Label: TYPHIM VI- salmonella typhi ty2 vi polysaccharide antigen injection, solution
- NDC Code(s): 50090-1758-0, 50090-1758-9
- Packager: A-S Medication Solutions
- This is a repackaged label.
- Source NDC Code(s): 49281-790
- Category: VACCINE LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated October 22, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Typhim Vi®, Typhoid Vi Polysaccharide Vaccine, produced by Sanofi Pasteur SA, for intramuscular use, is a sterile solution containing the cell surface Vi polysaccharide extracted from Salmonella enterica serovar Typhi, S typhi Ty2 strain. The organism is grown in a semi-synthetic medium. Casein derived raw materials are used early in manufacturing during the fermentation process. The capsular polysaccharide is precipitated from the concentrated culture supernatant by the addition of hexadecyltrimethylammonium bromide, and the product is purified by differential centrifugation and precipitation. Each 0.5 mL dose may contain residual amounts of formaldehyde (not more than 100 mcg) used for the inactivation of the bacterial culture. The potency of the purified polysaccharide is assessed by molecular size and O-acetyl content. Phenol, 0.25%, is added as a preservative. The vaccine contains residual polydimethylsiloxane or fatty-acid ester-based antifoam. The vaccine is a clear, colorless solution. Each dose of 0.5 mL is formulated to contain 25 mcg of purified Vi polysaccharide in a colorless isotonic phosphate buffered saline (pH 7 ± 0.3), 4.150 mg of Sodium Chloride, 0.065 mg of Disodium Phosphate, 0.023 mg of Monosodium Phosphate, and 0.5 mL of Sterile Water for Injection. The vial stopper and the plunger stopper of the syringe are not made with natural rubber latex.
-
CLINICAL PHARMACOLOGY
Typhoid fever is an infectious disease caused by S typhi. Humans are the only natural host and reservoir for S typhi; infections result from the consumption of food or water that has been contaminated by the excretions of an acute case or a carrier. S typhi organisms efficiently invade the human intestinal mucosae ultimately leading to bacteremia; following a typical 10- to 14-day incubation period, a systemic illness occurs. The clinical presentation of typhoid fever exhibits a broad range of severity and can be debilitating. Classical cases have fever, myalgia, anorexia, abdominal discomfort and headaches; the fever increases step-wise over a period of days and then may remain at 102°F to 106°F over 10 to 14 days before decreasing in a step-wise manner. Skin lesions known as rose spots may be present. Constipation is common in older children and adults, while diarrhea may occur in younger children. Among the less common but most severe complications are intestinal perforation and hemorrhage, and death. The course is typically more severe without appropriate antimicrobial therapy. The case fatality rate was reported to be approximately 10% to 20% in the pre-antibiotic era. (1) (2) (3) During the period of 1983 to 1991 in the US, the case fatality rate reported to the Centers for Disease Control and Prevention (CDC) was 0.2% (9/4010). (4) Infection of the gallbladder can lead to the chronic carrier state.
Typhoid fever is still endemic in many countries of the world where it is predominantly a disease of school-age children and may be a major public health problem. Most cases of typhoid fever in the US are thought to be acquired during foreign travel. During the periods of 1975 to 1984 and 1983 to 1984, respectively, 62% and 70% of the cases of typhoid fever reported to the CDC were acquired during foreign travel; this compares to 33% of cases during 1967-1972. (5)
In 1992, 414 cases of typhoid fever were reported to the CDC. Of these 414 cases, 1 (0.2%) case occurred in an infant under one year of age; 77 (18.6%) cases occurred in persons one to nine years of age; 81 (19.6%) cases occurred in persons 10 to 19 years of age; 251 (60.6%) cases occurred in individuals ≥20 years of age; the age was not available for 4 (1%) cases. One death was reported in 1991. (4) Domestic surveillance could underestimate the risk of typhoid fever in travelers since the disease is unlikely to be reported for persons who received diagnosis and treatment overseas. (6)
Approximately 2% to 4% of acute typhoid fever cases develop into a chronic carrier state. The chronic carrier state occurs more frequently with advanced age, and among females than males. (2) (7) These non-symptomatic carriers are the natural reservoir for S typhi and can serve to maintain the disease in its endemic state or to directly infect new individuals. Outbreaks of typhoid fever are often traced to food handlers who are asymptomatic carriers. (8)
Two formulations were utilized in studies of the Typhoid Vi Polysaccharide Vaccine. These included the liquid formulation which is identical to Typhim Vi vaccine and a lyophilized formulation.
