Label: VITAMIN B COMPLEX- thiamine hydrochloride, riboflavin 5 phosphate sodium, dexpanthenol and niacinamide injection
- NDC Code(s): 67457-146-30
- Packager: Mylan Institutional LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated September 15, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Vitamin B-Complex 100 Injection is a sterile solution for intramuscular or slow intravenous injection comprised of vitamins which may be categorized as belonging to the vitamin B complex group.
Each mL contains: Thiamine Hydrochloride 100 mg, Riboflavin 5’ Phosphate Sodium 2 mg, Pyridoxine Hydrochloride 2 mg, Dexpanthenol 2 mg, Niacinamide 100 mg, with Benzyl Alcohol 2% as preservative, in Water for Injection. Sodium Hydroxide and/or Hydrochloric Acid may have been used to adjust pH.
-
INDICATIONS AND USAGE
In disorders requiring parenteral administration of vitamins, i.e. pre- and post-operative treatment, when requirements are increased as in fever, severe burns, increased metabolism, pregnancy, gastrointestinal disorders interfering with intake or absorption of vitamins, prolonged or wasting diseases, alcoholism and where other deficiencies exist.
- CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
-
ADVERSE REACTIONS
Mild transient diarrhea, polycythemia vera, peripheral vascular thrombosis, itching transitory exanthema, feeling of swelling of entire body, anaphylactic shock and death. Sensitivity to the ingredients listed may occur (see WARNINGS). Use should be discontinued upon observance of any untoward reaction. Pain upon intramuscular injection may be noted.
-
DOSAGE AND ADMINISTRATION
Usually 0.25 to 2 mL by intramuscular or slow intravenous injection. High concentrations given intravenously may be diluted using parenteral infusion solutions. (See PRECAUTIONS.)
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever the solution and container permit (see HOW SUPPLIED).
-
HOW SUPPLIED
Vitamin B-Complex 100 Injection is available in a 30 mL multiple-dose vial individually boxed.
Phase separation due to reduced solubility can occur under certain conditions of shipping or storage (e.g. accidental freezing), which may produce visible particles. Do not use product if these do not redissolve on warming to body temperature and shaking well.
