Label: NASAL DECONGESTANT MAXIMUM STRENGTH- pseudoephedrine hcl tablet, film coated
- NDC Code(s): 68016-473-24, 68016-473-96
- Packager: Chain Drug Consortium
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 6, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- diabetes
- heart disease
- high blood pressure
- thyroid disease
- difficulty in urination due to enlargement of the prostate gland
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
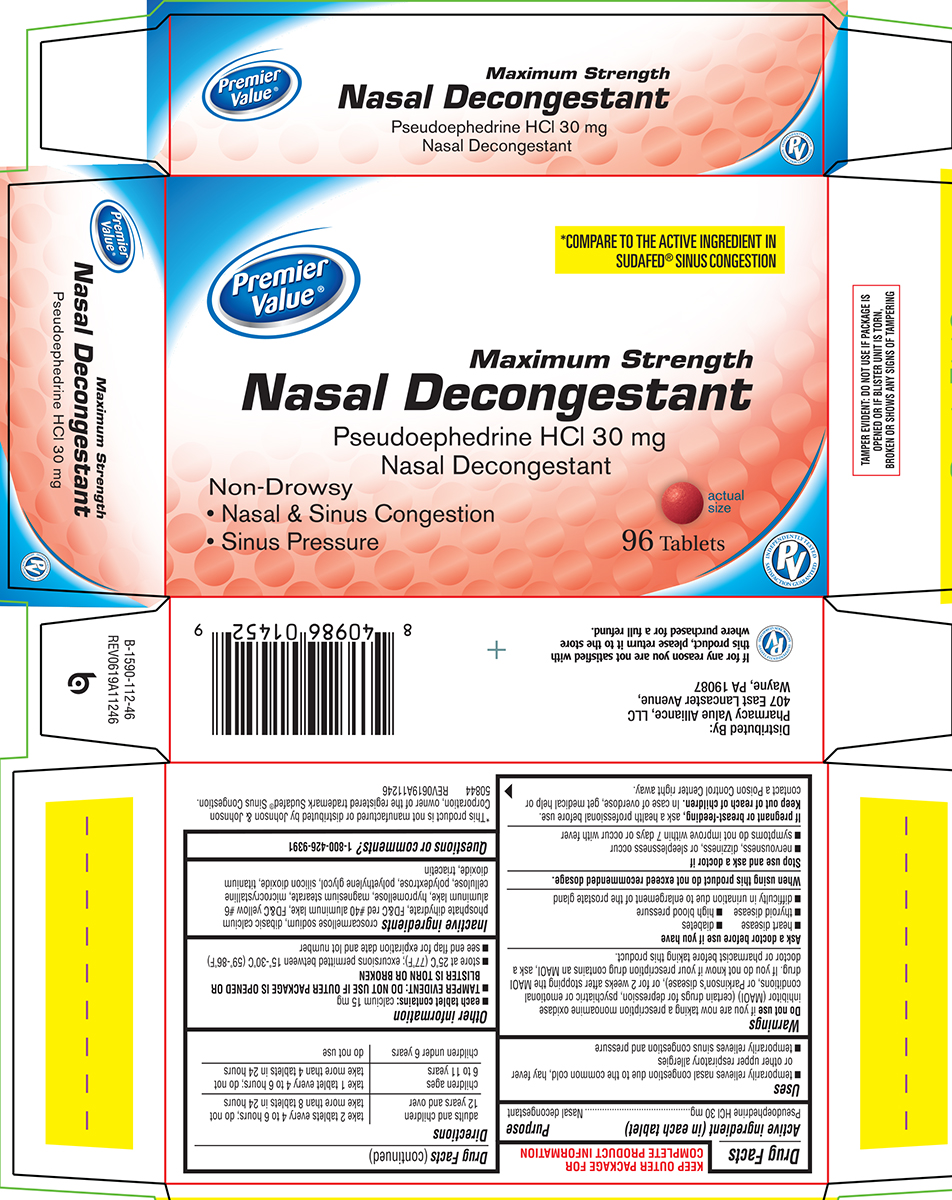
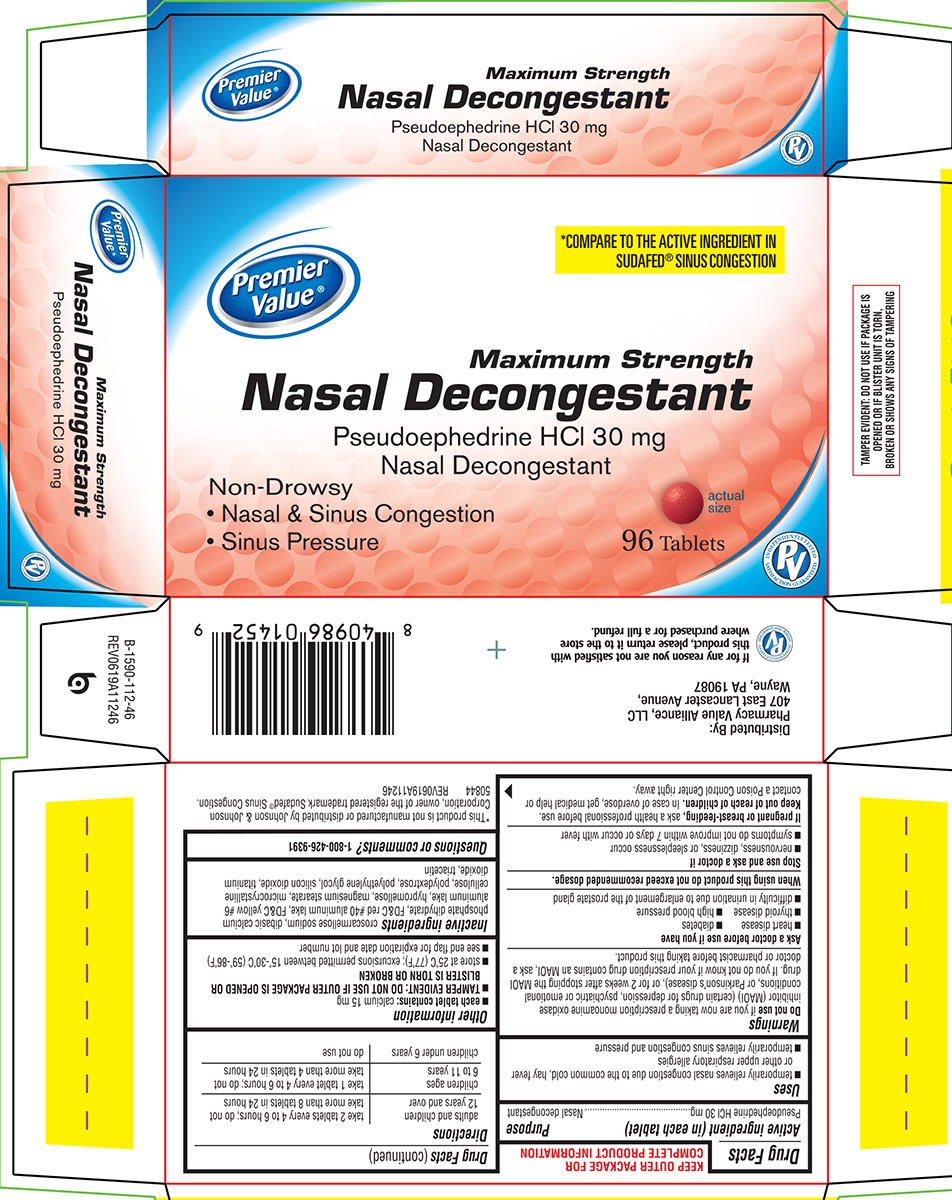
Principal Display Panel
Premier
Value®*COMPARE TO THE ACTIVE INGREDIENT IN
SUDAFED® SINUS CONGESTIONMaximum Strength
Nasal DecongestantPseudoephedrine HCl 30 mg
Nasal DecongestantNon-Drowsy
• Nasal & Sinus Congestion
• Sinus Pressure96 Tablets
actual
sizePV
INDEPENDENTLY TESTED
SATISFACTION GUARANTEEDTAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN,
BROKEN OR SHOWS ANY SIGNS OF TAMPERING*This product is not manufactured or distributed by Johnson & Johnson
Corporation, owner of the registered trademark Sudafed® Sinus Congestion.
50844 REV0619A11246Distributed By:
Pharmacy Value Alliance, LLC
407 East Lancaster Avenue,
Wayne, PA 19087
If for any reason you are not satisfied with
this product, please return it to the store
where purchased for a full refund.
44-112
-
INGREDIENTS AND APPEARANCE
NASAL DECONGESTANT MAXIMUM STRENGTH
pseudoephedrine hcl tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-473 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PSEUDOEPHEDRINE HYDROCHLORIDE (UNII: 6V9V2RYJ8N) (PSEUDOEPHEDRINE - UNII:7CUC9DDI9F) PSEUDOEPHEDRINE HYDROCHLORIDE 30 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color red Score no score Shape ROUND Size 7mm Flavor Imprint Code 44;112 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-473-24 1 in 1 CARTON 08/25/1981 1 24 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:68016-473-96 4 in 1 CARTON 08/25/1981 2 24 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 08/25/1981 Labeler - Chain Drug Consortium (101668460) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(68016-473) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(68016-473) , pack(68016-473) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(68016-473) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 868734088 manufacture(68016-473) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(68016-473) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(68016-473)