Label: BRAVECTO- fluralaner tablet, chewable
-
NDC Code(s):
0061-4340-01,
0061-4341-01,
0061-4342-01,
0061-4343-01, view more0061-4344-01
- Packager: Merck Sharp & Dohme Corp.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated October 29, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Caution:
- Description:
-
Indications:
Bravecto kills adult fleas and is indicated for the treatment and prevention of flea infestations (Ctenocephalides felis), and the treatment and control of tick infestations [Ixodes scapularis (black-legged tick), Dermacentor variabilis (American dog tick), Rhipicephalus sanguineus (brown dog tick), and Haemaphysalis longicornis (Asian longhorned tick)] for 12 weeks in dogs and puppies 6 months of age and older, and weighing 4.4 pounds or greater.
Bravecto is also indicated for the treatment and control of Amblyomma americanum (lone star tick) infestations for 8 weeks in dogs and puppies 6 months of age and older, and weighing 4.4 pounds or greater.
-
Dosage and Administration:
Bravecto should be administered orally as a single dose every 12 weeks according to the Dosage Schedule below to provide a minimum dose of 11.4 mg/lb (25 mg/kg) body weight.
Bravecto may be administered every 8 weeks in case of potential exposure to Amblyomma americanum ticks (see Effectiveness).
Bravecto should be administered with food.
Dosage Schedule Body Weight Ranges
(lb)Fluralaner Content
(mg)Chews Administered - *
- Dogs over 123.0 lb should be administered the appropriate combination of chews
4.4 – 9.9 112.5 One >9.9 – 22.0 250 One >22.0 – 44.0 500 One >44.0 – 88.0 1000 One >88.0 – 123.0* 1400 One Treatment with Bravecto may begin at any time of the year and can continue year round without interruption.
- Contraindications:
-
Warnings:
Not for human use. Keep this and all drugs out of the reach of children. Keep the product in the original packaging until use, in order to prevent children from getting direct access to the product. Do not eat, drink or smoke while handling the product. Wash hands thoroughly with soap and water immediately after use of the product.
Keep Bravecto in a secure location out of reach of dogs, cats, and other animals to prevent accidental ingestion or overdose.
-
Precautions:
Fluralaner is a member of the isoxazoline class. This class has been associated with neurologic adverse reactions including tremors, ataxia, and seizures. Seizures have been reported in dogs receiving isoxazoline class drugs, even in dogs without a history of seizures. Use with caution in dogs with a history of seizures or neurologic disorders.
Adverse events have been reported following use in breeding females. Before use in breeding female dogs, refer to Post-Approval Experience and Animal Safety sections.
Bravecto has not been shown to be effective for 12-weeks duration in puppies less than 6 months of age. Bravecto is not effective against Amblyomma americanum ticks beyond 8 weeks after dosing (see Effectiveness).
-
Adverse Reactions:
In a well-controlled U.S. field study, which included 294 dogs (224 dogs were administered Bravecto every 12 weeks and 70 dogs were administered an oral active control every 4 weeks and were provided with a tick collar); there were no serious adverse reactions. All potential adverse reactions were recorded in dogs treated with Bravecto over a 182-day period and in dogs treated with the active control over an 84-day period. The most frequently reported adverse reaction in dogs in the Bravecto and active control groups was vomiting.
Percentage of Dogs with Adverse Reactions in the Field Study Adverse Reaction
(AR)Bravecto Group: Percentage of Dogs with the AR During the 182-Day Study (n=224 dogs) Active Control Group: Percentage of Dogs with the AR During the 84-Day Study (n=70 dogs) Vomiting 7.1 14.3 Decreased Appetite 6.7 0.0 Diarrhea 4.9 2.9 Lethargy 5.4 7.1 Polydipsia 1.8 4.3 Flatulence 1.3 0.0 In a well-controlled laboratory dose confirmation study, one dog developed edema and hyperemia of the upper lips within one hour of receiving Bravecto. The edema improved progressively through the day and had resolved without medical intervention by the next morning.
Post-Approval Experience (2022):
The following adverse events are based on post-approval adverse drug experience reporting for fluralaner. Not all adverse events are reported to FDA/CVM. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using these data.