Refrigeration of the product may cause darkening of the solution due to the riboflavin content. The colour does not affect the safety or efficacy of the product.
PROTECT FROM LIGHT: Store in carton until contents are used. Store under refrigeration 2° to 8°C (36° to 46°F). Do not permit to freeze.
Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505 U.S.A.Manufactured by:
Mylan Institutional
Galway, Ireland0415L107
Revised: 9/2021
MI:VITBIJ:R4 -
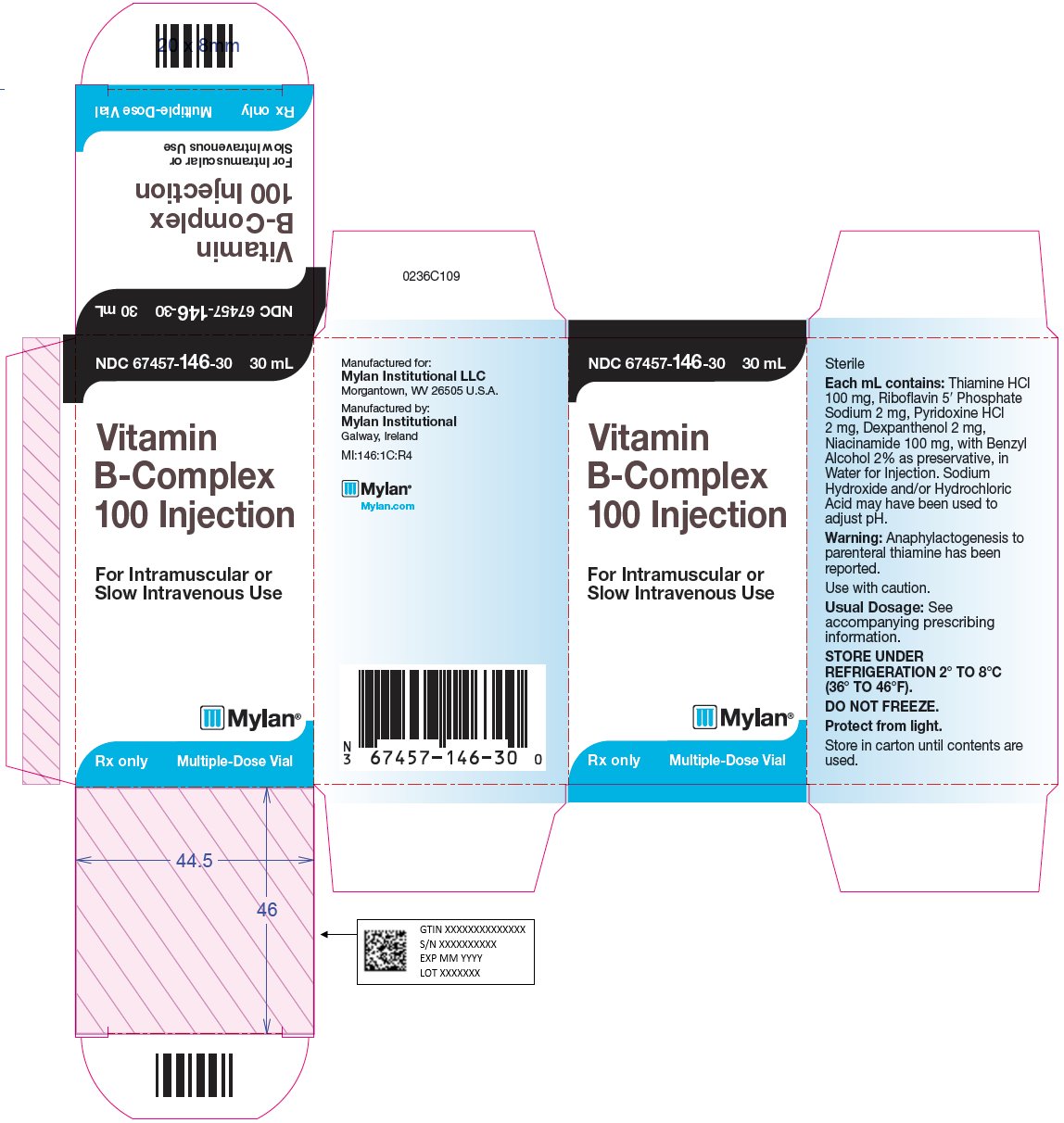
PRINCIPAL DISPLAY PANEL - 30 mL
NDC 67457-146-30 30 mL
Vitamin
B-Complex
100 InjectionFor Intramuscular or
Slow Intravenous UseRx only Multi-Dose Vial
Sterile
Each mL contains: Thiamine HCl
100 mg, Riboflavin 5' Phosphate
Sodium 2 mg, Pyridoxine HCl
2 mg, Dexpanthenol 2 mg,
Niacinamide 100 mg, with Benzyl
Alcohol 2% as preservative, in
Water for Injection. Sodium
Hydroxide and/or Hydrochloric
Acid may have been used to
adjust pH.Warning: Anaphylactogenesis to
parenteral thiamine has been
reported.Use with caution.
Usual Dosage: See
accompanying prescribing
information.STORE UNDER
REFRIGERATION 2° TO 8°C
(36° TO 46°F).DO NOT FREEZE.
Protect from light.
Store in carton until contents are
used.Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505 U.S.A.Manufactured by:
Mylan Institutional
Galway, IrelandMI:146:1C:R4
Mylan.com
-
INGREDIENTS AND APPEARANCE
VITAMIN B COMPLEX
thiamine hydrochloride, riboflavin 5 phosphate sodium, dexpanthenol and niacinamide injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:67457-146 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength THIAMINE HYDROCHLORIDE (UNII: M572600E5P) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE 100 mg in 1 mL RIBOFLAVIN 5'-PHOSPHATE SODIUM (UNII: 20RD1DZH99) (FLAVIN MONONUCLEOTIDE - UNII:7N464URE7E) FLAVIN MONONUCLEOTIDE 2 mg in 1 mL DEXPANTHENOL (UNII: 1O6C93RI7Z) (DEXPANTHENOL - UNII:1O6C93RI7Z) DEXPANTHENOL 2 mg in 1 mL NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 100 mg in 1 mL PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE HYDROCHLORIDE 2 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) 20 mg in 1 mL WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67457-146-30 1 in 1 CARTON 05/10/2013 1 30 mL in 1 VIAL, MULTI-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug other 05/10/2013 Labeler - Mylan Institutional LLC (790384502)