Label: ENDODAN- oxycodone and aspirin tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 16590-947-30, 16590-947-60, 16590-947-90 - Packager: STAT RX USA LLC
- This is a repackaged label.
- Source NDC Code(s): 60951-310
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: CII
- Marketing Status: New Drug Application Authorized Generic
Drug Label Information
Updated May 23, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Each ENDODAN Tablet contains:
Oxycodone Hydrochloride, USP 4.8355 mg 1
Aspirin, USP 325 mg
ENDODAN Tablets also contain the following inactive ingredients: D and C Yellow 10, FD and C Yellow 6, microcrystalline cellulose and corn starch.
The oxycodone hydrochloride component is Morphinan-6-one, 4,5-epoxy-14-hydroxy-3-methoxy-17-methyl-, hydrochloride, (5a)-., a white to off-white, hygroscopic crystals or powder, odorless, soluble in water; slightly soluble in alcohol and is represented by the following structural formula:
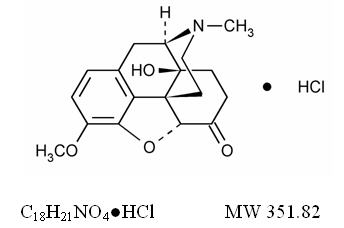
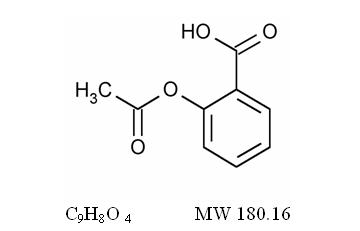
The aspirin component is 2-(acetyloxy)-, Benzoic acid, a white crystal, commonly tabular or needle-like, or white, crystalline powder. Is odorless or has a faint odor. Is stable in dry air; in moist air it gradually hydrolyzes to salicylic and acetic acids. Slightly soluble in water; freely soluble in alcohol; soluble in chloroform and in ether; sparingly soluble in absolute ether and is represented by the following structural formula:
1 4.8355 mg oxycodone HCl is equivalent to 4.3346 mg of oxycodone as the free base.
-
CLINICAL PHARMACOLOGY
Central Nervous SystemOxycodone is a semisynthetic pure opioid agonist whose principal therapeutic action is analgesia. Other pharmacological effects of oxycodone include anxiolysis, euphoria and feelings of relaxation. These effects are mediated by receptors (notably μ and κ) in the central nervous system for endogenous opioid-like compounds such as endorphins and enkephalins. Oxycodone produces respiratory depression through direct activity at respiratory centers in the brain stem and depresses the cough reflex by direct effect on the center of the medulla.
Aspirin (acetylsalicylic acid) works by inhibiting the body’s production of prostaglandins, including prostaglandins involved in inflammation. Prostaglandins cause pain sensations by stimulating muscle contractions and dilating blood vessels throughout the body. In the CNS, aspirin works on the hypothalamus heat-regulating center to reduce fever, however, other mechanisms may be involved.
Gastrointestinal Tract and Other Smooth MuscleOxycodone reduces motility by increasing smooth muscle tone in the stomach and duodenum. In the small intestine, digestion of food is delayed by decreases in propulsive contractions. Other opioid effects include contraction of biliary tract smooth muscle, spasm of the Sphincter of Oddi, increased ureteral and bladder sphincter tone, and a reduction in uterine tone.
Aspirin can produce gastrointestinal injury (lesions, ulcers) through a mechanism that is not yet completely understood, but may involve a reduction in eicosanoid synthesis by the gastric mucosa. Decreased production of prostaglandins may compromise the defenses of the gastric mucosa and the activity of substances involved in tissue repair and ulcer healing.
Cardiovascular SystemOxycodone may produce a release of histamine and may be associated with orthostatic hypotension, and other symptoms, such as pruritus, flushing, red eyes, and sweating.
Platelet AggregationAspirin affects platelet aggregation by irreversibly inhibiting prostaglandin cyclo-oxygenase. This effect lasts for the life of the platelet and prevents the formation of the platelet aggregating factor thromboxane A2. Nonacetylated salicylates do not inhibit this enzyme and have no effect on platelet aggregation. At somewhat higher doses, aspirin reversibly inhibits the formation of prostaglandin 12 (prostacyclin), which is an arterial vasodilator and inhibits platelet aggregation.
PharmacokineticsAbsorption and Distribution
The mean absolute oral bioavailability of oxycodone in cancer patients was reported to be about 87%. Oxycodone has been shown to be 45% bound to human plasma proteins in vitro. The volume of distribution after intravenous administration is 211.9 ±186.6 L.
Aspirin is hydrolyzed primarily to salicylic acid in the gut wall and during first-pass metabolism through the liver. Salicylic acid is absorbed rapidly from the stomach, but most of the absorption occurs in the proximal small intestine. Following absorption, salicylate is distributed to most body tissues and fluids, including fetal tissues, breast milk, and the CNS. High concentrations are found in the liver and kidneys. Salicylate is variably bound to serum proteins, particularly albumin.
