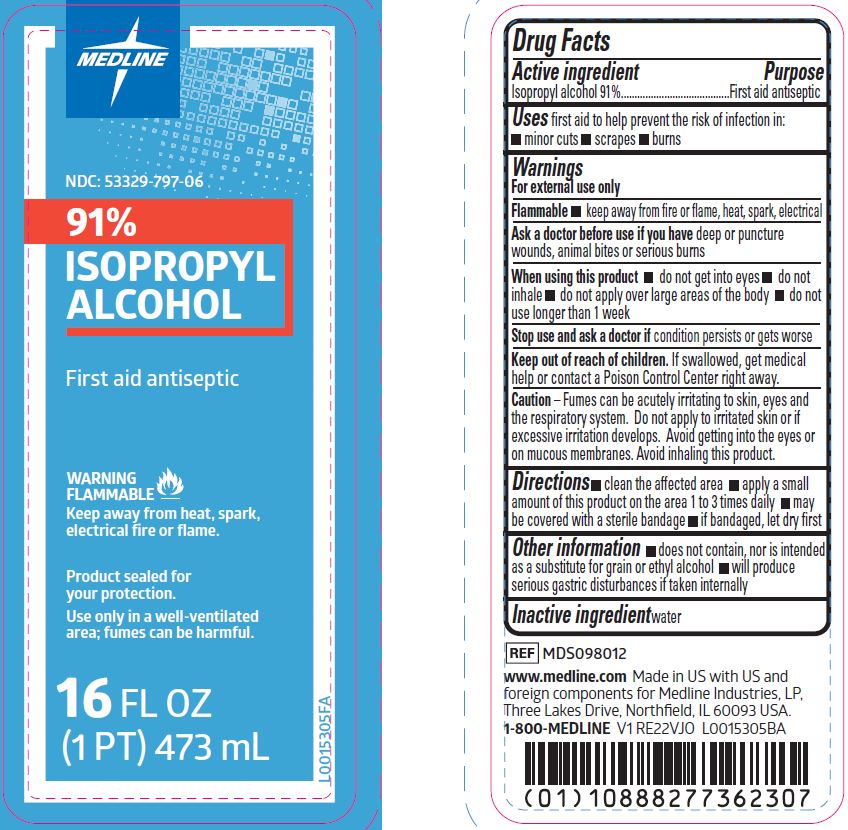
Label: ISOPROPYL ALCOHOL solution
- NDC Code(s): 53329-797-06
- Packager: Medline Industries, LP
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 29, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- Flammable
- Ask a doctor before use if you have
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children.
- Caution
- Directions
- Other information
- Inactive ingredient
- Manufacturing Information
- Package Label
-
INGREDIENTS AND APPEARANCE
ISOPROPYL ALCOHOL
isopropyl alcohol solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-797 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 91 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-797-06 473 mL in 1 BOTTLE; Type 0: Not a Combination Product 08/06/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 08/06/2015 Labeler - Medline Industries, LP (025460908) Registrant - Medline Industries, LP (025460908)