The protective efficacy of each of these formulations of the Typhoid Vi Polysaccharide Vaccine was assessed independently in two trials conducted in areas where typhoid fever is endemic. A single intramuscular dose of 25 mcg was used in these efficacy studies. A randomized double-blind controlled trial with Typhim Vi vaccine (liquid formulation) was conducted in five villages west of Katmandu, Nepal. There were 6,908 vaccinated subjects: 3,454 received Typhim Vi vaccine and 3,454 in the control group received a 23-valent pneumococcal polysaccharide vaccine. Of the 6,908 subjects, 6,439 subjects were in the target population of 5 to 44 years of age. In addition, 165 children ages 2 to 4 years and 304 adults over 44 years of age were included in the study. The overall protective efficacy of Typhim Vi vaccine was 74% (95% confidence interval (CI): 49% to 87%) for blood culture confirmed cases of typhoid fever during 20 months of post-vaccination follow-up. (9) (10) (11)
The protective efficacy of the Typhoid Vi Polysaccharide Vaccine, lyophilized formulation, was evaluated in a randomized double-blind controlled trial conducted in South Africa. There were 11,384 vaccinated children 5 to 15 years of age; 5,692 children received the Vi capsular polysaccharide vaccine and 5,692 in the control group received Meningococcal Polysaccharide (Groups A+C) Vaccine. The protective efficacy for the Vi capsular polysaccharide (lyophilized formulation) group for blood culture confirmed cases of typhoid fever was 55% (95% CI: 30% to 70%) overall during 3 years of post-vaccination follow-up, and was 61%, 52% and 50%, respectively, for years 1, 2, and 3. Vaccination was associated with an increase in anti-Vi antibodies as measured by radioimmunoassay (RIA) and enzyme-linked immunosorbent assay. Antibody levels remained elevated at 6 and 12 months post-vaccination. (11) (12)
Because of the low incidence of typhoid fever, efficacy studies were not feasible in a US population.
Controlled comparative efficacy studies of Typhim Vi vaccine and other types of typhoid vaccines have not been performed.
An increase in serum anti-capsular antibodies is thought to be the basis of protection provided by Typhim Vi vaccine. However, a specific correlation of post-vaccination antibody levels with subsequent protection is not available, and the level of Vi antibody that will provide protection has not been determined. Also, limitations exist for comparing immunogenicity results from subjects in endemic areas, where some subjects have baseline serological evidence of prior S typhi exposure, to naive populations such as most American travelers.
