by: Crown Laboratories, Inc. Johnson City, TN 37604
Usual Dosage: 2 to 3 applications daily. See package insert for full prescribing information.
See end of carton for batch number and expiration date.

-
Triderm (Triamcinolone Acetonide Cream USP), 0.025% tube label
NDC 0316-0165-15
Rx Only
TRIDERM CREAM TM
Triamcinolone Acetonide Cream USP, 0.025%
15 grams
WARNING: Keep out of reach of children
For External Use Only
Not for Ophthalmic UseStore at 20 o-25 oC (68 o-77 oF) [see USP Controlled Room Temperature]
Avoid excessive heat. Protect from freezing.
Each gram contains 0.25 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
USUAL DOSAGE: 2 or 4 applications daily. See package insert for full prescribing information.
TO OPEN: Use cap to puncture seal.
IMPORTANT: Do not use if seal has been punctured or is not visible. See crimp of tube for batch number and expiration date.
Manufactured and Distributed by:
Crown Laboratories, Inc.
Johnson City, TN 37604

-
Triderm (Triamcinolone Acetonide Cream USP), 0.025% carton label
NDC 0316-0165-15
Rx Only
TRIDERM CREAM TM
Triamcinolone Acetonide Cream USP, 0.025%
15 grams
WARNING: Keep out of reach of children
For External Use Only
Not for Ophthalmic UseUSUAL DOSAGE: 2 or 4 applications daily. See package insert for full prescribing information.
See end of carton for batch number and expiration date.
Each gram contains 0.25 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
Directions for puncturing seal: Remove cap. Turn cap upside down and place puncture tip onto tube seal; push down until seal is punctured. Screw cap back on to close
IMPORTANT: Do not use if seal has been punctured or is not visible.
Store at 20 o-25 oC (68 o-77 oF) [see USP Controlled Room Temperature]
Avoid excessive heat. Protect from freezing.
Manufactured and Distributed by: Crown Laboratories, Inc., Johnson City, TN 37604

-
Triderm (Triamcinolone Acetonide Cream USP), 0.5% tube label
NDC 0316-0175-15
Rx Only
TRIDERM CREAM TM
Triamcinolone Acetonide Cream USP, 0.5%
15 grams
WARNING: Keep out of reach of children
For External Use Only
Not for Ophthalmic UseStore at 20 o-25 oC (68 o-77 oF) [see USP Controlled Room Temperature]
Avoid excessive heat. Protect from freezing.
Each gram contains 5 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
USUAL DOSAGE: 2 or 3 applications daily. See package insert for full prescribing information.
TO OPEN: Use cap to puncture seal.
IMPORTANT: Do not use if seal has been punctured or is not visible. See crimp of tube for batch number and expiration date.
Manufactured and Distributed by:
Crown Laboratories, Inc.
Johnson City, TN 37604

-
Triderm (Triamcinolone Acetonide Cream USP), 0.5% carton label
NDC 0316-0175-15
Rx Only
TRIDERM CREAM TM
Triamcinolone Acetonide Cream USP, 0.5%
15 grams
WARNING: Keep out of reach of children
For External Use Only
Not for Ophthalmic UseUSUAL DOSAGE: 2 or 3 applications daily. See package insert for full prescribing information.
See end of carton for batch number and expiration date.
Each gram contains 5 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
Directions for puncturing seal: Remove cap. Turn cap upside down and place puncture tip onto tube seal; push down until seal is punctured. Screw cap back on to close
IMPORTANT: Do not use if seal has been punctured or is not visible.
Store at 20 o-25 oC (68 o-77 oF) [see USP Controlled Room Temperature]
Avoid excessive heat. Protect from freezing.
Manufactured and Distributed by: Crown Laboratories, Inc., Johnson City, TN 37604

-
INGREDIENTS AND APPEARANCE
TRIDERM
triamcinolone acetonide creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0316-0175 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRIAMCINOLONE ACETONIDE (UNII: F446C597KA) (TRIAMCINOLONE ACETONIDE - UNII:F446C597KA) TRIAMCINOLONE ACETONIDE 5 mg in 1 g Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) WATER (UNII: 059QF0KO0R) CETYL PALMITATE (UNII: 5ZA2S6B08X) SORBIC ACID (UNII: X045WJ989B) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) MINERAL OIL (UNII: T5L8T28FGP) POLAWAX POLYSORBATE (UNII: Q504PL8E0V) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0316-0175-15 1 in 1 CARTON 03/25/2015 1 15 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:0316-0175-16 1 in 1 CARTON 09/01/2017 2 454 g in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA088042 03/19/1984 TRIDERM
triamcinolone acetonide creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0316-0170 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRIAMCINOLONE ACETONIDE (UNII: F446C597KA) (TRIAMCINOLONE ACETONIDE - UNII:F446C597KA) TRIAMCINOLONE ACETONIDE 1 mg in 1 g Inactive Ingredients Ingredient Name Strength POTASSIUM SORBATE (UNII: 1VPU26JZZ4) MINERAL OIL (UNII: T5L8T28FGP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SORBITOL (UNII: 506T60A25R) CETYL PALMITATE (UNII: 5ZA2S6B08X) SORBIC ACID (UNII: X045WJ989B) WATER (UNII: 059QF0KO0R) POLAWAX POLYSORBATE (UNII: Q504PL8E0V) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0316-0170-01 1 in 1 CARTON 03/19/1984 1 28.4 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:0316-0170-03 1 in 1 CARTON 03/19/1984 2 85.2 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:0316-0170-02 3 g in 1 POUCH; Type 0: Not a Combination Product 11/20/2007 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA088042 03/19/1984 TRIDERM
triamcinolone acetonide creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0316-0165 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRIAMCINOLONE ACETONIDE (UNII: F446C597KA) (TRIAMCINOLONE ACETONIDE - UNII:F446C597KA) TRIAMCINOLONE ACETONIDE 0.25 mg in 1 g Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SORBITOL (UNII: 506T60A25R) CETYL PALMITATE (UNII: 5ZA2S6B08X) SORBIC ACID (UNII: X045WJ989B) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) WATER (UNII: 059QF0KO0R) MINERAL OIL (UNII: T5L8T28FGP) POLAWAX POLYSORBATE (UNII: Q504PL8E0V) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0316-0165-15 1 in 1 CARTON 03/25/2015 1 15 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:0316-0165-85 1 in 1 CARTON 03/25/2015 2 85.2 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:0316-0165-16 1 in 1 CARTON 03/25/2015 3 454 g in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA088042 03/19/1984 Labeler - Crown Laboratories (079035945) Registrant - Crown Laboratories (079035945) Establishment Name Address ID/FEI Business Operations Crown Laboratories 079035945 manufacture(0316-0170, 0316-0165, 0316-0175)






