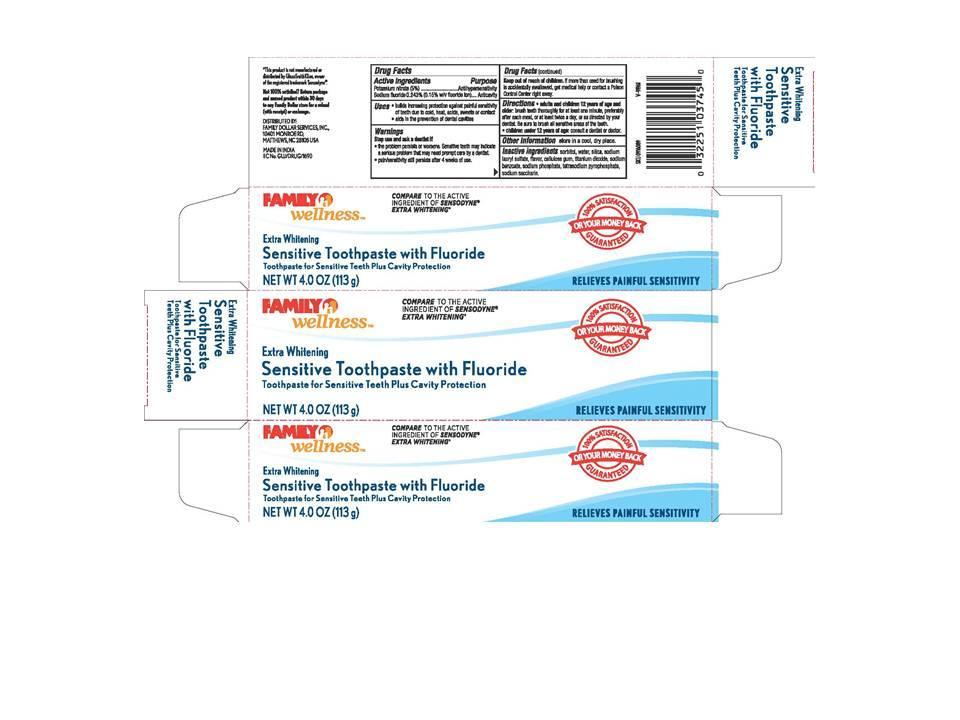
Label: FAMILY WELLNESS SENSITIVE EXTRA WHITENING- potassium nitrate and sodium fluoride paste
-
Contains inactivated NDC Code(s)
NDC Code(s): 55319-900-01 - Packager: Family Wellness
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 11, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- USES
- Purpose
- WARNINGS
- Keep out of reach of children
-
Directions
Adults and children 12 years and older Brush teeth for at least one minute, preferably after each meal, or at least twice a day or as directed by your dentist. Be sure to brush sensitive areas of the teeth.
Children under 12 years of age Consult a dentist or doctor
Other information Store in a cool, dry place.
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FAMILY WELLNESS SENSITIVE EXTRA WHITENING
potassium nitrate and sodium fluoride pasteProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55319-900 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POTASSIUM NITRATE (UNII: RU45X2JN0Z) (NITRATE ION - UNII:T93E9Y2844) POTASSIUM NITRATE 5 mg in 1 g SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 1.5 mg in 1 g Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) WATER (UNII: 059QF0KO0R) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM PHOSPHATE (UNII: SE337SVY37) SACCHARIN SODIUM (UNII: SB8ZUX40TY) Product Characteristics Color white (Sensitive) Score Shape Size Flavor MINT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55319-900-01 113 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 04/11/2013 Labeler - Family Wellness (024472631)