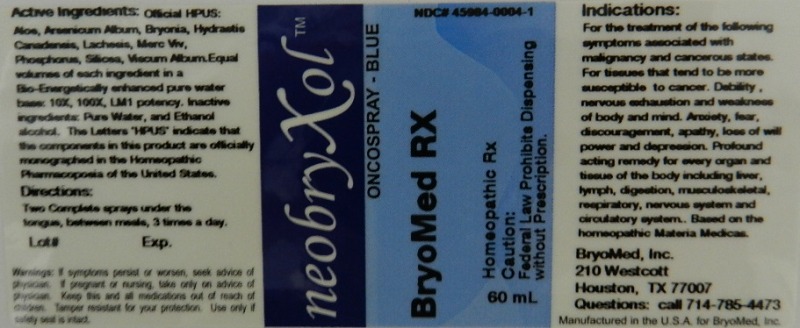
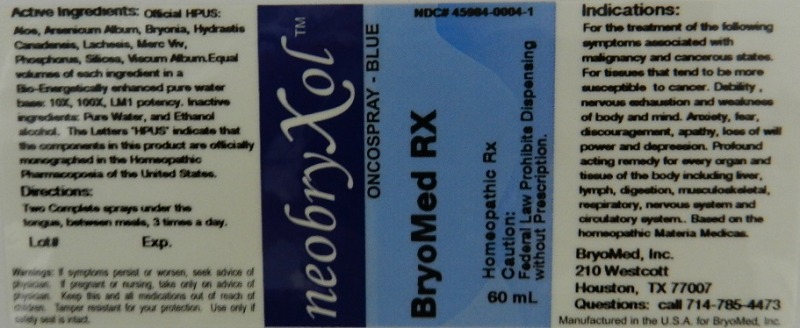
Label: ONCOSPRAY BLUE- homepathic rx liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 45984-0004-1 - Packager: Bryomed Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 9, 2012
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Inactive Ingredients
- Dosage and Administration
-
Purpose
Indications:
For the treatment of the following symptoms associated with malignancy and cancerous states.
- for tissues that tend to be more susceptible to cancer
- debility, nervous exhaustion and weakness of body and mind
- anxiety, fear, discouragement, apathy, loss of will power and depression
- profound acting remedy for every organ and tissue of the body including liver, lymph, digestion, musculoskeletal, respiratory, nervous system and circulatory system
Reference image oncospray.jpg
- for tissues that tend to be more susceptible to cancer
-
Warnings
Warnings: If symptoms persist or worsen, seek advice of physician. If pregnant or nursing, take only on advice of physician. Keep this and all medications out of reach of children.
Tamper resistant for your protection. Use only if safety seal is intact.
Caution: Federal law prohibits dispensing without prescription.
Reference image oncospray.jpg
- KEEP OUT OF REACH OF CHILDREN
-
Indications and Usage
For the treatment of the following symptoms associated with malignancy and cancerous states. For tissues that tend to be more susceptible to cancer. Debility, nervous exhaustion and weakness of body and mind. Anxiety, fear, discouragement, apathy, loss of will power and depression. Profound acting remedy for every organ and tissue of the body including liver, lymph, digestion, musculoskeletal, respiratory, nervous system and circulatory system.
Based on the homeopathic Materia Medicas.
Reference image oncospray.jpg - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ONCOSPRAY BLUE
homepathic rx liquidProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:45984-0004 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALOE (UNII: V5VD430YW9) (ALOE - UNII:V5VD430YW9) ALOE 10 [hp_X] in 60 mL ARSENIC TRIOXIDE (UNII: S7V92P67HO) (ARSENIC TRIOXIDE - UNII:S7V92P67HO) ARSENIC TRIOXIDE 10 [hp_X] in 60 mL BRYONIA ALBA ROOT (UNII: T7J046YI2B) (BRYONIA ALBA ROOT - UNII:T7J046YI2B) BRYONIA ALBA ROOT 10 [hp_X] in 60 mL GOLDENSEAL (UNII: ZW3Z11D0JV) (GOLDENSEAL - UNII:ZW3Z11D0JV) GOLDENSEAL 10 [hp_X] in 60 mL LACHESIS MUTA VENOM (UNII: VSW71SS07I) (LACHESIS MUTA VENOM - UNII:VSW71SS07I) LACHESIS MUTA VENOM 10 [hp_X] in 60 mL PHOSPHORUS (UNII: 27YLU75U4W) (PHOSPHORUS - UNII:27YLU75U4W) PHOSPHORUS 10 [hp_X] in 60 mL SILICON DIOXIDE (UNII: ETJ7Z6XBU4) (SILICON DIOXIDE - UNII:ETJ7Z6XBU4) SILICON DIOXIDE 10 [hp_X] in 60 mL VISCUM ALBUM WHOLE (UNII: E6839Q6DO1) (VISCUM ALBUM WHOLE - UNII:E6839Q6DO1) VISCUM ALBUM WHOLE 10 [hp_X] in 60 mL MERCURY (UNII: FXS1BY2PGL) (MERCURY - UNII:FXS1BY2PGL) MERCURY 10 [hp_X] in 60 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:45984-0004-1 60 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/09/2012 Labeler - Bryomed Inc. (078382220) Registrant - Bryomed Inc. (078382220)