Metabolism and Elimination
A high portion of oxycodone is N-dealkylated to noroxycodone during first-pass metabolism. Oxymorphone, is formed by the O-demethylation of oxycodone. The metabolism of oxycodone to oxymorphone is catalyzed by CYP2D6. Free and conjugated noroxycodone, free and conjugated oxycodone, and oxymorphone are excreted in human urine following a single oral dose of oxycodone. Approximately 8% to 14% of the dose is excreted as free oxycodone over 24 hours after administration. Following a single, oral dose of oxycodone, the mean ± SD elimination half-life is 3.51 ± 1.43 hours.
The biotransformation of aspirin occurs primarily in the liver by the microsomal enzyme system. With a plasma half-life of approximately 15 minutes, aspirin is rapidly hydrolyzed to salicylate. At low doses, salicylate elimination follows first-order kinetics. The plasma half-life of salicylate is approximately 2 to 3 hours.
Approximately 10% of aspirin is excreted as unchanged salicylate in the urine. The major metabolites excreted in the urine are salicyluric acid (75%), salicyl phenolic glucuronide (10%), salicyl acyl glucuronide (5%), and gentisic and gentisuric acid (less than 1%) each. Eighty to 100% of a single dose is excreted in the urine within 24 to 72 hours.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
ENDODAN tablets are contraindicated in patients with known hypersensitivity to oxycodone or aspirin, and in any situation where opioids or aspirin are contraindicated. Aspirin is contraindicated for patients with hemophilia.
Reye Syndrome: Aspirin should not be used in children or teenagers for viral infections, with or without fever, because of the risk of Reye syndrome with concomitant use of aspirin in certain viral illnesses.
Allergy: Aspirin is contraindicated in patients with known allergy to nonsteroidal anti-inflammatory drug products and in patients with the syndrome of asthma, rhinitis, and nasal polyps. Aspirin may cause severe urticaria, angioedema, or bronchospasm (asthma).
Oxycodone is contraindicated in patients with known hypersensitivity to oxycodone. Oxycodone is contraindicated in any situation where opioids are contraindicated including patients with significant respiratory depression (in unmonitored settings or the absence of resuscitative equipment) and patients with acute or severe bronchial asthma or hypercarbia. Oxycodone is contraindicated in the setting of suspected or known paralytic ileus.
-
WARNINGS
Misuse, Abuse and Diversion of OpioidsOxycodone is an opioid agonist of the morphine-type. Such drugs are sought by drug abusers and people with addiction disorders and are subject to criminal diversion.
Oxycodone can be abused in a manner similar to other opioid agonists, legal or illicit. This should be considered when prescribing or dispensing ENDODAN tablets in situations where the physician or pharmacist is concerned about an increased risk of misuse, abuse, or diversion. Concerns about misuse, addiction, and diversion should not prevent the proper management of pain.
Healthcare professionals should contact their State Professional Licensing Board, or State Controlled Substances Authority for information on how to prevent and detect abuse or diversion of this product.
Administration of ENDODAN (Oxycodone and Aspirin Tablets, USP) tablets should be closely monitored for the following potentially serious adverse reactions and complications:
Respiratory DepressionRespiratory depression is a hazard with the use of oxycodone, one of the active ingredients in ENDODAN tablets, as with all opioid agonists. Elderly and debilitated patients are at particular risk for respiratory depression as are non-tolerant patients given large initial doses of oxycodone or when oxycodone is given in conjunction with other agents that depress respiration. Oxycodone should be used with extreme caution in patients with acute asthma, chronic obstructive pulmonary disorder (COPD), cor pulmonale, or preexisting respiratory impairment. In such patients, even usual therapeutic doses of oxycodone may decrease respiratory drive to the point of apnea. In these patients alternative non-opioid analgesics should be considered, and opioids should be employed only under careful medical supervision at the lowest effective dose.
In case of respiratory depression, a reversal agent such as naloxone hydrochloride may be utilized (see OVERDOSAGE ).
Head Injury and Increased Intracranial PressureThe respiratory depressant effects of opioids include carbon dioxide retention and secondary elevation of cerebrospinal fluid pressure, and may be markedly exaggerated in the presence of head injury, other intracranial lesions or a pre-existing increase in intracranial pressure. Oxycodone produces effects on pupillary response and consciousness which may obscure neurologic signs of worsening in patients with head injuries.
Hypotensive EffectOxycodone may cause severe hypotension particularly in individuals whose ability to maintain blood pressure has been compromised by a depleted blood volume, or after concurrent administration with drugs which compromise vasomotor tone such as phenothiazines. Oxycodone, like all opioid analgesics of the morphine-type, should be administered with caution to patients in circulatory shock, since vasodilation produced by the drug may further reduce cardiac output and blood pressure. Oxycodone may produce orthostatic hypotension in ambulatory patients.