The following adverse events reported for dogs are listed in decreasing order of reporting frequency:
Vomiting, lethargy, diarrhea (with and without blood), anorexia, pruritis, polydipsia, seizure, allergic reactions (including hives, swelling, erythema), dermatitis (including crusts, pustules, rash), tremors and ataxia. In some cases, birth defects (including limb deformities and cleft palate), stillbirth, and abortion have been reported after treatment of breeding females.
Contact Information:
To report suspected adverse events, for technical assistance or to obtain a copy of the Safety Data Sheet (SDS), contact Merck Animal Health at 1-800-224-5318. Additional information can be found at www.bravecto.com.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or online at http://www.FDA.gov/reportanimalae.
-
Clinical Pharmacology:
Peak fluralaner concentrations are achieved between 2 hours and 3 days following oral administration, and the elimination half-life ranges between 9.3 to 16.2 days. Quantifiable drug concentrations can be measured (lower than necessary for effectiveness) through 112 days. Due to reduced drug bioavailability in the fasted state, fluralaner should be administered with food.
Mode of Action:
Fluralaner is for systemic use and belongs to the class of isoxazoline-substituted benzamide derivatives. Fluralaner is an inhibitor of the arthropod nervous system. The mode of action of fluralaner is the antagonism of the ligand-gated chloride channels (gamma-aminobutyric acid (GABA)-receptor and glutamate-receptor).
-
Effectiveness:
Bravecto began to kill fleas within two hours after administration in a well-controlled laboratory study. In a European laboratory study, Bravecto killed fleas and Ixodes ricinus ticks and reduced the numbers of live fleas and Ixodes ricinus ticks on dogs by > 98% within 12 hours for 12 weeks. In a well-controlled laboratory study, Bravecto demonstrated 100% effectiveness against adult fleas 48 hours post-infestation for 12 weeks. In well-controlled laboratory studies, Bravecto demonstrated ≥ 93% effectiveness against Dermacentor variabilis, Ixodes scapularis, Rhipicephalus sanguineus, and Haemaphysalis longicornis ticks 48 hours post-infestation for 12 weeks. Bravecto demonstrated ≥90% effectiveness against Amblyomma americanum 72 hours post-infestation for 8 weeks, but failed to demonstrate ≥90% effectiveness beyond 8 weeks.
In a well-controlled U.S. field study, a single dose of Bravecto reduced fleas by ≥ 99.7% for 12 weeks. Dogs with signs of flea allergy dermatitis showed improvement in erythema, alopecia, papules, scales, crusts, and excoriation as a direct result of eliminating flea infestations.
Palatability: In a well-controlled U.S. field study, which included 559 doses administered to 224 dogs, 80.7% of dogs voluntarily consumed Bravecto within 5 minutes, an additional 12.5% voluntarily consumed Bravecto within 5 minutes when offered with food, and 6.8% refused the dose or required forced administration.
-
Animal Safety:
Margin of Safety Study: In a margin of safety study, Bravecto was administered orally to 8- to 9-week-old puppies at 1, 3, and 5× the maximum label dose of 56 mg/kg at three, 8-week intervals. The dogs in the control group (0×) were untreated.
There were no clinically-relevant, treatment-related effects on physical examinations, body weights, food consumption, clinical pathology (hematology, clinical chemistries, coagulation tests, and urinalysis), gross pathology, histopathology, or organ weights. Diarrhea, mucoid and bloody feces were the most common observations in this study, occurring at a similar incidence in the treated and control groups. Five of the twelve treated dogs that experienced one or more of these signs did so within 6 hours of the first dosing. One dog in the 3× treatment group was observed to be dull, inappetant, with evidence of bloody diarrhea, vomiting, and weight loss beginning five days after the first treatment. One dog in the 1× treatment group vomited food 4 hours following the first treatment.
Reproductive Safety Study: Bravecto was administered orally to intact, reproductively- sound male and female Beagles at a dose of up to 168 mg/kg (equivalent to 3× the maximum label dose) on three to four occasions at 8-week intervals. The dogs in the control group (0×) were untreated.
