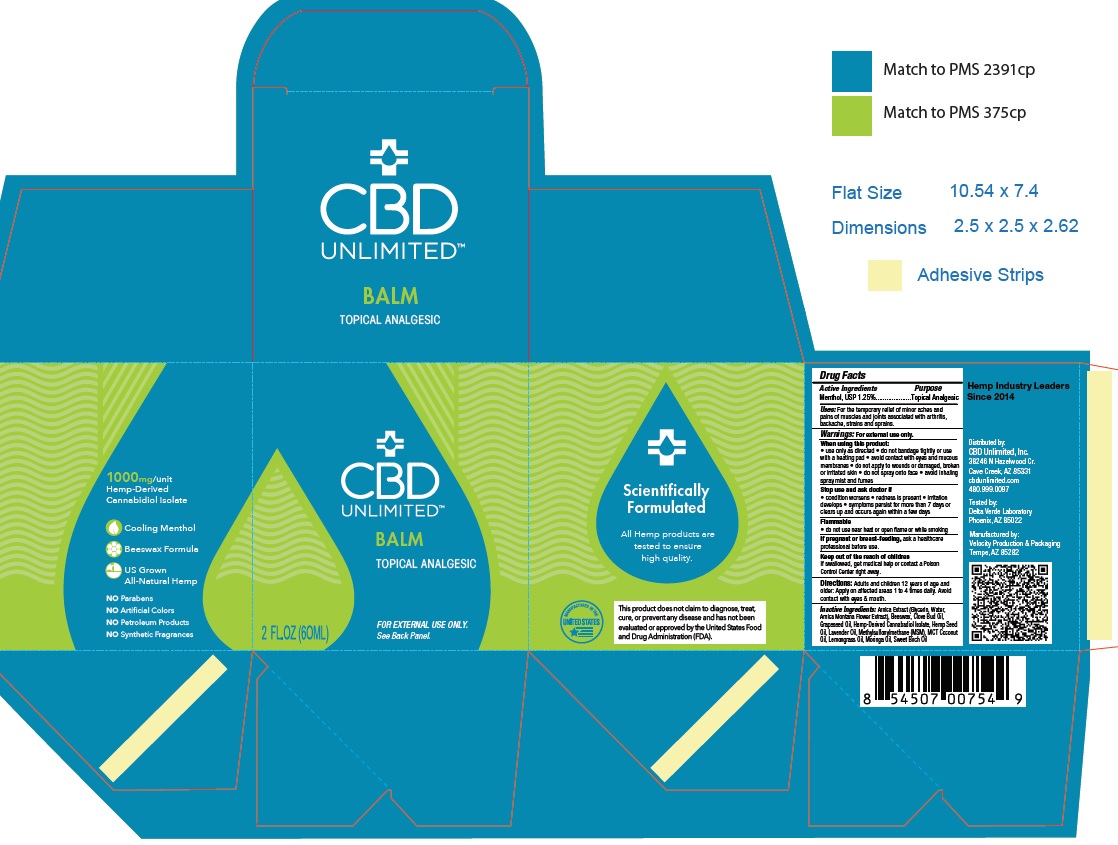
Label: CBD UNLIMITED BALM TOPICAL ANALGESIC- menthol cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 73045-003-01 - Packager: ENDEXX CORPORATION
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 5, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings:
Warnings: For external use only.
When using this product:
• use only as directed• do not bandage tightly or use with a heating pad
• avoid contact with eyes and mucous membranes
• do not apply to wounds or damaged, broken or irritated skin
• do not spray onto face
• avoid inhaling spray mist and fumes
Stop use and ask doctor if
• condition worsens
• redness is present
• irritation develops
• symptoms persist for more than 7 days or clears up and occurs again within a few days
Flammable
• do not use near heat or open _ame or while smoking
If pregnant or breast-feeding, ask a healthcare
professional before use.
Keep out of the reach of children
- Keep Out of Reach of Children
- Directions:
- Inactive Ingredients
- Image
-
INGREDIENTS AND APPEARANCE
CBD UNLIMITED BALM TOPICAL ANALGESIC
menthol creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73045-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 125 mg in 1 mL Inactive Ingredients Ingredient Name Strength MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) YELLOW WAX (UNII: 2ZA36H0S2V) CANNABIDIOL (UNII: 19GBJ60SN5) OLIVE OIL (UNII: 6UYK2W1W1E) HETEROTHECA INULOIDES FLOWER (UNII: W9NZ9OZF68) MORINGA OLEIFERA SEED OIL (UNII: REM6A5QMC0) GRAPE SEED OIL (UNII: 930MLC8XGG) Cannabis Sativa Seed Oil (UNII: 69VJ1LPN1S) Clove Oil (UNII: 578389D6D0) Lavender Oil (UNII: ZBP1YXW0H8) East Indian Lemongrass Oil (UNII: UP0M8M3VZW) Methyl Salicylate (UNII: LAV5U5022Y) Dimethyl Sulfone (UNII: 9H4PO4Z4FT) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73045-003-01 60 mL in 1 JAR; Type 0: Not a Combination Product 05/05/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 05/05/2020 Labeler - ENDEXX CORPORATION (002119594) Registrant - ENDEXX CORPORATION (002119594) Establishment Name Address ID/FEI Business Operations Velocity Production & Packaging 081076893 MANUFACTURE(73045-003)