Label: SINUS AND ALLERGY RELIEF PE- chlorpheniramine maleate, phenylephrine hcl tablet
- NDC Code(s): 36800-642-08
- Packager: Topco Associates, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated July 19, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- high blood pressure
- heart disease
- thyroid disease
- diabetes
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
- a breathing problem such as emphysema or chronic bronchitis
When using this product
- do not exceed recommended dosage
- excitability may occur, especially in children
- drowsiness may occur
- avoid alcoholic beverages
- alcohol, sedatives and tranquilizers may increase drowsiness
- use caution when driving a motor vehicle or operating machinery
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
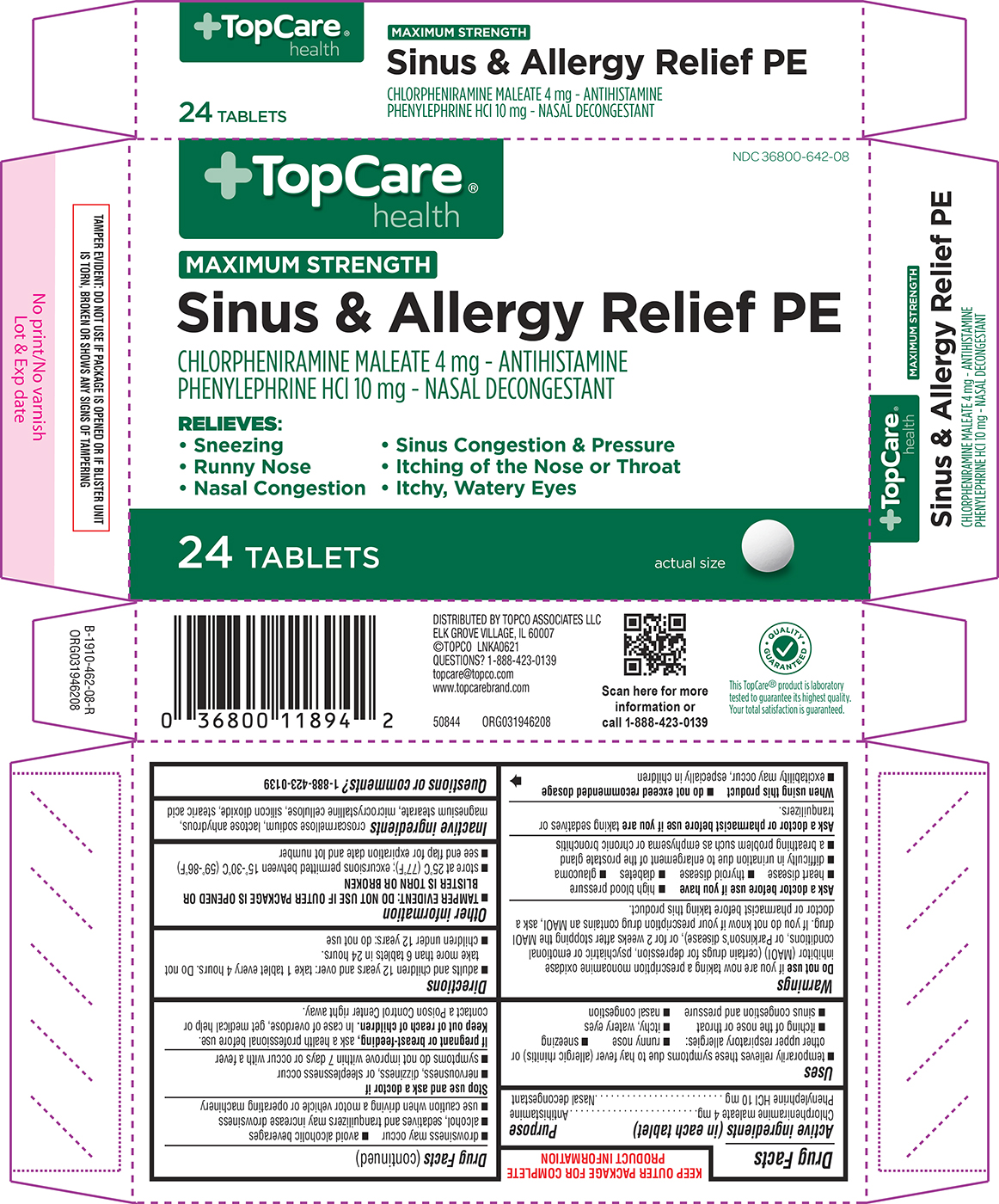
Principal display panel
+TopCare®
healthNDC 36800-642-08
MAXIMUM STRENGTH
Sinus & Allergy Relief PECHLORPHENIRAMINE MALEATE 4 mg - ANTIHISTAMINE
PHENYLEPHRINE HCl 10 mg - NASAL DECONGESTANTRELIEVES:
• Sneezing • Sinus Congestion & Pressure
• Runny Nose • Itching of the Nose or Throat
• Nasal Congestion • Itchy, Watery Eyes24 TABLETS
actual size
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER UNIT
IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERINGDISTRIBUTED BY TOPCO ASSOCIATES LLC
ELK GROVE VILLAGE, IL 60007
©TOPCO LNKA0621
QUESTIONS? 1-888-423-0139
topcare@topco.com
www.topcarebrand.com50844 ORG031946208
QUALITY GUARANTEED
This TopCare® product is laboratory
tested to guarantee its highest quality.