In endemic regions (Nepal, South Africa, Indonesia) where trials were conducted, pre-vaccination geometric mean antibody levels suggest that infection with S typhi had previously occurred in a large percentage of the vaccinees. In these populations, specific antibody levels increased four-fold or greater in 68% to 87.5% of older children and adult subjects following vaccination. For 43 persons 15 to 44 years of age in the Nepal pilot study, geometric mean specific antibody levels pre- and 3 weeks post-vaccination were, respectively, 0.38 and 3.68 mcg antibody/mL by RIA; 79% had a four-fold or greater rise in Vi antibody levels. (9) (12)
Immunogenicity and safety trials were conducted in an adult US population. A single dose of Typhim Vi vaccine induced a four-fold or greater increase in antibody levels in 88% and 96% of this adult population for 2 studies, respectively, following vaccination (see Table 1). (10) (13)
Table 1 (10) (13): Vi ANTIBODY LEVELS IN US ADULTS 18 TO 40 YEARS OF AGE GIVEN TYPHIM Vi VACCINE GEOMETRIC MEAN ANTIBODY LEVELS (mcg antibody/mL by RIA) N Pre Post
(4 weeks)% ≥4 FOLD
INCREASE(95% CI) (95% CI) (95% CI) Trial 1
(1 lot)54 0.16
(0.13 to 0.21)3.23
(2.59 to 4.03)96% (52/54)
(87% to 100%)Trial 2
(2 lots combined)97 0.17
(0.14 to 0.21)2.86
(2.26 to 3.62)88% (85/97)
(81% to 94%)No studies of safety and immunogenicity have been conducted in US children. A double-blind randomized controlled trial evaluating the safety and immunogenicity of Typhim Vi vaccine was performed in 175 Indonesian children. The percentage of 2- to 5-year-old children achieving a four-fold or greater increase in antibody levels at 4 weeks post-vaccination was 96.3% (52/54) (95% CI: 87.3% to 99.6%), and in the study subset of 2-year-old children was 94.4% (17/18) (95% CI: 72.7% to 99.9%). The geometric mean antibody levels (mcg antibody/mL by RIA) for the 2- to 5-year-old children and the subset of 2-year-olds were, respectively, 5.81 (4.36 to 7.77) and 5.76 (3.48 to 9.53). (10) (11)
In the US Reimmunization Study, adults previously immunized with Typhim Vi vaccine in other studies were reimmunized with a 25 mcg dose at 27 or 34 months after the primary dose. Data on antibody response to primary immunization, decline following primary immunization, and response to reimmunization are presented in Table 2. Antibody levels attained following reimmunization at 27 or 34 months after the primary dose were similar to levels attained following the primary immunization. (10) (13) This response is typical for a T-cell independent polysaccharide vaccine in that reimmunization does not elicit higher antibody levels than primary immunization. The safety of reimmunization was also evaluated in this study (see ADVERSE REACTIONS section).
Table 2 (10) (13): US STUDIES IN 18- TO 40-YEAR-OLD ADULTS: KINETICS AND PERSISTENCE OF Vi ANTIBODY* RESPONSE TO PRIMARY IMMUNIZATION WITH TYPHIM Vi VACCINE, AND RESPONSE TO REIMMUNIZATION AT 27 OR 34 MONTHS PRE-DOSE 1 1
MONTH11 MONTHS 18 MONTHS 27 MONTHS 34 MONTHS 1 MONTH POST-REIMMUNIZATION† - *
- mcg antibody/mL by RIA
- †
- Includes available data from all reimmunized subjects (subjects initially randomized to Typhim Vi vaccine, and subjects initially randomized to placebo who received open label Typhim Vi vaccine two weeks later).
- ‡
- Group 1: Reimmunized at 27 months following primary immunization.
- §
- Not Done.
- ¶
- Antibody levels pre-reimmunization.
- #
- Group 2: Reimmunized at 34 months following primary immunization.
GROUP 1‡ N
Level*
95% CI43
0.19
(0.14-0.26)43
3.01
(2.22-4.06)39
1.97
(1.31-3.00)ND§ 43
1.07¶
(0.71-1.62)ND 43
3.04
(2.17-4.26)GROUP 2# N
Level
95% CI12
0.14
(0.11-0.18)12
3.78
(2.18-6.56)ND 10
1.21
(0.63-2.35)ND 12
0.76¶
(0.37-1.55)12
3.31
(1.61-6.77)Concurrently Administered Vaccines
Concomitant Administration of Typhim Vi and Menactra®, Meningococcal (Groups A, C, Y and W-135) Polysaccharide Diphtheria Toxoid Conjugate Vaccine, Manufactured by Sanofi Pasteur SA
In a double-blind, randomized, controlled clinical trial, 945 participants aged 18 through 55 years received Typhim Vi and Menactra vaccines concomitantly (N=469), or Typhim Vi vaccine followed one month later by Menactra vaccine (N=476). Sera were obtained approximately 28 days after each respective vaccination. The antibody response to Typhim Vi vaccine and to Menactra vaccine components were similar between groups.
-
INDICATIONS AND USAGE
Typhim Vi vaccine is indicated for active immunization for the prevention of typhoid fever caused by S typhi and is approved for use in persons two years of age or older.
Immunization with Typhim Vi vaccine should occur at least two weeks prior to expected exposure to S typhi.
