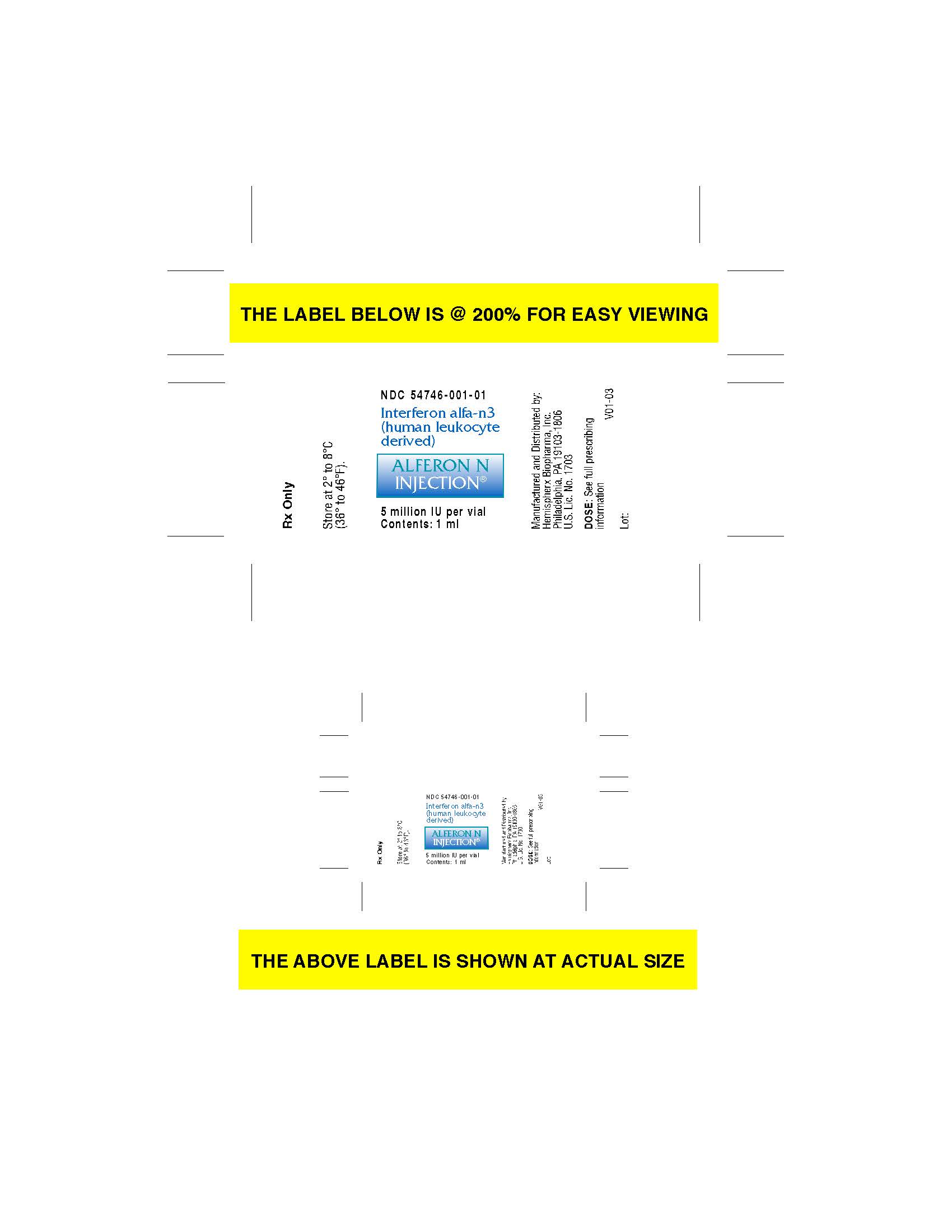
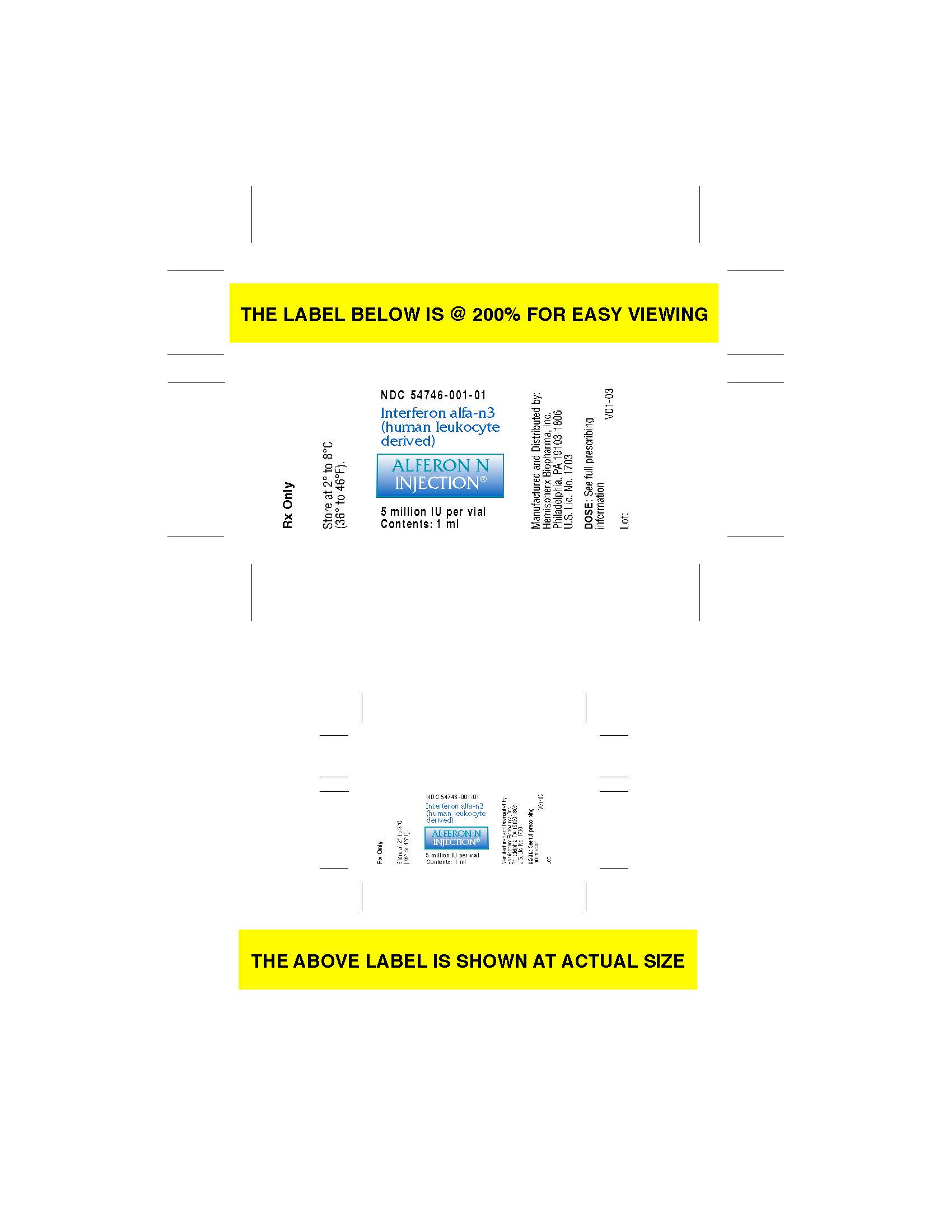
Label: ALFERON- interferon alfa-n3 injection
-
Contains inactivated NDC Code(s)
NDC Code(s): 54746-001-01 - Packager: AIM ImmunoTech Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated October 28, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Storage
- carton label
- vial label
-
INGREDIENTS AND APPEARANCE
ALFERON
interferon alfa-n3 injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54746-001 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength INTERFERON ALFA-N3 (UNII: 47BPR3V3MP) (INTERFERON ALFA-N3 - UNII:47BPR3V3MP) INTERFERON ALFA-N3 5000000 [arb'U] in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54746-001-01 00 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 10/10/1989 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA103158 10/10/1989 Labeler - AIM ImmunoTech Inc (058608076) Registrant - AIM ImmunoTech Inc (119520661) Establishment Name Address ID/FEI Business Operations AIM ImmunoTech Inc. 119520661 api manufacture(54746-001)