There were no clinically-relevant, treatment-related effects on the body weights, food consumption, reproductive performance, semen analysis, litter data, gross necropsy (adult dogs) or histopathology findings (adult dogs and puppies). One adult 3× treated dog suffered a seizure during the course of the study (46 days after the third treatment). Abnormal salivation was observed on 17 occasions: in six treated dogs (11 occasions) after dosing and four control dogs (6 occasions).
The following abnormalities were noted in 7 pups from 2 of the 10 dams in only the treated group during gross necropsy examination: limb deformity (4 pups), enlarged heart (2 pups), enlarged spleen (3 pups), and cleft palate (2 pups). During veterinary examination at Week 7, two pups from the control group had inguinal testicles, and two and four pups from the treated group had inguinal and cryptorchid testicles, respectively. No undescended testicles were observed at the time of necropsy (days 50 to 71).
In a well-controlled field study Bravecto (fluralaner) was used concurrently with other medications, such as vaccines, anthelmintics, antibiotics, and steroids. No adverse reactions were observed from the concurrent use of Bravecto with other medications.
- Storage Information:
- How Supplied:
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 112.5 MG Chew Blister Pack Carton
BRAVECTO®
(fluralaner)
Chews4.4-9.9 lb
1 Chew
Kills fleas and ticks
For dogs and puppies 6 months of age and older
See back panel for dosing intervals
Caution: Federal (USA) law restricts this drug to
use by or on the order of a licensed veterinarian.
CONTENTS: 1 CHEW CONTAINING 112.5 MG
FLURALANER
Approved by FDA under NADA # 141-426
-
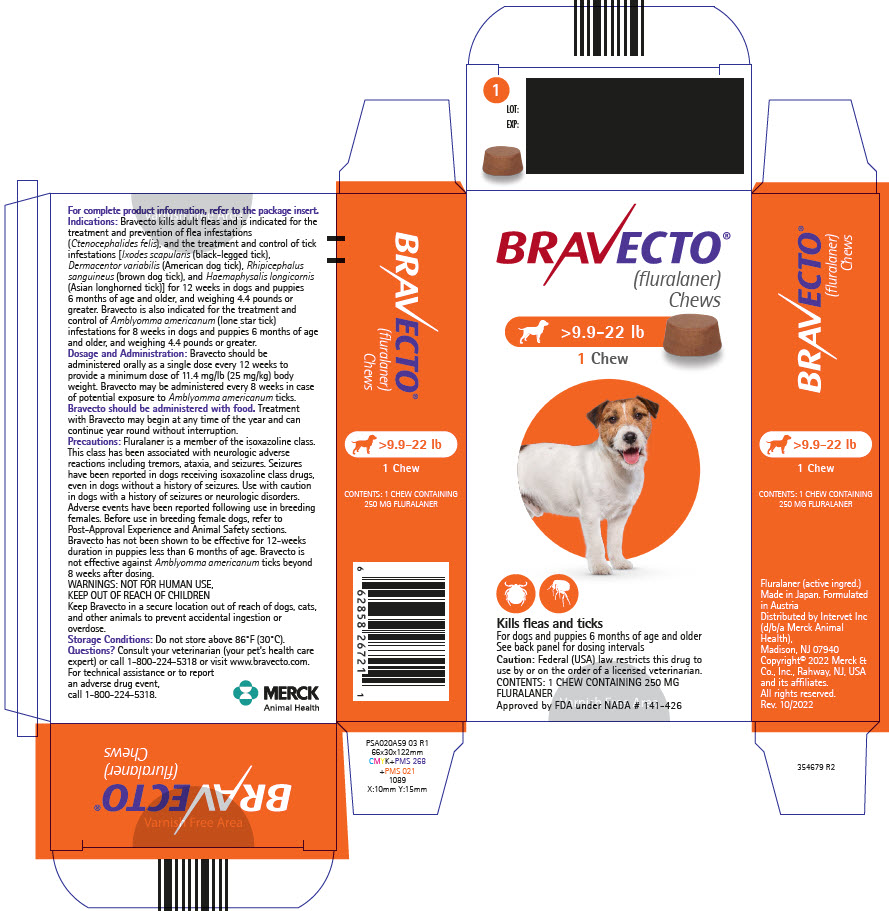
PRINCIPAL DISPLAY PANEL - 250 MG Chew Blister Pack Carton
BRAVECTO®
(fluralaner)
Chews>9.9-22 lb
1 Chew
Kills fleas and ticks
For dogs and puppies 6 months of age and older
See back panel for dosing intervals
Caution: Federal (USA) law restricts this drug to
use by or on the order of a licensed veterinarian.