Typhim Vi vaccine is not indicated for routine immunization of individuals in the United States (US). (14)
Selective immunization against typhoid fever is recommended under the following circumstances: 1) travelers to areas where a recognized risk of exposure to typhoid exists, particularly ones who will have prolonged exposure to potentially contaminated food and water, 2) persons with intimate exposure (ie, continued household contact) to a documented typhoid carrier, and 3) workers in microbiology laboratories who frequently work with S typhi. (14)
Typhoid vaccination is not required for international travel, but is recommended for travelers to such areas as Africa, Asia, and Central and South America where there is a recognized risk of exposure to S typhi. Current CDC advisories should be consulted with regard to specific locales. Vaccination is particularly recommended for travelers who will have prolonged exposure to potentially contaminated food and water. However, even travelers who have been vaccinated should use caution in selecting food and water. (15)
There is no evidence to support the use of typhoid vaccine to control common source outbreaks, disease following natural disaster or in persons attending rural summer camps. (14)
An optimal reimmunization schedule has not been established. Reimmunization every two years under conditions of repeated or continued exposure to the S typhi organism is recommended at this time.
For recommended primary immunization and reimmunization see DOSAGE AND ADMINISTRATION section.
Typhim Vi vaccine should not be used to treat a patient with typhoid fever or a chronic typhoid carrier.
- CONTRAINDICATIONS
-
WARNINGS
Allergic reactions have been reported rarely in the post-marketing experience (see ADVERSE REACTIONS section).
The safety and immunogenicity of Typhim Vi vaccine in children under two years of age has not been established. As with other polysaccharide vaccines, the antibody response may be inadequate. The decision whether to vaccinate children under 2 years of age depends upon the risk incurred by the child on the basis of the epidemiological context.
Typhim Vi vaccine provides protection against the risk of infection related to Salmonella typhi, but gives no protection against Salmonella paratyphi A or B, non-S typhi species of Salmonella enterica serovar Typhi, or other bacteria that cause enteric disease.
If the vaccine is used in persons deficient in producing antibodies, whether due to genetic defect, immunodeficiency disease, or immunosuppressive therapy, the expected immune response may not be obtained. This includes patients with asymptomatic or symptomatic HIV-infection, severe combined immunodeficiency, hypogammaglobulinemia, or agammaglobulinemia; altered immune states due to diseases such as leukemia, lymphoma, or generalized malignancy; or an immune system compromised by treatment with corticosteroids, alkylating drugs, antimetabolites or radiation. (16)
As with any vaccine, vaccination with Typhim Vi vaccine may not protect 100% of individuals.
-
PRECAUTIONS
General
Care is to be taken by the health-care provider for the safe and effective use of Typhim Vi vaccine.
Epinephrine injection (1:1000) must be immediately available following immunization should an anaphylactic or other allergic reaction occur due to any component of the vaccine.
Prior to an injection of any vaccine, all known precautions should be taken to prevent adverse reactions. This includes a review of the patient's history with respect to possible hypersensitivity to the vaccine or similar vaccines.
Acute infection or febrile illness may be reason for delaying use of Typhim Vi vaccine except when, in the opinion of the physician, withholding the vaccine entails a greater risk.
Syncope (fainting) has been reported following vaccination with Typhim Vi. Procedures should be in place to prevent falling injury and manage syncopal reactions.
Safety and immunogenicity data from controlled trials are not available for Typhim Vi vaccine following previous immunization with whole-cell typhoid or live, oral typhoid vaccine (see ADVERSE REACTIONS section).
Information for Vaccine Recipients or Parents/Guardians
Before administration, healthcare providers should inform patients, parents or guardians of the benefits and risks of immunization with Typhim Vi vaccine.
Prior to administration of Typhim Vi vaccine, healthcare providers should ask patients, parents and guardians about the recent health status of the patient to be immunized.
Typhim Vi vaccine is indicated in persons traveling to endemic or epidemic areas. Current CDC advisories should be consulted with regard to specific locales.
Travelers should take all necessary precautions to avoid contact with or ingestion of contaminated food and water.