Alcohol WarningPatients who consume three or more alcoholic drinks every day should be counseled about the bleeding risks involved with chronic, heavy alcohol use while taking aspirin.
Coagulation AbnormalitiesEven low doses of aspirin can inhibit platelet function leading to an increase in bleeding time. This can adversely affect patients with inherited (hemophilia) or acquired (liver disease or vitamin K deficiency) bleeding disorders.
GI Side EffectsGI side effects include stomach pain, heartburn, nausea, vomiting, and gross GI bleeding. Although minor upper GI symptoms, such as dyspepsia, are common and can occur anytime during therapy, physicians should remain alert for signs of ulceration and bleeding, even in the absence of previous GI symptoms. Physicians should inform patients about the signs and symptoms of GI side effects and what steps to take if they occur.
Peptic Ulcer DiseasePatients with a history of active peptic ulcer disease should avoid using aspirin, which can cause gastric mucosal irritation and bleeding.
-
PRECAUTIONS
GeneralOpioid analgesics should be used with caution when combined with CNS depressant drugs, and should be reserved for cases where the benefits of opioid analgesia outweigh the known risks of respiratory depression, altered mental state, and postural hypotension.
ENDODAN tablets should be given with caution to patients with CNS depression, elderly or debilitated patients, patients with severe impairment of hepatic, pulmonary, or renal function, hypothyroidism, Addison's disease, prostatic hypertrophy, urethral stricture, acute alcoholism, delirium tremens, kyphoscoliosis with respiratory depression, myxedema, and toxic psychosis.
ENDODAN tablets may obscure the diagnosis or clinical course in patients with acute abdominal conditions. Oxycodone may aggravate convulsions in patients with convulsive disorders, and all opioids may induce or aggravate seizures in some clinical settings.
Following administration of ENDODAN tablets, anaphylactic reactions have been reported in patients with a known hypersensitivity to codeine, a compound with a structure similar to morphine and oxycodone. The frequency of this possible cross-sensitivity is unknown.
Aspirin has been associated with elevated hepatic enzymes, blood urea nitrogen and serum creatinine, hyperkalemia, proteinuria, and prolonged bleeding time.
HemorrhageAspirin may increase the likelihood of hemorrhage due to its effect on the gastric mucosa and platelet function (prolongation of bleeding time). Salicylates should be used with caution in the presence of peptic ulcer or coagulation abnormalities.
PregnancyAspirin can cause fetal harm when administered to a pregnant woman. Salicylates readily cross the placenta and by inhibiting prostaglandin synthesis, may cause constriction of ductus arteriosus, resulting in pulmonary hypertension and increased fetal mortality and, possibly other untoward fetal effects. Aspirin use in pregnancy can also result in alteration in maternal and neonatal hemostasis mechanisms. Maternal aspirin use during later stages of pregnancy may cause low birth weight, increased incidence of intracranial hemorrhage in premature infants, stillbirths and neonatal death. The use of aspirin during pregnancy especially in the third trimester should be avoided. If ENDODAN tablets are used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Renal FailureAvoid aspirin in patients with severe renal failure (glomerular filtration rate less than 10 mL/minute).
Hepatic InsufficiencyAvoid aspirin in patients with severe hepatic insufficiency.
Interactions with Other CNS DepressantsPatients receiving other opioid analgesics, general anesthetics, phenothiazines, other tranquilizers, centrally-acting anti-emetics, sedative-hypnotics or other CNS depressants (including alcohol) concomitantly with ENDODAN tablets may exhibit an additive CNS depression. When such combined therapy is contemplated, the dose of one or both agents should be reduced.
Interactions with Mixed Agonist/Antagonist Opioid AnalgesicsAgonist/antagonist analgesics (i.e., pentazocine, nalbuphine, and butorphanol) should be administered with caution to a patient who has received or is receiving a course of therapy with a pure opioid agonist analgesic such as oxycodone. In this situation, mixed agonist/antagonist analgesics may reduce the analgesic effect of oxycodone and/or may precipitate withdrawal symptoms in these patients.
Ambulatory Surgery and Postoperative UseOxycodone and other morphine-like opioids have been shown to decrease bowel motility. Ileus is a common postoperative complication, especially after intra-abdominal surgery with use of opioid analgesia. Caution should be taken to monitor for decreased bowel motility in postoperative patients receiving opioids. Standard supportive therapy should be implemented.
Use in Pancreatic/Biliary Tract DiseaseOxycodone may cause spasm of the sphincter of Oddi and should be used with caution in patients with biliary tract disease, including acute pancreatitis. Opioids like oxycodone may cause increases in the serum amylase level.
