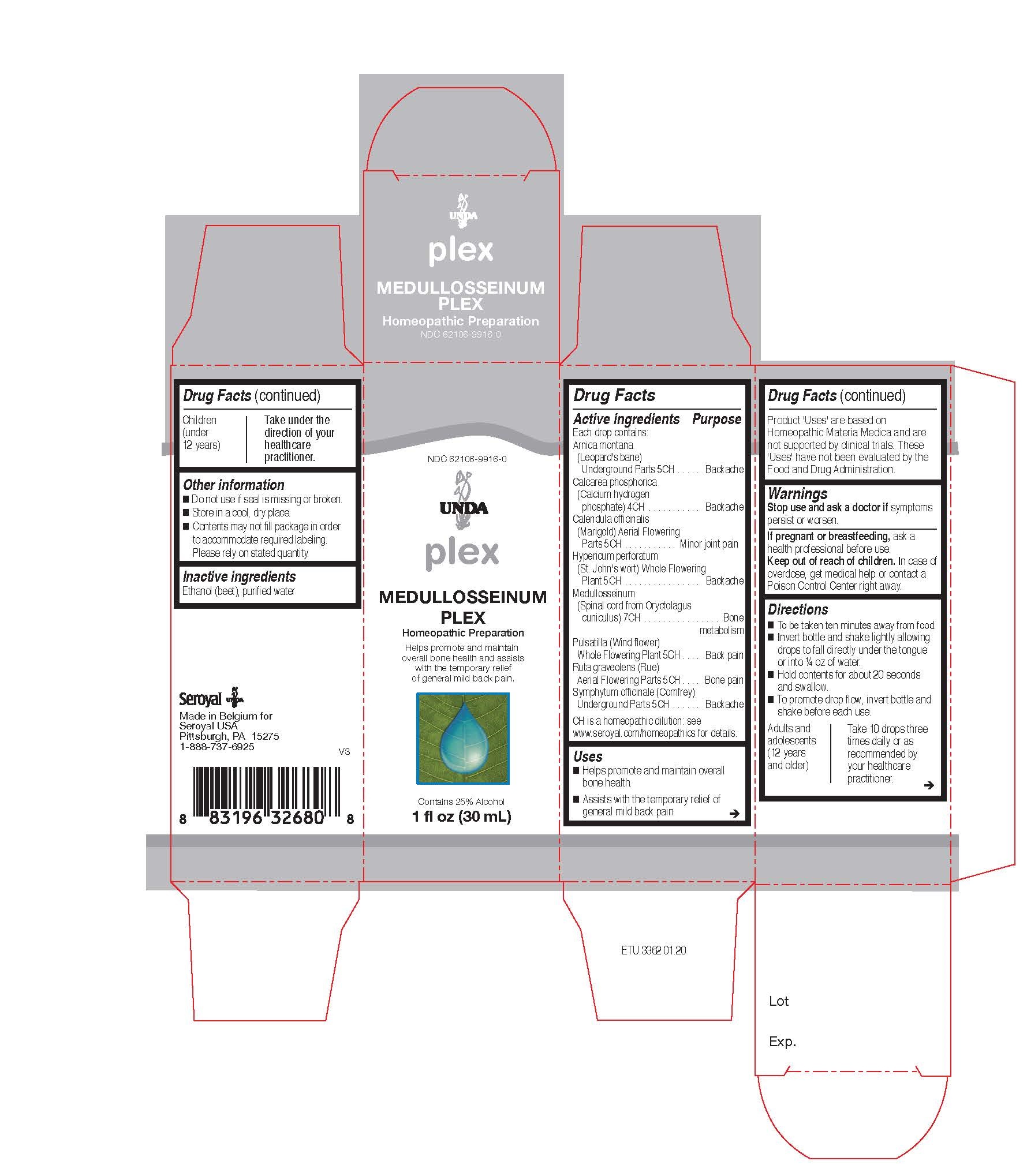
Label: MEDULLOSSEINUM PLEX- arnica montana, calcarea phosphorica, calendula officinalis, hypericum perforatum, medullosseinum, pulsatilla, ruta graveolens, symphytum officinale liquid
- NDC Code(s): 62106-9916-0
- Packager: Seroyal USA
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated October 9, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active ingredients
Each drop contains equal parts of:
Arnica montana (Leopard's bane) Underground Parts 5CH
Calcarea phosphorica (Calcium hydrogen phosphate) 4CH
Calendula officinalis (Marigold) Flowering Top 5CH
Hypericum perforatum (St. John's wort) Whole Plant 5CH
Medullosseinum (Spinal cord from Oryctolagus cuniculus) 7CH
Pulsatilla (Wind flower) Whole Flowering Plant 5CH
Ruta graveolens (Rue) Aerial Parts 5CH
Symphytum officinale (Comfrey) Underground Parts 5CH - PURPOSE
- WARNINGS
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- OVERDOSAGE
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
-
DOSAGE & ADMINISTRATION
Directions
To be taken ten minutes away from food.