One dose of vaccine should be given at least 2 weeks prior to expected exposure.
Reimmunization consisting of a single-dose for US travelers every two years under conditions of repeated or continued exposure to the S typhi organism is recommended at this time. (14)
As part of the child's or adult's immunization record, the date, lot number, and manufacturer of the vaccine administered should be recorded. (17)
Drug Interactions
Concomitant Vaccine Administration
Typhim Vi was concomitantly administered with Menactra vaccine in individuals 18 through 55 years of age (see CLINICAL PHARMACOLOGY and ADVERSE REACTIONS).
No studies have been conducted in the US to evaluate interactions or immunological interference between the concurrent use of Typhim Vi vaccine and drugs (including antibiotics and antimalarial drugs), immune globulins or other vaccines (including common travelers vaccines such as tetanus, poliomyelitis, hepatitis A, and yellow fever).
Typhim Vi vaccine must not be mixed with any vaccine in the same syringe. Separate injection sites should be used in case of concomitant administration.
CARCINOGENESIS, MUTAGENESIS, IMPAIRMENT OF FERTILITY
Typhim Vi vaccine has not been evaluated for its carcinogenic potential, mutagenic potential or impairment of fertility.
PREGNANCY
Animal reproduction studies have not been conducted with Typhim Vi vaccine. It is not known whether Typhim Vi vaccine can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Typhim Vi vaccine should be given to a pregnant woman only if clearly needed. (14)
When possible, delaying vaccination until the second or third trimester to minimize the possibility of teratogenicity is a reasonable precaution. (18)
NURSING MOTHERS
It is not known whether Typhim Vi vaccine is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Typhim Vi vaccine is administered to a nursing woman.
There is no data on the use of this product in nursing mothers.
PEDIATRIC USE
Safety and effectiveness of Typhim Vi vaccine have been established in children 2 years of age and older. (10) (11) (See DOSAGE AND ADMINISTRATION section.)
For children below the age of 2 years, safety and effectiveness have not been established.
-
ADVERSE REACTIONS
Adverse event information is derived from clinical trials and worldwide post-marketing experience.
Data From Clinical Trials
Because clinical trials are conducted under widely varying conditions, adverse reactions rates observed in the clinical trials of a vaccine cannot be directly compared to rates in the clinical trials of another vaccine and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to vaccine use and for approximating rates.
Safety of Typhim Vi vaccine, the US licensed liquid formulation, has been assessed in clinical trials in more than 4,000 subjects both in countries of high and low endemicity. In addition, the safety of the lyophilized formulation has been assessed in more than 6,000 individuals. The adverse reactions were predominately minor and transient local reactions. Local reactions such as injection site pain, erythema, and induration almost always resolved within 48 hours of vaccination. Elevated oral temperature, above 38°C (100.4°F), was observed in approximately 1% of vaccinees in all studies. No serious or life-threatening systemic events were reported in these clinical trials. (10) (11)
Adverse reactions from two trials evaluating Typhim Vi vaccine lots in the US (18- to 40-year-old adults) are summarized in Table 3. No severe or unusual side effects were observed. Most subjects reported pain and/or tenderness (pain upon direct pressure). Local adverse experiences were generally limited to the first 48 hours. (10) (11)
Table 3 (10) (11): PERCENTAGE OF 18- TO 40-YEAR-OLD US ADULTS PRESENTING WITH LOCAL OR SYSTEMIC REACTIONS WITHIN 48 HOURS AFTER THE FIRST IMMUNIZATION WITH TYPHIM Vi VACCINE REACTION Trial 1
Placebo
N = 54Trial 1
Typhim Vi vaccine
N = 54
(1 Lot)Trial 2
Typhim Vi vaccine
N = 98
(2 Lots combined)Local Tenderness 7 (13.0%) 53 (98.0%) 95 (96.9%) Pain 4 (7.4%) 22 (40.7%) 26 (26.5%) Induration 0 8 (14.8%) 5 (5.1%) Erythema 0 2 (3.7%) 5 (5.1%) Systemic Malaise 8 (14.8%) 13 (24.0%) 4 (4.1%) Headache 7 (13.0%) 11 (20.4%) 16 (16.3%) Myalgia 0 4 (7.4%) 3 (3.1%) Nausea 2 (3.7%) 1 (1.9%) 8 (8.2%) Diarrhea 2 (3.7%) 0 3 (3.1%) Feverish (subjective) 0 6 (11.1%) 3 (3.1%) Fever ≥100°F 0 1 (1.9%) 0 Vomiting 0 1 (1.9%) 0 No studies were conducted in US children. Adverse reactions from a trial in Indonesia in children one to twelve years of age are summarized in Table 4. (10) (11) No severe or unusual side effects were observed.