Tolerance and Physical DependenceTolerance is the need for increasing doses of opioids to maintain a defined effect such as analgesia (in the absence of disease progression or other external factors). Physical dependence is manifested by withdrawal symptoms after abrupt discontinuation of a drug or upon administration of an antagonist. Physical dependence and tolerance are not unusual during chronic opioid therapy.
The opioid abstinence or withdrawal syndrome is characterized by some or all of the following: restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other symptoms also may develop, including: irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory, respiratory rate, or heart rate.
In general, opioids should not be abruptly discontinued (see DOSAGE AND ADMINISTRATION: Cessation of Therapy).
Information for Patients/Caregivers
The following information should be provided to patients receiving ENDODAN tablets by their physician, nurse, pharmacist, or caregiver:
- Patients should be aware that ENDODAN tablets contain oxycodone, which is a morphine-like substance.
- Patients should be instructed to keep ENDODAN tablets in a secure place out of the reach of children. In the case of accidental ingestions, emergency medical care should be sought immediately.
- When ENDODAN tablets are no longer needed, the unused tablets should be destroyed by flushing down the toilet.
- Patients should be advised not to adjust the medication dose themselves. Instead, they must consult with their prescribing physician.
- Patients should be advised that ENDODAN tablets may impair mental and/or physical ability required for the performance of potentially hazardous tasks (e.g., driving, operating heavy machinery).
- Patients should not combine ENDODAN tablets with alcohol, opioid analgesics, tranquilizers, sedatives, or other CNS depressants unless under the recommendation and guidance of a physician. When co-administered with another CNS depressant, ENDODAN tablets can cause dangerous additive central nervous system or respiratory depression, which can result in serious injury or death.
- The safe use of ENDODAN tablets during pregnancy has not been established; thus, women who are planning to become pregnant or are pregnant should consult with their physician before taking ENDODAN tablets.
- Nursing mothers should consult with their physicians about whether to discontinue nursing or discontinue ENDODAN tablets because of the potential for serious adverse reactions to nursing infants.
- Patients who are treated with ENDODAN tablets for more than a few weeks should be advised not to abruptly discontinue the medication. Patients should consult with their physician for a gradual discontinuation dose schedule to taper off the medication.
- Patients should be advised that ENDODAN tablets are a potential drug of abuse. They should protect it from theft, and it should never be given to anyone other than the individual for whom it was prescribed.
- Patients should be advised that ENDODAN tablets may cause or worsen constipation. They should discuss any past history of constipation with their prescribing physician so a management plan may be initiated.
Laboratory Tests
Although oxycodone may cross-react with some drug urine tests, no available studies were found which determined the duration of detectability of oxycodone in urine drug screens. However, based on pharmacokinetic data, the approximate duration of detectability for a single dose of oxycodone is roughly estimated to be one to two days following drug exposure.
Urine testing for opiates may be performed to determine illicit drug use and for medical reasons such as evaluation of patients with altered states of consciousness or monitoring efficacy of drug rehabilitation efforts. The preliminary identification of opiates in urine involves the use of an immunoassay screening and thin-layer chromatography (TLC). Gas chromatography/mass spectrometry (GC/MS) may be utilized as a third-stage identification step in the medical investigational sequence for opiate testing after immunoassay and TLC. The identities of 6-keto opiates (e.g., oxycodone) can further be differentiated by the analysis of their methoxime-trimethylsilyl (MO-TMS) derivative.
Drug/Drug Interactions with OxycodoneOpioid analgesics may enhance the neuromuscular-blocking action of skeletal muscle relaxants and produce an increase in the degree of respiratory depression.
Patients receiving CNS depressants such as other opioid analgesics, general anesthetics, phenothiazines, other tranquilizers, centrally-acting anti-emetics, sedative-hypnotics or other CNS depressants (including alcohol) concomitantly with ENDODAN tablets may exhibit an additive CNS depression. When such combined therapy is contemplated, the dose of one or both agents should be reduced.
Agonist/antagonist analgesics (i.e., pentazocine, nalbuphine, naltrexone, and butorphanol) should be administered with caution to a patient who has received or is receiving a pure opioid agonist such as oxycodone. These agonist/antagonist analgesics may reduce the analgesic effect of oxycodone or may precipitate withdrawal symptoms.
Drug/Drug Interactions with AspirinAngiotensin Converting Enzyme (ACE) Inhibitors: The hyponatremic and hypotensive effects of ACE inhibitors may be diminished by the concomitant administration of aspirin due to its indirect effect on the renin-angiotensin conversion pathway.
Acetazolamide: Concurrent use of aspirin and acetazolamide can lead to high serum concentrations of acetazolamide (and toxicity) due to competition at the renal tubule for secretion.
Anticoagulant Therapy (Heparin and Warfarin): Patients on anticoagulation therapy are at increased risk for bleeding because of drug-drug interactions and the effect on platelets. Aspirin can displace warfarin from protein binding sites, leading to prolongation of both the prothrombin time and the bleeding time. Aspirin can increase the anticoagulant activity of heparin, increasing bleeding risk.
