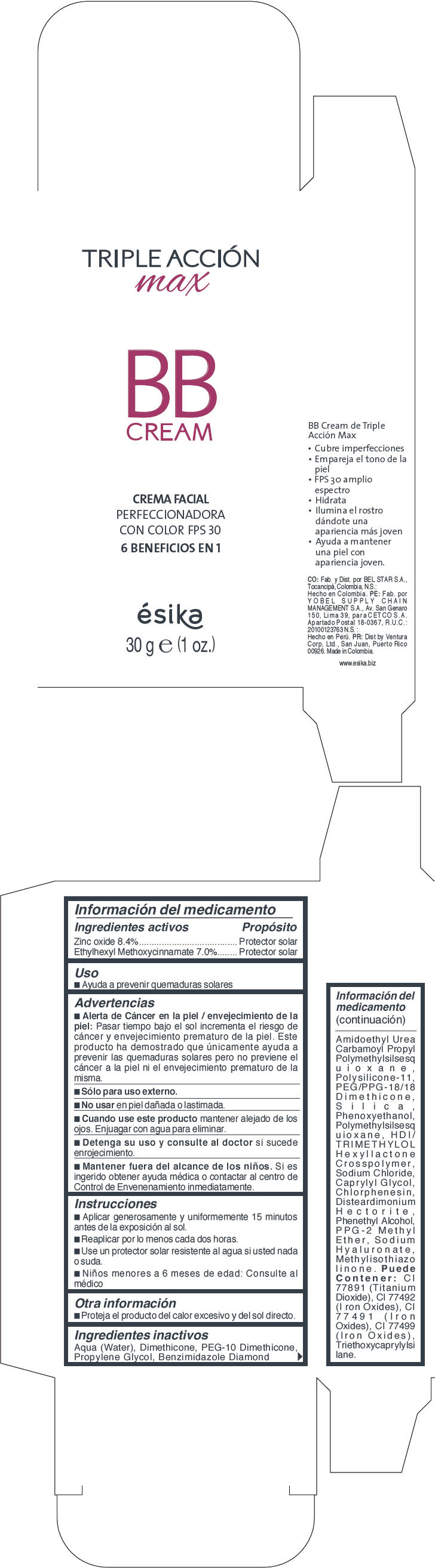
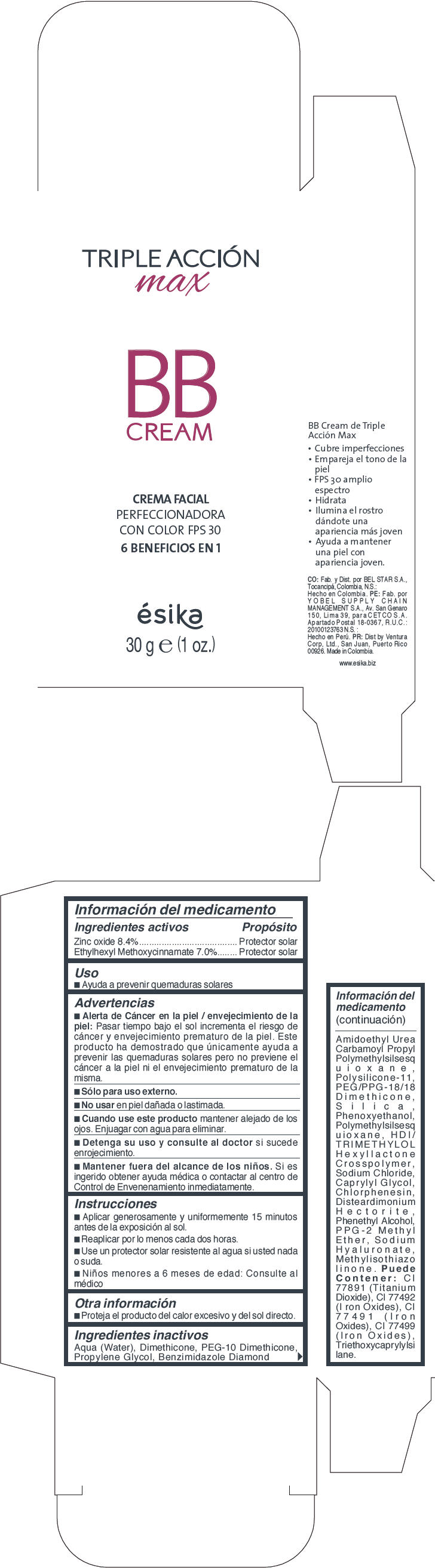
Label: ESIKA TRIPLE ACCION MAX BB LIGHT BEIGE- zinc oxide and octinoxate cream
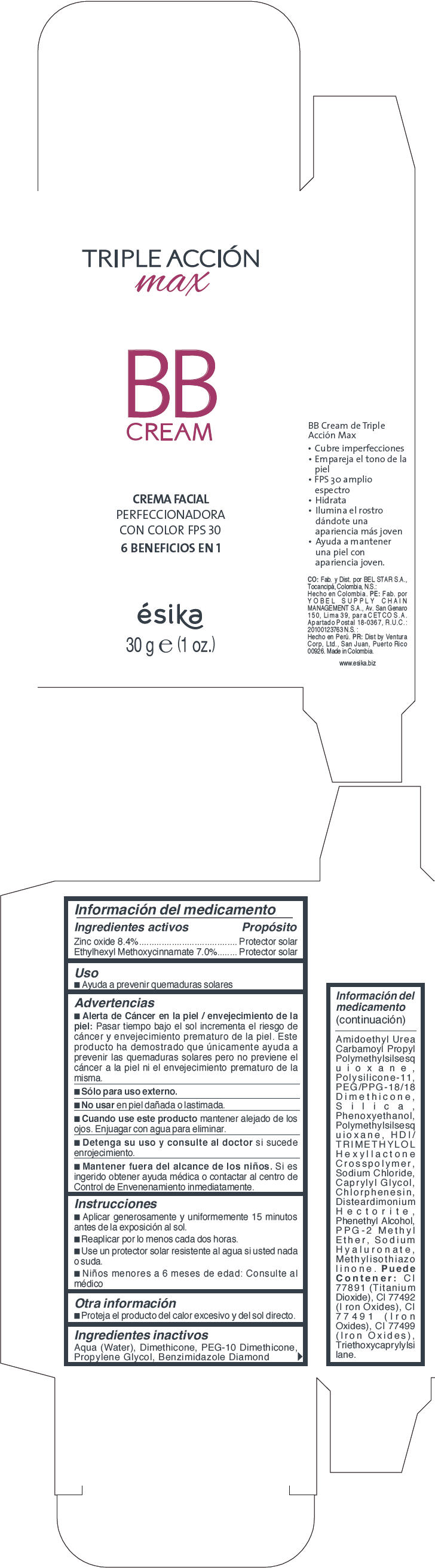
ESIKA TRIPLE ACCION MAX BB DARK BEIGE- zinc oxide and octinoxate cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 13537-998-04, 13537-998-05, 13537-998-06, 13537-999-01, view more13537-999-02, 13537-999-03 - Packager: Ventura Corporation LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 11, 2014
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uso
-
Advertencias
- Alerta de Cáncer en la piel / envejecimiento de la piel : Pasar tiempo bajo el sol incrementa el riesgo de cáncer y envejecimiento prematuro de la piel. Este producto ha demostrado que unicamente ayuda a prevenir las quemaduras solares pero no previene el cáncer a la piel ni el envejecimiento prematuro de la misma.
- Sólo para uso externo.
- Instrucciones
- Otra información
-
Ingredientes inactivos
Aqua (Water),Dimethicone, PEG-10,Dimethicone, Propylene Glycol, Benzimidazole Diamond Amidoethyl Urea, Carbamoyl Propyl Polymethylsilsesquioxane, Polysilicone-11, PEG/PPG-18/18 Dimethicone, Silica, Phenoxyethanol, Polymethylsilsesquioxane, HDI/TRIMETHYLOL Hexyllactone Crosspolymer, Sodium Chloride, Caprylyl Glycol, Chlorphenesin, Disteardimonium Hectorite, Phenethyl Alcohol, PPG-2,Methyl Ether, Sodium Hyaluronate, Methylisothiazolinone. Puede Contener:CI 77891 (Titanium Dioxide ) , CI 77492 ( Iron Oxides), CI 77491 (Iron Oxides),CI 77499 (Iron Oxides), Triethoxycaprylylsilane.
