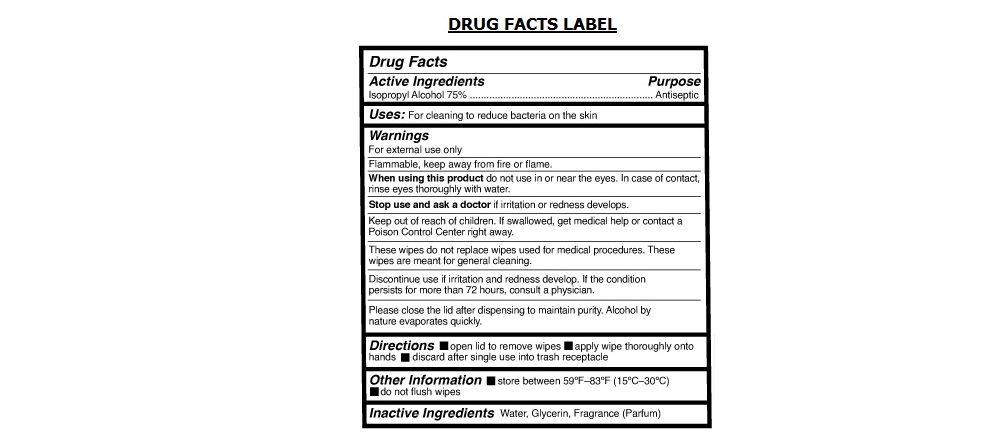
Label: CLEANSHOT SANITIZING ALCOHOL WIPES- isopropyl alcohol cloth
- NDC Code(s): 73779-916-50
- Packager: CIC Coatings, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 23, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Purpose
- INDICATIONS & USAGE
-
Warnings
For external use only
Flammable, keep away from fire or flame.
When using this product do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or redness develops.
These wipes do not replace wipes used for medical procedures. These wipes are meant for general cleaning.
Discontinue use if irritation and redness develop. If the condition persists for more than 72 hours, consult a physician.
Please close the lid after dispensing to maintain purity. Alcohol by nature evaporates quickly.
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- SPL UNCLASSIFIED SECTION
- Packaging
-
INGREDIENTS AND APPEARANCE
CLEANSHOT SANITIZING ALCOHOL WIPES
isopropyl alcohol clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73779-916 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 75 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73779-916-50 500 in 1 CANISTER 10/15/2020 10/31/2024 1 5.4 mL in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 10/15/2020 10/31/2024 Labeler - CIC Coatings, LLC (832380344) Establishment Name Address ID/FEI Business Operations CIC Coatings, LLC 832380344 manufacture(73779-916)