Anticonvulsants: Salicylate can displace protein-bound phenytoin and valproic acid, leading to a decrease in the total concentration of phenytoin and an increase in serum valproic acid levels.
Beta Blockers: The hypotensive effects of beta blockers may be diminished by the concomitant administration of aspirin due to inhibition of renal prostaglandins, leading to decreased renal blood flow, and salt and fluid retention.
Diuretics: The effectiveness of diuretics in patients with underlying renal or cardiovascular disease may be diminished by the concomitant administration of aspirin due to inhibition of renal prostaglandins, leading to decreased renal blood flow and salt and fluid retention.
Methotrexate: Aspirin may enhance the serious side and toxicity of methotrexate due to displacement from its plasma protein binding sites and/or reduced renal clearance.
Nonsteroidal Anti-inflammatory Drugs (NSAID's): The concurrent use of aspirin with other NSAID's should be avoided because this may increase bleeding or lead to decreased renal function. Aspirin may enhance the serious side effects and toxicity of ketorolac, due to displacement from its plasma protein binding sites and/or reduced renal clearance.
Oral Hypoglycemics Agents: Aspirin may increase the serum glucose-lowering action of insulin and sulfonylureas leading to hypoglycemia.
Uricosuric Agents: Salicylates antagonize the uricosuric action of probenecid or sulfinpyrazone.
Drug/Laboratory Test InteractionsDepending on the sensitivity/specificity and the test methodology, the individual components of ENDODAN tablets may cross-react with assays used in the preliminary detection of cocaine (primary urinary metabolite, benzoylecgonine) or marijuana (cannabinoids) in human urine. A more specific alternate chemical method must be used in order to obtain a confirmed analytical result. The preferred confirmatory method is gas chromatography/mass spectrometry (GC/MS). Moreover, clinical considerations and professional judgment should be applied to any drug-of-abuse test result, particularly when preliminary positive results are used.
Salicylates may increase the protein bound iodine (PBI) result by competing for the protein binding sites on pre-albumin and possibly thyroid-binding globulins.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Animal studies to evaluate the carcinogenic potential of oxycodone and aspirin have not been performed.
Mutagenesis
The combination of oxycodone and aspirin has not been evaluated for mutagenicity. Oxycodone alone was negative in a bacterial reverse mutation assay (Ames), an in vitro chromosome aberration assay with human lymphocytes without metabolic activation and an in vivo mouse micronucleus assay. Oxycodone was clastogenic in the human lymphocyte chromosomal assay in the presence of metabolic activation and in the mouse lymphoma assay with or without metabolic activation. Aspirin induced chromosome aberrations in cultured human fibroblasts.
Fertility
Animal studies to evaluate the effects of oxycodone on fertility have not been performed. Aspirin has been shown to inhibit ovulation in rats.
Pregnancy
Teratogenic Effects
Oxycodone: Pregnancy Category BReproduction studies in rats and rabbits demonstrated that oral administration of oxycodone was not teratogenic or embryo-fetal toxic.
Aspirin: Pregnancy Category D (see PRECAUTIONS)
Salicylates readily cross the placenta and by inhibiting prostaglandin synthesis, may cause constriction of ductus arteriosus resulting in pulmonary hypertension and increased fetal mortality and, possibly other untoward fetal effects. Aspirin use in pregnancy can also result in alteration in maternal and neonatal hemostasis mechanisms. Maternal aspirin use during later stages of pregnancy may cause low birth weight, increased incidence of intracranial hemorrhage in premature infants, stillbirths and neonatal death. Use during pregnancy, especially in the third trimester, should be avoided.
Safe use of ENDODAN (Oxycodone and Aspirin Tablets, USP) in pregnancy has not been established relative to possible adverse effects on fetal development. Therefore, ENDODAN tablets should not be used in pregnant women unless, in the judgment of the physician, the potential benefits outweigh the possible hazards.
Nonteratogenic EffectsOpioids can cross the placental barrier and have the potential to cause neonatal respiratory depression. Opioid use during pregnancy may result in a physically drug-dependent fetus. After birth, the neonate may suffer severe withdrawal symptoms. Aspirin may produce anemia, ante- or postpartum hemorrhage, prolonged gestation and labor, and oligohydramnios.
Labor and DeliveryENDODAN tablets are not recommended for use in women during and immediately prior to labor and delivery due to its potential effects on respiratory function in the newborn. Aspirin should be avoided one week prior to and during labor and delivery because it can result in excessive blood loss at delivery. Prolonged gestation and prolonged labor due to prostaglandin inhibition have been reported.