CONTENTS: 1 CHEW CONTAINING 250 MG
FLURALANER
Approved by FDA under NADA # 141-426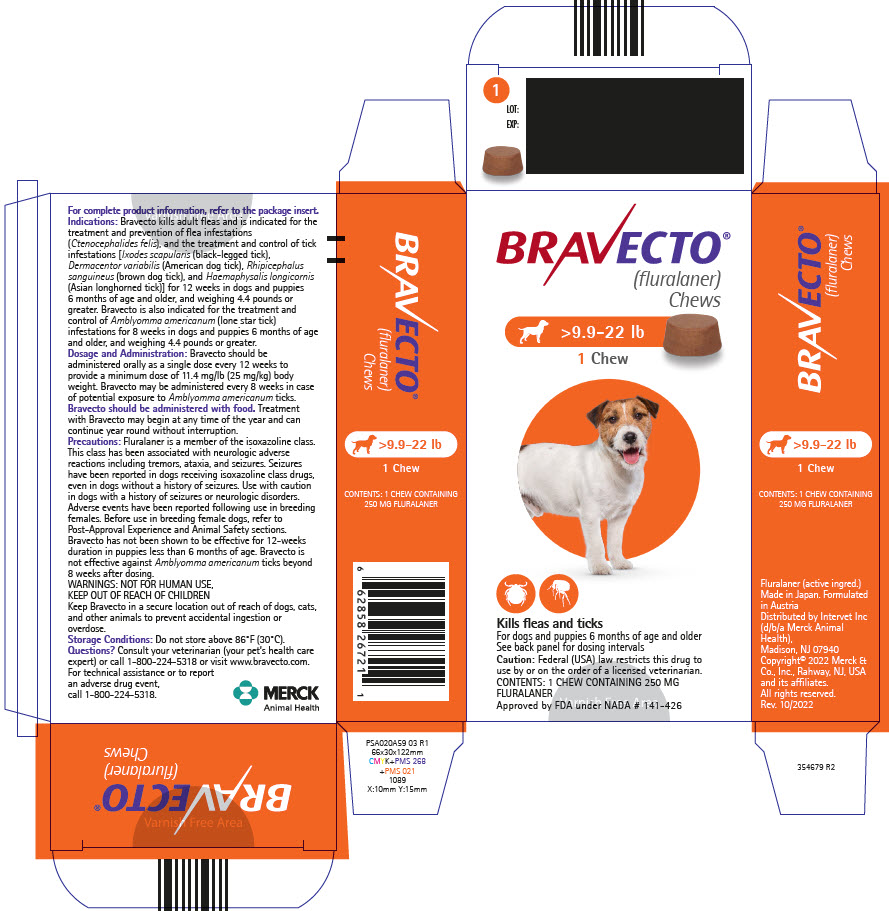
-
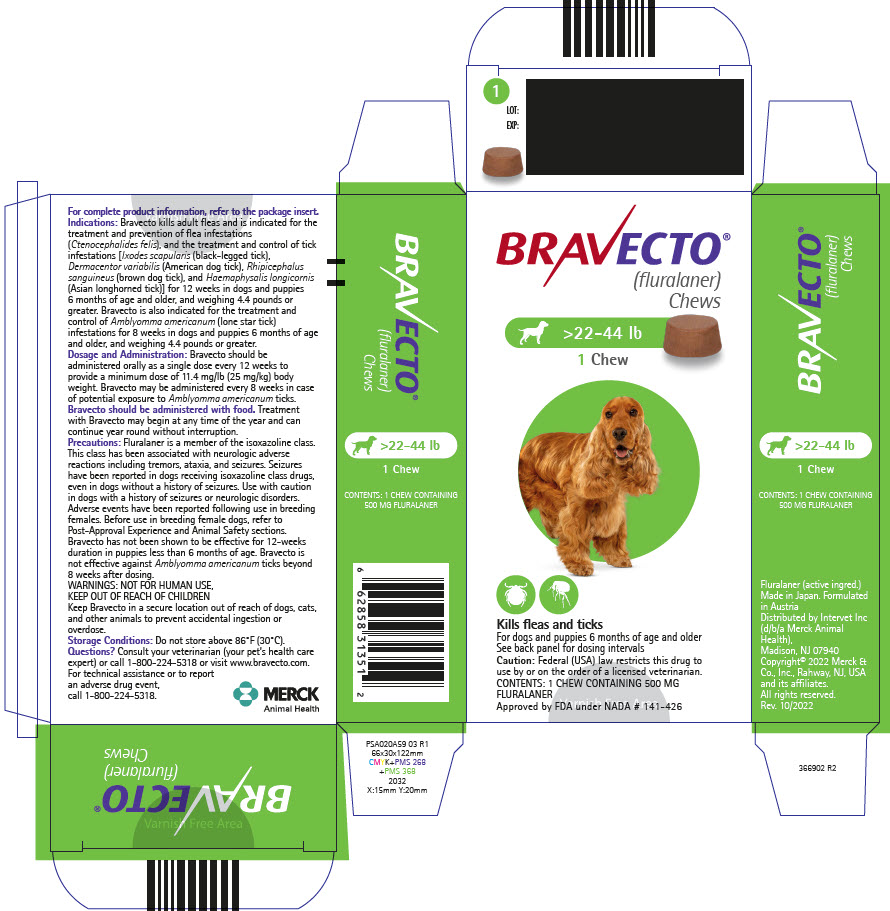
PRINCIPAL DISPLAY PANEL - 500 MG Chew Blister Pack Carton
BRAVECTO®
(fluralaner)
Chews>22-44 lb
1 Chew
Kills fleas and ticks
For dogs and puppies 6 months of age and older
See back panel for dosing intervals
Caution: Federal (USA) law restricts this drug to
use by or on the order of a licensed veterinarian.