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 30 g Tube Carton - Light Beige
- PRINCIPAL DISPLAY PANEL - 30 g Tube Carton - Dark Beige
-
INGREDIENTS AND APPEARANCE
ESIKA TRIPLE ACCION MAX BB LIGHT BEIGE
zinc oxide and octinoxate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-999 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Zinc Oxide (UNII: SOI2LOH54Z) (Zinc Oxide - UNII:SOI2LOH54Z) Zinc Oxide 0.084 g in 1 g Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 0.07 g in 1 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Dimethicone (UNII: 92RU3N3Y1O) Propylene Glycol (UNII: 6DC9Q167V3) PEG/PPG-18/18 Dimethicone (UNII: 9H0AO7T794) Silicon Dioxide (UNII: ETJ7Z6XBU4) Phenoxyethanol (UNII: HIE492ZZ3T) Sodium Chloride (UNII: 451W47IQ8X) Caprylyl Glycol (UNII: 00YIU5438U) Chlorphenesin (UNII: I670DAL4SZ) Disteardimonium Hectorite (UNII: X687XDK09L) Phenylethyl Alcohol (UNII: ML9LGA7468) Dimethyl Ether (UNII: AM13FS69BX) Hyaluronate Sodium (UNII: YSE9PPT4TH) Methylisothiazolinone (UNII: 229D0E1QFA) Titanium Dioxide (UNII: 15FIX9V2JP) Ferric Oxide Yellow (UNII: EX438O2MRT) Ferric Oxide Red (UNII: 1K09F3G675) Ferrosoferric Oxide (UNII: XM0M87F357) Triethoxycaprylylsilane (UNII: LDC331P08E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-999-02 1 in 1 CARTON 1 NDC:13537-999-01 30 g in 1 TUBE 2 NDC:13537-999-03 1 g in 1 PACKET Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 03/10/2014 ESIKA TRIPLE ACCION MAX BB DARK BEIGE
zinc oxide and octinoxate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-998 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Zinc Oxide (UNII: SOI2LOH54Z) (Zinc Oxide - UNII:SOI2LOH54Z) Zinc Oxide 0.084 g in 1 g Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 0.07 g in 1 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Dimethicone (UNII: 92RU3N3Y1O) Propylene Glycol (UNII: 6DC9Q167V3) PEG/PPG-18/18 Dimethicone (UNII: 9H0AO7T794) Silicon Dioxide (UNII: ETJ7Z6XBU4) Phenoxyethanol (UNII: HIE492ZZ3T) Sodium Chloride (UNII: 451W47IQ8X) Caprylyl Glycol (UNII: 00YIU5438U) Chlorphenesin (UNII: I670DAL4SZ) Disteardimonium Hectorite (UNII: X687XDK09L) Phenylethyl Alcohol (UNII: ML9LGA7468) Dimethyl Ether (UNII: AM13FS69BX) Hyaluronate Sodium (UNII: YSE9PPT4TH) Methylisothiazolinone (UNII: 229D0E1QFA) Titanium Dioxide (UNII: 15FIX9V2JP) Ferric Oxide Yellow (UNII: EX438O2MRT) Ferric Oxide Red (UNII: 1K09F3G675) Ferrosoferric Oxide (UNII: XM0M87F357) Triethoxycaprylylsilane (UNII: LDC331P08E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-998-05 1 in 1 CARTON 1 NDC:13537-998-04 30 g in 1 TUBE 2 NDC:13537-998-06 1 g in 1 PACKET Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 03/10/2014 Labeler - Ventura Corporation LTD (602751344) Establishment Name Address ID/FEI Business Operations Bel Star S.A. (Colombia) 880160197 MANUFACTURE(13537-999, 13537-998)