Table 4 (10) (11): PERCENTAGE OF INDONESIAN CHILDREN ONE TO TWELVE YEARS OF AGE PRESENTING WITH LOCAL OR SYSTEMIC REACTIONS WITHIN 48 HOURS AFTER THE FIRST IMMUNIZATION WITH TYPHIM Vi VACCINE REACTIONS N = 175 - *
- Subjective feeling of fever.
Local Soreness 23 (13.0%) Pain 25 (14.3%) Erythema 12 (6.9%) Induration 5 (2.9%) Impaired Limb Use 0 Systemic Feverishness* 5 (2.9%) Headache 0 Decreased Activity 3 (1.7%) In the US Reimmunization Study, subjects who had received Typhim Vi vaccine 27 or 34 months earlier, and subjects who had never previously received a typhoid vaccination, were randomized to placebo or Typhim Vi vaccine, in a double-blind study. Safety data from the US Reimmunization Study are presented in Table 5. (10) (11) (13) In this study 5/30 (17%) primary immunization subjects and 10/45 (22%) reimmunization subjects had a local reaction. No severe or unusual side effects were observed. Most subjects reported pain and/or tenderness (pain upon direct pressure). Local adverse experiences were generally limited to the first 48 hours. (10) (11) (13)
Table 5 (10) (11) (13): US REIMMUNIZATION STUDY, SUBJECTS PRESENTING WITH LOCAL AND SYSTEMIC REACTIONS WITHIN 48 HOURS AFTER IMMUNIZATION WITH TYPHIM Vi VACCINE REACTION PLACEBO
(N = 32)FIRST IMMUNIZATION
(N = 30)REIMMUNIZATION
(N = 45*)- *
- At 27 or 34 months following a previous dose given in different studies.
Local Tenderness 2 (6%) 28 (93%) 44 (98%) Pain 1 (3%) 13 (43%) 25 (56%) Induration 0 5 (17%) 8 (18%) Erythema 0 1 (3%) 5 (11%) Systemic Malaise 1 (3%) 11 (37%) 11 (24%) Headache 5 (16%) 8 (27%) 5 (11%) Myalgia 0 2 (7%) 1 (2%) Nausea 0 1 (3%) 1 (2%) Diarrhea 0 0 1 (2%) Feverish (subjective) 0 3 (10%) 2 (4%) Fever ≥100°F 1 (3%) 0 1 (2%) Vomiting 0 0 0 Solicited Injection Site and Systemic Reactions When Given With Menactra Vaccine
The majority (70%-77%) of solicited injection site reactions at the Typhim Vi and at the Menactra injection sites were reported as Grade 1 and resolved within 3 days post-vaccination. The most common systemic reactions were headache (41% when Menactra and Typhim Vi were given concomitantly; 42% when Typhim Vi was given with Placebo, and 33% when Menactra vaccine was given alone one month after Typhim Vi vaccination) and fatigue (38% when Menactra vaccine and Typhim Vi were given concomitantly; 35% when Typhim Vi was given with Placebo, and 27% when Menactra vaccine was given alone one month after Typhim Vi vaccination). Fever ≥40.0°C and seizures were not reported.
Data From Worldwide Post-marketing Experience
In addition to reports in clinical trials, worldwide voluntary adverse events reports received since market introduction of Typhim Vi vaccine are listed below. This list includes serious events and/or events which were included based on severity, frequency of reporting or a plausible causal connection to Typhim Vi vaccine. Because these events were reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to vaccination.