CONTENTS: 1 CHEW CONTAINING 500 MG
FLURALANER
Approved by FDA under NADA # 141-426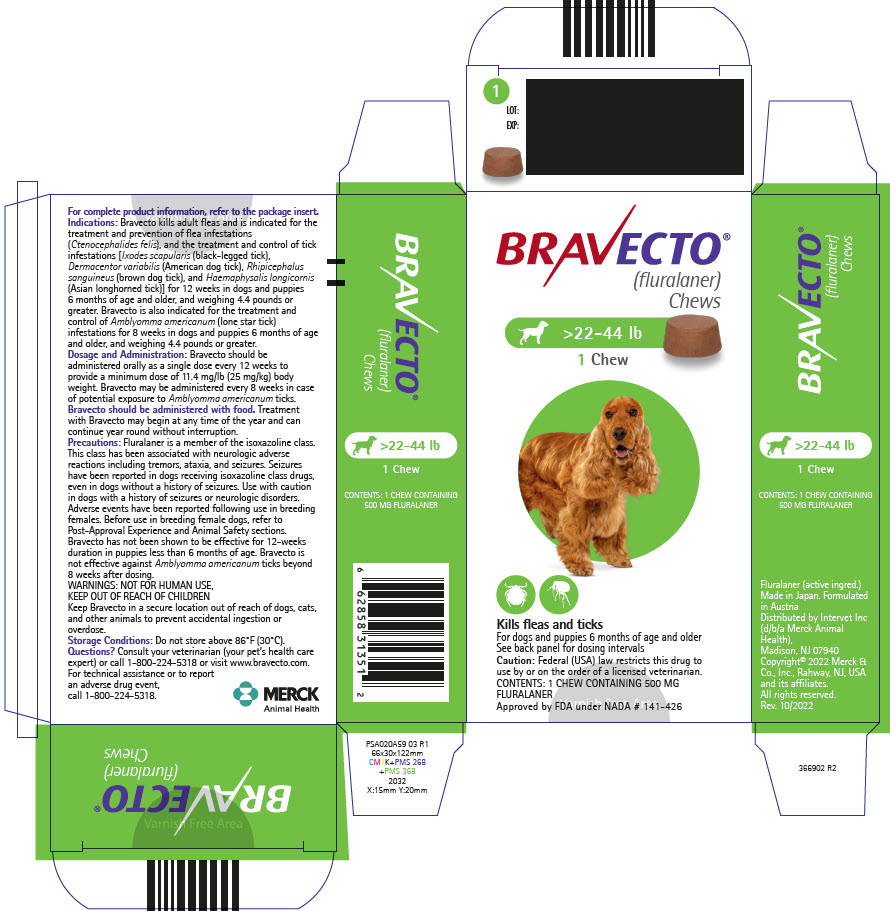
-
PRINCIPAL DISPLAY PANEL - 1000 MG Chew Blister Pack Carton
BRAVECTO®
(fluralaner)
Chews>44-88 lb
1 Chew
Kills fleas and ticks
For dogs and puppies 6 months of age and older
See back panel for dosing intervals
Caution: Federal (USA) law restricts this drug to
use by or on the order of a licensed veterinarian.