Invert bottle and shake lightly allowing drops to fall directly under the tongue or into ¼ oz of water.
Hold contents for about 20 seconds and swallow.
To promote drop flow, invert bottle and shake before each use.Adults and adolescents (12 years and older)
Take 10 drops three times daily or as recommended by your healthcare practitioner.
Children (under 12 years)
Take under the direction of your healthcare practitioner.
-
INDICATIONS & USAGE
Uses
Helps promote and maintain overall bone health.
Assists with the temporary relief of general mild back pain.Directions
To be taken ten minutes away from food.
Invert bottle and shake lightly allowing drops to fall directly under the tongue or into ¼ oz of water.
Hold contents for about 20 seconds and swallow.
To promote drop flow, invert bottle and shake before each use.Adults and adolescents (12 years and older)
Take 10 drops three times daily or as recommended by your healthcare practitioner.
Children (under 12 years)
Take under the direction of your healthcare practitioner.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MEDULLOSSEINUM PLEX
arnica montana, calcarea phosphorica, calendula officinalis, hypericum perforatum, medullosseinum, pulsatilla, ruta graveolens, symphytum officinale liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62106-9916 Route of Administration SUBLINGUAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYPERICUM PERFORATUM (UNII: XK4IUX8MNB) (HYPERICUM PERFORATUM - UNII:XK4IUX8MNB) HYPERICUM PERFORATUM 5 [hp_C] in 30 mL ARNICA MONTANA ROOT (UNII: MUE8Y11327) (ARNICA MONTANA ROOT - UNII:MUE8Y11327) ARNICA MONTANA ROOT 5 [hp_C] in 30 mL CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: L11K75P92J) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS 4 [hp_C] in 30 mL COMFREY ROOT (UNII: M9VVZ08EKQ) (COMFREY ROOT - UNII:M9VVZ08EKQ) COMFREY ROOT 5 [hp_C] in 30 mL PULSATILLA VULGARIS (UNII: I76KB35JEV) (PULSATILLA VULGARIS - UNII:I76KB35JEV) PULSATILLA VULGARIS 5 [hp_C] in 30 mL RUTA GRAVEOLENS FLOWERING TOP (UNII: N94C2U587S) (RUTA GRAVEOLENS FLOWERING TOP - UNII:N94C2U587S) RUTA GRAVEOLENS FLOWERING TOP 5 [hp_C] in 30 mL CALENDULA OFFICINALIS FLOWERING TOP (UNII: 18E7415PXQ) (CALENDULA OFFICINALIS FLOWERING TOP - UNII:18E7415PXQ) CALENDULA OFFICINALIS FLOWERING TOP 5 [hp_C] in 30 mL ORYCTOLAGUS CUNICULUS SPINAL CORD (UNII: 49SKT79UB3) (ORYCTOLAGUS CUNICULUS SPINAL CORD - UNII:49SKT79UB3) ORYCTOLAGUS CUNICULUS SPINAL CORD 7 [hp_C] in 30 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62106-9916-0 1 in 1 CARTON 10/22/2015 1 30 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 10/22/2015 Labeler - Seroyal USA (018361118) Establishment Name Address ID/FEI Business Operations SAN'UP 401010287 manufacture(62106-9916)