- Gastrointestinal disorders
Nausea, vomiting, diarrhea, abdominal pain - General disorders and administration site condition
Injection site pain, inflammation, induration, and erythema; lymphadenopathy, fever, asthenia, malaise, flu-like episode - Immune system disorders
Anaphylaxis, allergic-type reactions such as pruritus, rash, urticaria, angioedema, difficulty breathing, hypotension; serum sickness - Musculoskeletal and connective tissue disorders
Myalgia, arthralgia, cervical pain - Nervous system disorders
Syncope with and without convulsions, headache, loss of consciousness, tremor - Respiratory system disorders
Asthma
Reporting of Adverse Events
Reporting by parents and patients of all adverse events occurring after vaccine administration should be encouraged. Adverse events following immunization with vaccine should be reported by the health-care provider to the US Department of Health and Human Services (DHHS) Vaccine Adverse Event Reporting System (VAERS). Reporting forms and information about reporting requirements or completion of the form can be obtained from VAERS through a toll-free number 1-800-822-7967 or visit the VAERS website at http//www.vaers.org. (17)
Health-care providers also should report these events to the Pharmocovigilance Department, Sanofi Pasteur Inc., Discovery Drive, Swiftwater, PA 18370, or call 1-800-822-2463.
-
DOSAGE AND ADMINISTRATION
For intramuscular use only.
Dosage
The immunizing dose for adults and children is a single injection of 0.5 mL.
A reimmunizing dose is 0.5 mL. Reimmunization consisting of a single dose for US travelers every two years under conditions of repeated or continued exposure to the S typhi organism is recommended at this time. (14)
Preparation for Administration
The syringe or vial and its packaging should be inspected prior to use for evidence of leakage, premature activation of the plunger, or a faulty tip seal. If any of these conditions exists, do NOT administer the vaccine.
- Syringe
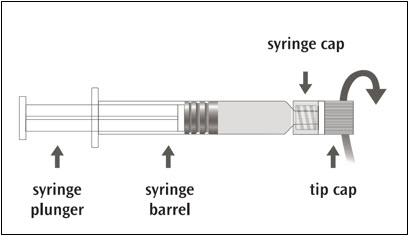
Picture A: Luer-Lok™ syringe
Step 1: Holding the syringe cap in one hand (avoid holding the syringe plunger or barrel), unscrew the tip cap by twisting it counterclockwise. 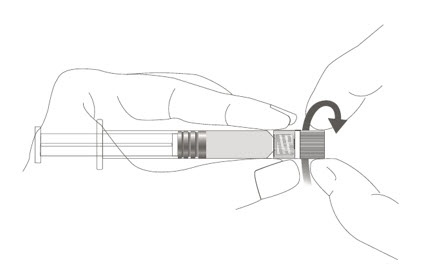
Step 2: To attach the needle to the syringe, gently twist the needle clockwise into the syringe until slight resistance is felt. 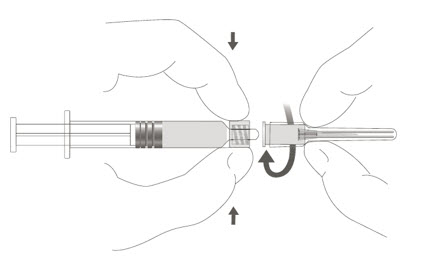
The syringe is intended for single use only, must not be reused, and must be disposed of properly and promptly following its use.
- Vial
Tear off upper seal of vial cap. Cleanse top of rubber stopper of the vial with a suitable antiseptic.
Use a separate sterile syringe and needle or a sterile disposable unit for each individual patient to prevent the transmission of infectious agents from person to person. Needles should not be recapped and should be properly disposed.
Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. If any of this conditions exists, do NOT administer the vaccine.
In adults, the intramuscular injection is typically given in the deltoid. In children, the intramuscular injection is given either in the deltoid or the anterolateral thigh.
Do NOT inject this vaccine into the gluteal area or areas where there may be a nerve trunk.
Do NOT inject intravenously.