Nursing MothersOrdinarily, nursing should not be undertaken while a patient is receiving ENDODAN tablets because of the possibility of sedation and/or respiratory depression in the infant. Oxycodone is excreted in breast milk in low concentrations, and there have been rare reports of somnolence and lethargy in babies of nursing mothers taking an oxycodone/acetaminophen product. Salicylic acid has also been detected in breast milk. Adverse effects on platelet function in the nursing infant exposed to aspirin in breast milk may be a potential risk. Furthermore, the risk of Reye Syndrome caused by salicylate in breast milk is unknown. Because of the potential for serious adverse reactions in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the potential benefits to the woman and the possible hazards to the nursing infant.
Pediatric UseENDODAN tablets should not be administered to pediatric patients. Reye Syndrome is a rare but serious disease which can follow flu or chicken pox in children and teenagers. While the cause of Reye Syndrome is unknown, some reports claim aspirin (or salicylates) may increase the risk of developing this disease.
Geriatric UseSpecial precaution should be given when determining the dosing amount and frequency of ENDODAN tablets for geriatric patients, since clearance of oxycodone may be slightly reduced in this patient population when compared to younger patients.
Hepatic ImpairmentIn a pharmacokinetic study of oxycodone in patients with end-stage liver disease, oxycodone plasma clearance decreased and the elimination half-life increased. Care should be exercised when oxycodone is used in patients with hepatic impairment.
Renal ImpairmentIn a study of patients with end stage renal impairment, mean elimination half-life was prolonged in uremic patients due to increased volume of distribution and reduced clearance. Oxycodone should be used with caution in patients with renal impairment.
-
ADVERSE REACTIONS
Serious adverse reactions that may be associated with ENDODAN tablet use include respiratory depression, apnea, respiratory arrest, circulatory depression, hypotension, and shock (see OVERDOSAGE).
The most frequently observed non-serious adverse reactions include lightheadedness, dizziness, drowsiness or sedation, nausea, and vomiting. These effects seem to be more prominent in ambulatory than in nonambulatory patients, and some of these adverse reactions may be alleviated if the patient lies down. Other adverse reactions include euphoria, dysphoria, constipation and pruritus.
Aspirin may increase the likelihood of hemorrhage due to its effect on the gastric mucosa and platelet function. Furthermore, aspirin has the potential to cause anaphylaxis in hypersensitive patients as well as angioedema especially in patients with chronic urticaria. Other adverse reactions due to aspirin use include anorexia, reversible hepatotoxicity, leukopenia, thrombocytopenia, purpura, decreased plasma iron concentration, and shortened erythrocyte survival time.
Other adverse reactions obtained from postmarketing experiences with ENDODAN tablets are listed by organ system and in decreasing order of severity and/or frequency as follows:
Body as a Whole
allergic reaction, malaise, asthenia, headache, anaphylaxis, fever, hypothermia, thirst, increased sweating, accident, accidental overdose, non-accidental overdose.
Cardiovascular
tachycardia, dysrhythmias, hypotension, orthostatic hypotension, bradycardia, palpitations
Central and Peripheral Nervous System
stupor, paresthesia, agitation, cerebral edema, coma, confusion, dizziness, headache, subdural or intracranial hemorrhage, lethargy, seizures, anxiety, mental impairment
Fluid and Electrolyte
dehydration, hyperkalemia, metabolic acidosis, respiratory alkalosis
Gastrointestinal
hemorrhagic gastric/duodenal ulcer, gastric/peptic ulcer, dyspepsia, abdominal pain, diarrhea, eructation, dry mouth, gastrointestinal bleeding, intestinal perforation, nausea, vomiting, transient elevations of hepatic enzymes, hepatitis, Reye syndrome, pancreatitis, intestinal obstruction, ileus
Hearing and Vestibular
hearing loss, tinnitus. Patients with high frequency loss may have difficulty perceiving tinnitus. In these patients, tinnitus cannot be used as a clinical indicator of salicylism.
Hematologic
unspecified hemorrhage, purpura, reticulocytosis, prolongation of prothrombin time, disseminated intravascular coagulation, ecchymosis, thrombocytopenia.
Hypersensitivity
acute anaphylaxis, angioedema, asthma, bronchospasm, laryngeal edema, urticaria, anaphylactoid reaction
Metabolic and Nutritional
hypoglycemia, hyperglycemia, acidosis, alkalosis
Musculoskeletal
rhabdomyolysis
Ocular
miosis, visual disturbances, red eye.
