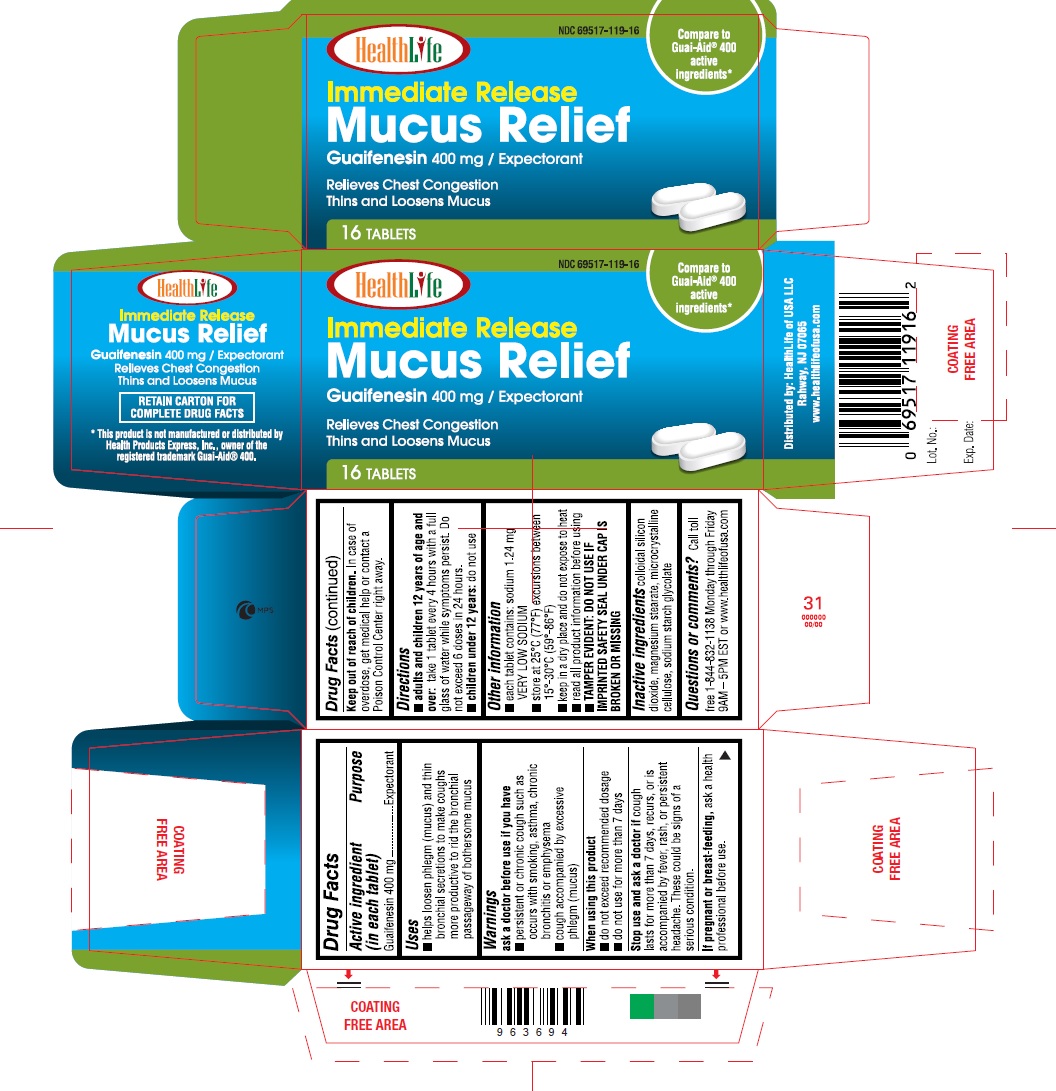
Label: MUCUS RELIEF- guaifenesin 400 mg tablet
-
NDC Code(s):
69517-119-02,
69517-119-05,
69517-119-10,
69517-119-12, view more69517-119-16, 69517-119-25, 69517-119-50
- Packager: HealthLife of USA LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 21, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
ask a doctor before use if you have
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis or emphysema
- cough accompanied by excessive phlegm (mucus)
- Directions:
- Other information
- Inactive ingredients
-
Questions or Comments
Call toll free 1-844-832-1138 Monday through Friday 9AM – 5PM EST or www.healthlifeofusa.com
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
MUCUS RELIEF
guaifenesin 400 mg tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69517-119 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 400 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STEARIC ACID (UNII: 4ELV7Z65AP) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color white Score no score Shape OVAL Size 17mm Flavor Imprint Code EB Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69517-119-25 25 in 1 CARTON 04/13/2016 1 2 in 1 POUCH; Type 0: Not a Combination Product 2 NDC:69517-119-50 50 in 1 CARTON 04/13/2016 2 2 in 1 POUCH; Type 0: Not a Combination Product 3 NDC:69517-119-02 2 in 1 POUCH 04/13/2016 3 2 in 1 POUCH; Type 0: Not a Combination Product 4 NDC:69517-119-10 1000 in 1 BOTTLE 04/13/2016 4 NDC:69517-119-05 500 in 1 BOTTLE 4 NDC:69517-119-12 12 in 1 BOTTLE 4 1 in 1 CARTON; Type 0: Not a Combination Product 5 NDC:69517-119-16 1 in 1 CARTON 06/05/2017 5 16 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 04/13/2016 Labeler - HealthLife of USA LLC (079656178) Establishment Name Address ID/FEI Business Operations Elysium Pharmaceutical Ltd. 915664486 manufacture(69517-119)