Label: LORATADINE ODT- loratadine tablet, orally disintegrating
-
Contains inactivated NDC Code(s)
NDC Code(s): 68016-527-31 - Packager: Chain Drug Consortium, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 8, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
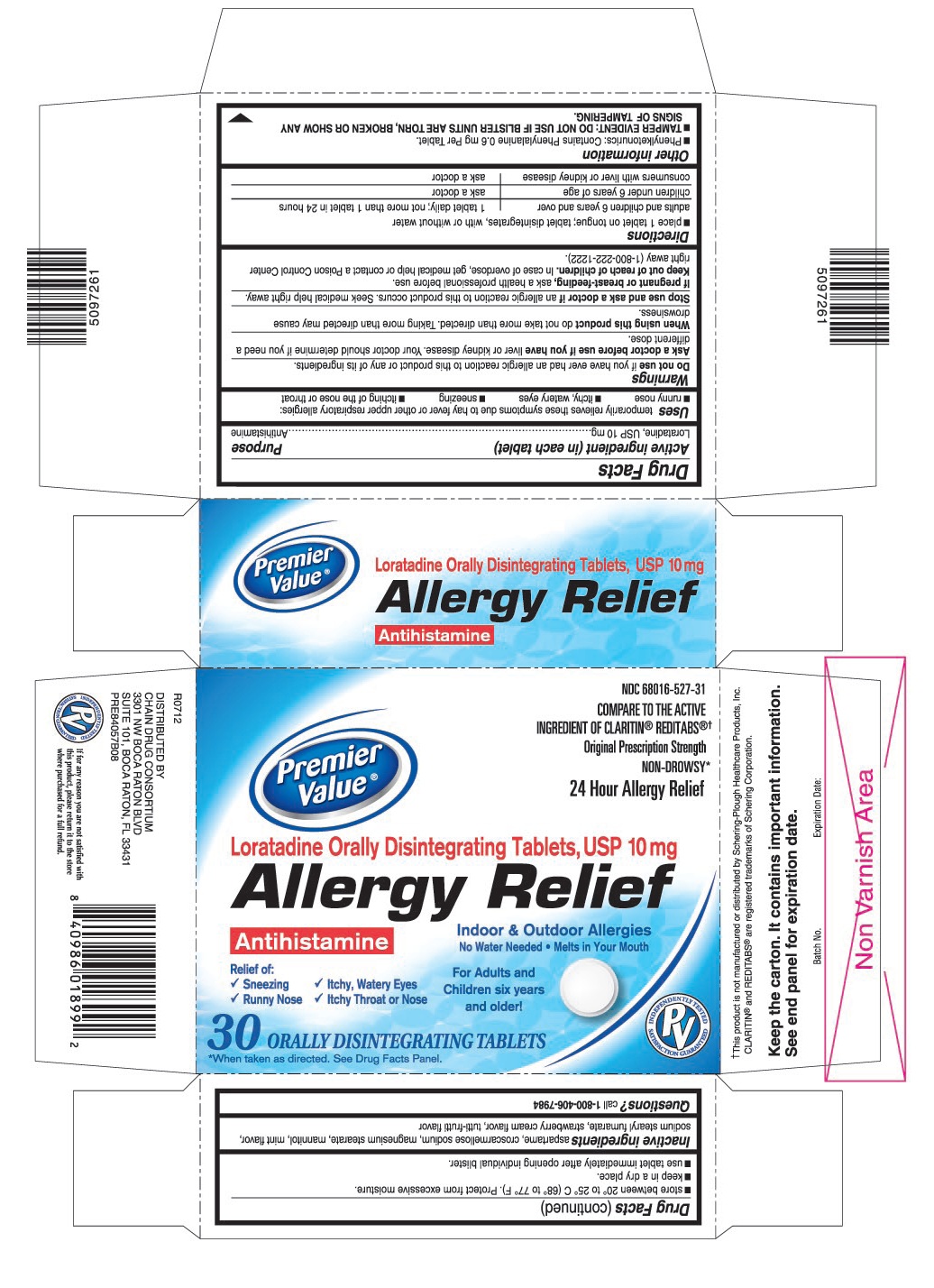
- ACTIVE INGREDIENT (IN EACH TABLET)
- PURPOSE
- USES
-
WARNINGS
Ask a doctor before use if you have
liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product
do not take more than directed. Taking more than directed may cause drowsiness.
- DIRECTIONS
-
OTHER INFORMATION
- •
- Phenylketonurics: Contains Phenylalanine 0.6 mg Per Tablet.
- •
- TAMPER EVIDENT: DO NOT USE IF BLISTER UNITS ARE TORN, BROKEN OR SHOW ANY SIGNS OF TAMPERING.
- •
- store between 20° to 25° C (68° to 77° F). Protect from excessive moisture.
- •
- keep in a dry place.
- •
- use tablet immediately after opening individual blister.
- INACTIVE INGREDIENTS
- QUESTIONS?
-
PRINCIPAL DISPLAY PANEL
Premier Value®
NDC 68016-527-31
Original Prescription Strength
NON-DROWSY*
24 Hour Allergy Relief
Loratadine Orally Disintegrating Tablets, 10 mg
Allergy Relief
Antihistamine
Indoor & Outdoor Allergies
No water needed. Melts in your mouth.
Relief of:
- •
- Sneezing
- •
- Itchy, Watery Eyes
- •
- Runny Nose
- •
- Itchy Throat or Nose
For Adults and Children six years and older!
30 ORALLY DISINTEGRATING TABLETS
COMPARE TO THE ACTIVE INGREDIENT OF CLARITIN®REDITABS®†
*When taken as directed. See Drug Facts Panel.
†This product is not manufactured or distributed by Schering-Plough HealthCare Products, Inc. CLARITIN® and REDITABS® are registered trademarks of Schering Corporation.
-
INGREDIENTS AND APPEARANCE
LORATADINE ODT
loratadine tablet, orally disintegratingProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-527 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 10 mg Inactive Ingredients Ingredient Name Strength ASPARTAME (UNII: Z0H242BBR1) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) SODIUM STEARYL FUMARATE (UNII: 7CV7WJK4UI) Product Characteristics Color white (white to off-white) Score no score Shape ROUND (flat faced beveled edge) Size 10mm Flavor STRAWBERRY, TUTTI FRUTTI, MINT Imprint Code RC17 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-527-31 30 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077153 08/31/2007 Labeler - Chain Drug Consortium, LLC (101668460) Registrant - Ranbaxy Pharmaceuticals Inc. (937890044) Establishment Name Address ID/FEI Business Operations Ohm Laboratories Inc. 051565745 MANUFACTURE(68016-527)