Psychiatric
drug dependence, drug abuse, somnolence, depression, nervousness, hallucination
Reproductive
prolonged pregnancy and labor, stillbirths, lower birth weight infants, antepartum and postpartum bleeding, closure of patent ductus arteriosis
Respiratory System
bronchospasm, dyspnea, hyperpnea, pulmonary edema, tachypnea, aspiration, hypoventilation, laryngeal edema
Skin and Appendages
urticaria, rash, flushing
Urogenital
interstitial nephritis, papillary necrosis, proteinuria, renal insufficiency and failure, urinary retention
-
OVERDOSAGE
Signs and SymptomsSerious overdose with ENDODAN (Oxycodone and Aspirin Tablets, USP) is characterized by signs and symptoms of opioid and salicylate overdose. Oxycodone overdosage can be manifested by respiratory depression (a decrease in respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis), extreme somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, pupillary constriction (pupils may be dilated in the setting of hypoxia), and sometimes bradycardia and hypotension. In severe overdosage, apnea, circulatory collapse, cardiac arrest and death may occur. Early signs of acute aspirin (salicylate) overdose including tinnitus occur at plasma concentrations approaching 200 mcg/mL. Plasma concentrations of aspirin above 300 mcg/mL are toxic. Severe toxic effects are associated with levels above 400 mcg/mL. A single lethal dose of aspirin in adults is not known with certainty but death may be expected at 30 g. For real or suspected overdose, a Poison Control Center should be contacted immediately.
In acute salicylate overdose, severe acid-base and electrolyte disturbances may occur and are complicated by hyperthermia and dehydration, and coma. Respiratory alkalosis occurs early while hyperventilation is present, but is quickly followed by metabolic acidosis. Serious symptoms such as depression, coma, and respiratory failure progress rapidly.
Salicylism (chronic salicylate toxicity) may be noted by symptoms such as dizziness, tinnitus, difficulty hearing, nausea, vomiting, diarrhea, and mental confusion. More severe salicylism may result in respiratory alkalosis.
TreatmentPrimary attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and the institution of assisted or controlled ventilation. Supportive measures (including oxygen, intravenous fluids, and vasopressors) should be employed in the management of circulatory shock and pulmonary edema accompanying overdose as indicated. Cardiac arrest or arrhythmias may require cardiac massage or defibrillation. Treatment of acid-base disturbances and electrolyte disorders is also important. Because of the concern over salicylate toxicity, acid-base status should be followed closely with serial blood gas and serum pH determinations.
The opioid antagonist naloxone hydrochloride is a specific antidote against respiratory depression which may result from overdosage or unusual sensitivity to opioids including oxycodone. Therefore, an appropriate dose of naloxone hydrochloride should be administered (usual initial adult dose 0.4 mg-2 mg) preferably by the intravenous route, simultaneously with efforts at respiratory resuscitation. Since the duration of action of oxycodone may exceed that of the antagonist, the patient should be kept under continued surveillance and repeated doses of the antagonist should be administered as needed to maintain adequate respiration. Opioid antagonists should not be administered in the absence of clinically significant respiratory or circulatory depression secondary to oxycodone overdose. In patients who are physically dependent on any opioid agonist including oxycodone, an abrupt or complete reversal of opioid effects may precipitate an acute abstinence syndrome. The severity of the withdrawal syndrome produced will depend on the degree of physical dependence and the dose of the antagonist administered. Please see the prescribing information for the specific opioid antagonist for details of their proper use.
Gastric emptying and/or lavage may be useful in removing unabsorbed drug. This procedure is recommended as soon as possible after ingestion, even if the patient has vomited spontaneously. After lavage and/or emesis, administration of activated charcoal, as a slurry, is beneficial, if less than three hours have passed since ingestion. Charcoal adsorption should not be employed prior to lavage and emesis.
In severe cases of salicylate overdose, hyperthermia and hypovolemia are the major immediate threats to life. Children should be sponged with tepid water. Replacement fluid should be administered intravenously and augmented with correction of acidosis. Plasma electrolytes and pH should be monitored to promote alkaline diuresis of salicylate if renal function is normal. Infusion of glucose may be required to control hypoglycemia. With more severe acute toxicity respiratory alkalosis may occur.
Hemodialysis and peritoneal dialysis can be performed to reduce the body content of aspirin. In patients with renal insufficiency or in cases of life-threatening salicylate intoxication dialysis is usually required. Exchange transfusion may be indicated in infants and young children.
In case of real or suspected overdose, a poison control center should be consulted for the treatment of salicylism.
The toxicity of oxycodone and aspirin in combination is unknown.
-
DOSAGE AND ADMINISTRATION
Dosage should be adjusted according to the severity of the pain and the response of the patient. It may occasionally be necessary to exceed the usual dosage recommended below in cases of more severe pain or in those patients who have become tolerant to the analgesic effect of opioids. If pain is constant, the opioid analgesic should be given at regular intervals on an around-the-clock schedule. ENDODAN tablets are given orally.
The usual dosage is one tablet every 6 hours as needed for pain. The maximum daily dose of aspirin should not exceed 4 grams or 12 tablets.
Cessation of TherapyIn patients treated with ENDODAN tablets for more than a few weeks who no longer require therapy, doses should be tapered gradually to prevent signs and symptoms of withdrawal in the physically dependent patient.