Your total satisfaction is guaranteed.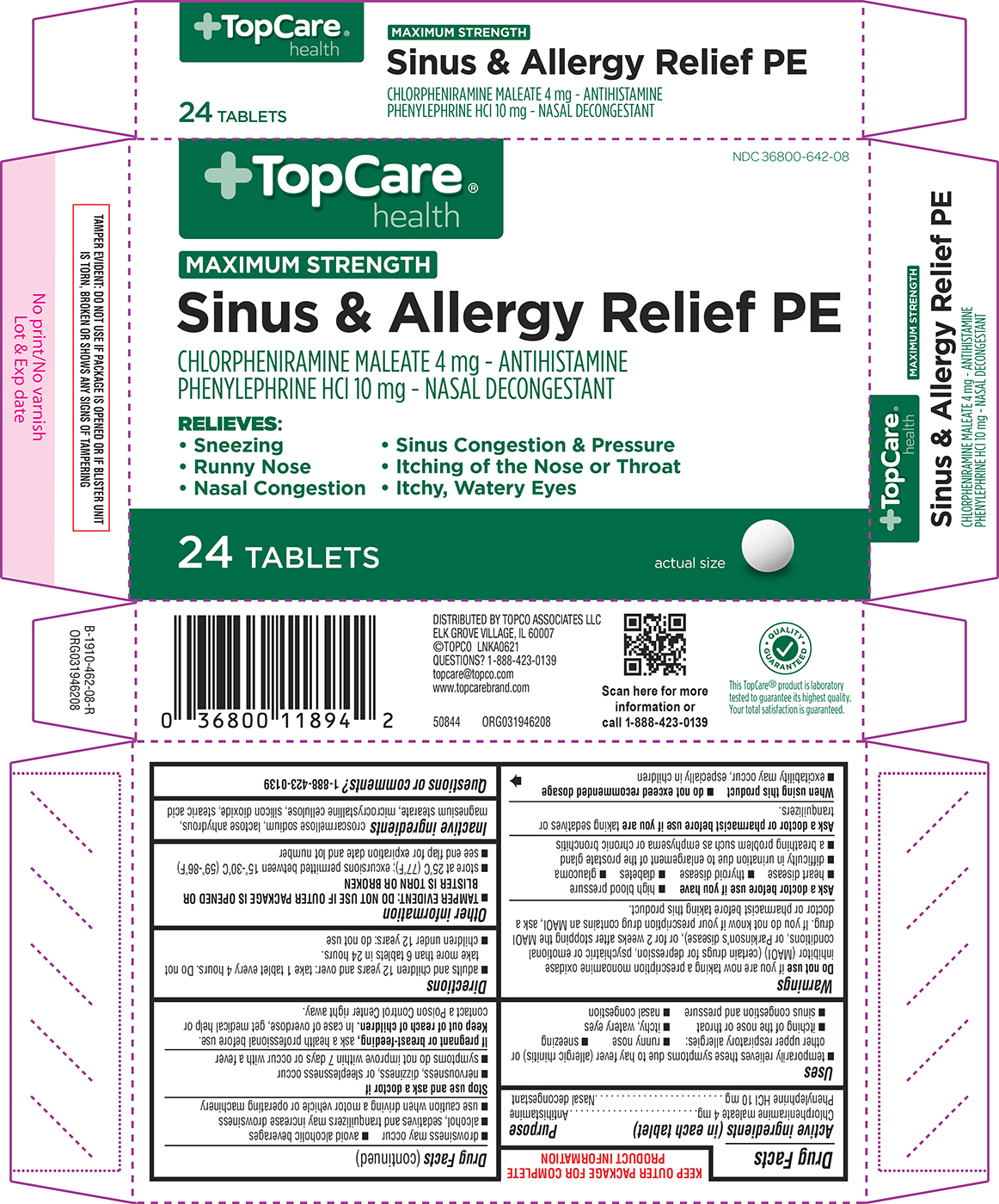
TopCare 44-462
-
INGREDIENTS AND APPEARANCE
SINUS AND ALLERGY RELIEF PE
chlorpheniramine maleate, phenylephrine hcl tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:36800-642 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 4 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white Score no score Shape ROUND Size 7mm Flavor Imprint Code 44;462 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:36800-642-08 1 in 1 CARTON 07/02/2021 1 24 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 07/02/2021 Labeler - Topco Associates, LLC (006935977) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(36800-642) , pack(36800-642) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(36800-642)