- HOW SUPPLIED
-
REFERENCES
- 1
- Levine MM, et al. New knowledge on pathogenesis of bacterial enteric infections as applied to vaccine development. Microbiol. Rev. 47: 510-550, 1983
- 2
- Levine MM. Typhoid Fever Vaccines. p 333-361. In Vaccines, Plotkin SA, Mortimer EA, eds. W.B. Saunders, 1988
- 3
- Levine MM, et al. Typhoid Fever Chapter 5, In: Vaccines and Immunotherapy. Stanley J. Cryz, Jr., Editor. pp 59-72, 1991
- 4
- CDC. Summary of Notifiable Diseases, United States 1992. MMWR 41: No. 55, 1993
- 5
- Ryan CA, et al. Salmonella typhi infections in the United States, 1975-1984: Increasing Role of Foreign Travel. Rev Infect Dis 11:1-8, 1989
- 6
- Woodruff BA, et al. A new look at typhoid vaccination. Information for the practicing physician. JAMA 265: 756-759, 1991
- 7
- Ames WR, et al. Age and sex as factors in the development of the typhoid carrier state, and a method for estimating carrier prevalence. Am J Public Health 33: 221-230, 1943
- 8
- CDC. Typhoid fever - Skagit County, Washington. MMWR 39: 749-751, 1990
- 9
- Acharya IL, et al. Prevention of typhoid fever in Nepal with the Vi capsular polysaccharide of Salmonella typhi. N Engl J Med 317: 1101-1104, 1987
- 10
- Unpublished data available from Sanofi Pasteur Inc., compiled 1991
- 11
- Unpublished data available from Sanofi Pasteur SA
- 12
- Klugman KP, et al. Protective activity of Vi capsular polysaccharide vaccine against typhoid fever. The Lancet, 1165-1169, 1987
- 13
- Keitel WA, et al. Clinical and serological responses following primary and booster immunization with Salmonella typhi Vi capsular polysaccharide vaccines. Vaccines 12: 195-199, 1994
- 14
- Recommendations of the Immunization Practices Advisory Committee (ACIP). Typhoid Immunization. MMWR 43: No. RR-14, 1994
- 15
- CDC. Health Information for International Travel 2001-2002. Atlanta: US Department of Health and Human Services, Public Health Service, 2001
- 16
- ACIP: Use of vaccines and immune globulins in persons with altered immunocompetence. MMWR 42: No. RR-4, 1993
- 17
- CDC. Vaccine Adverse Event Reporting System - United States. MMWR 39: 730-733, 1990
- 18
- ACIP: Update on Adult Immunization. MMWR 40: No. RR-12, 1991
- SPL UNCLASSIFIED SECTION
- SALMONELLA TYPHI TY2 VI POLYSACCHARIDE ANTIGEN
-
INGREDIENTS AND APPEARANCE
TYPHIM VI
salmonella typhi ty2 vi polysaccharide antigen injection, solutionProduct Information Product Type VACCINE Item Code (Source) NDC:50090-1758(NDC:49281-790) Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALMONELLA TYPHI TY2 VI POLYSACCHARIDE ANTIGEN (UNII: 7194H8W3KT) (SALMONELLA TYPHI TY2 VI POLYSACCHARIDE ANTIGEN - UNII:7194H8W3KT) SALMONELLA TYPHI TY2 VI POLYSACCHARIDE ANTIGEN 25 ug in 0.5 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 4.15 mg in 0.5 mL SODIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: 94255I6E2T) 0.065 mg in 0.5 mL SODIUM PHOSPHATE, MONOBASIC, DIHYDRATE (UNII: 5QWK665956) 0.023 mg in 0.5 mL PHENOL (UNII: 339NCG44TV) 1.25 mg in 0.5 mL WATER (UNII: 059QF0KO0R) 0.5 mL in 0.5 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50090-1758-0 1 in 1 PACKAGE 1 NDC:50090-1758-9 10 mL in 1 VIAL, MULTI-DOSE; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA103936 11/28/1994 Labeler - A-S Medication Solutions (830016429) Establishment Name Address ID/FEI Business Operations A-S Medication Solutions 830016429 RELABEL(50090-1758)