-
DRUG ABUSE AND DEPENDENCE
ENDODAN tablets are a Schedule II controlled substance. Oxycodone is a mu-agonist opioid with an abuse liability similar to morphine. Oxycodone, like morphine and other opioids used in analgesia, can be abused and is subject to criminal diversion.
Drug addiction is defined as an abnormal, compulsive use, use for non-medical purposes of a substance despite physical, psychological, occupational or interpersonal difficulties resulting from such use, and continued use despite harm or risk of harm. Drug addiction is a treatable disease, utilizing a multi-disciplinary approach, but relapse is common. Opioid addiction is relatively rare in patients with chronic pain but may be more common in individuals who have a past history of alcohol or substance abuse or dependence. Pseudoaddiction refers to pain relief seeking behavior of patients whose pain is poorly managed. It is considered an iatrogenic effect of ineffective pain management. The health care provider must assess continuously the psychological and clinical condition of a pain patient in order to distinguish addiction from pseudoaddiction and thus, be able to treat the pain adequately.
Physical dependence on a prescribed medication does not signify addiction. Physical dependence involves the occurrence of a withdrawal syndrome when there is sudden reduction or cessation in drug use or if an opiate antagonist is administered. Physical dependence can be detected after a few days of opioid therapy. However, clinically significant physical dependence is only seen after several weeks of relatively high dosage therapy. In this case, abrupt discontinuation of the opioid may result in a withdrawal syndrome. If the discontinuation of opioids is therapeutically indicated, gradual tapering of the drug over a 2-week period will prevent withdrawal symptoms. The severity of the withdrawal syndrome depends primarily on the daily dosage of the opioid, the duration of therapy and medical status of the individual.
The withdrawal syndrome of oxycodone is similar to that of morphine. This syndrome is characterized by yawning, anxiety, increased heart rate and blood pressure, restlessness, nervousness, muscle aches, tremor, irritability, chills alternating with hot flashes, salivation, anorexia, severe sneezing, lacrimation, rhinorrhea, dilated pupils, diaphoresis, piloerection, nausea, vomiting, abdominal cramps, diarrhea and insomnia, and pronounced weakness and depression.
“Drug-seeking” behavior is very common in addicts and drug abusers. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing or referral, repeated “loss” of prescriptions, tampering with prescriptions and reluctance to provide prior medical records or contact information for other treating physician(s). “Doctor shopping” to obtain additional prescriptions is common among drug abusers and people suffering from untreated addiction.
Abuse and addiction are separate and distinct from physical dependence and tolerance. Physicians should be aware that addiction may not be accompanied by concurrent tolerance and symptoms of physical dependence in all addicts. In addition, abuse of opioids can occur in the absence of true addiction and is characterized by misuse for non-medical purposes, often in combination with other psychoactive substances. Oxycodone, like other opioids, has been diverted for non-medical use. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests is strongly advised.
Proper assessment of the patient, proper prescribing practices, periodic re-evaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
Like other opioid medications, ENDODAN tablets are subject to the Federal Controlled Substances Act. After chronic use, ENDODAN tablets should not be discontinued abruptly when it is thought that the patient has become physically dependent on oxycodone.
Interactions with Alcohol and Drugs of AbuseOxycodone may be expected to have additive effects when used in conjunction with alcohol, other opioids, or illicit drugs that cause central nervous system depression.
-
HOW SUPPLIED
ENDODAN (oxycodone and aspirin tablets, USP), supplied as a yellow round tablet, with one face scored and the other debossed with “Endo” and “610”.
Available in:
Bottles of 100 NDC 60951-310-70
Store at 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F). [See USP Controlled Room Temperature.]
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
DEA Order Form Required.
Manufactured for:
Endo Pharmaceuticals Inc.
Chadds Ford, Pennsylvania 19317
ENDODAN® is a Registered Trademark of Endo Pharmaceuticals
© 2009 Endo Pharmaceuticals
Printed in U.S.A. 2005701/September, 2009
- PACKAGE LABEL - ENDODAN C-II 4.8355/325 MG TABLET
-
INGREDIENTS AND APPEARANCE
ENDODAN
oxycodone and aspirin tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:16590-947(NDC:60951-310) Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OXYCODONE HYDROCHLORIDE (UNII: C1ENJ2TE6C) (OXYCODONE - UNII:CD35PMG570) OXYCODONE HYDROCHLORIDE 4.8355 mg ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 325 mg Inactive Ingredients Ingredient Name Strength D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color yellow Score 2 pieces Shape ROUND Size 11mm Flavor Imprint Code Endo;610 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16590-947-30 30 in 1 BOTTLE 2 NDC:16590-947-60 60 in 1 BOTTLE 3 NDC:16590-947-90 90 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA authorized generic NDA007337 05/20/1994 Labeler - STAT RX USA LLC (786036330) Establishment Name Address ID/FEI Business Operations STAT RX USA LLC 786036330 relabel, repack