CONTENTS: 1 CHEW CONTAINING 1000 MG
FLURALANER
Approved by FDA under NADA # 141-426
-
PRINCIPAL DISPLAY PANEL - 1400 MG Chew Blister Pack Carton
BRAVECTO®
(fluralaner)
Chews>88-123 lb
1 Chew
Kills fleas and ticks
For dogs and puppies 6 months of age and older
See back panel for dosing intervals
Caution: Federal (USA) law restricts this drug to
use by or on the order of a licensed veterinarian.
CONTENTS: 1 CHEW CONTAINING 1400 MG
FLURALANER
Approved by FDA under NADA # 141-426
-
INGREDIENTS AND APPEARANCE
BRAVECTO
fluralaner tablet, chewableProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0061-4340 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FLURALANER (UNII: WSH8393RM5) (FLURALANER - UNII:WSH8393RM5) FLURALANER 112.5 mg Product Characteristics Color BROWN (Light brown to dark brown) Score no score Shape ROUND Size 11mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0061-4340-01 1 in 1 CARTON 1 1 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141426 05/16/2014 BRAVECTO
fluralaner tablet, chewableProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0061-4341 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FLURALANER (UNII: WSH8393RM5) (FLURALANER - UNII:WSH8393RM5) FLURALANER 250 mg Product Characteristics Color BROWN (Light brown to dark brown) Score no score Shape ROUND Size 16mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0061-4341-01 1 in 1 CARTON 1 1 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141426 05/16/2014 BRAVECTO
fluralaner tablet, chewableProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0061-4342 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FLURALANER (UNII: WSH8393RM5) (FLURALANER - UNII:WSH8393RM5) FLURALANER 500 mg Product Characteristics Color BROWN (Light brown to dark brown) Score no score Shape ROUND Size 20mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0061-4342-01 1 in 1 CARTON 1 1 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141426 05/16/2014 BRAVECTO
fluralaner tablet, chewableProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0061-4343 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FLURALANER (UNII: WSH8393RM5) (FLURALANER - UNII:WSH8393RM5) FLURALANER 1000 mg Product Characteristics Color BROWN (Light brown to dark brown) Score no score Shape ROUND Size 25mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0061-4343-01 1 in 1 CARTON 1 1 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141426 05/16/2014 BRAVECTO
fluralaner tablet, chewableProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0061-4344 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FLURALANER (UNII: WSH8393RM5) (FLURALANER - UNII:WSH8393RM5) FLURALANER 1400 mg Product Characteristics Color BROWN (Light brown to dark brown) Score no score Shape ROUND Size 29mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0061-4344-01 1 in 1 CARTON 1 1 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141426 05/16/2014 Labeler - Merck Sharp & Dohme Corp. (001317601) Establishment Name Address ID/FEI Business Operations HAMARI PFST, Ltd. 680242045 API MANUFACTURE Establishment Name Address ID/FEI Business Operations Gfm Gesellschaft Fuer Micronisierung Mbh 328866827 PARTICLE SIZE REDUCTION Establishment Name Address ID/FEI Business Operations LVA GmbH 300739641 ANALYSIS