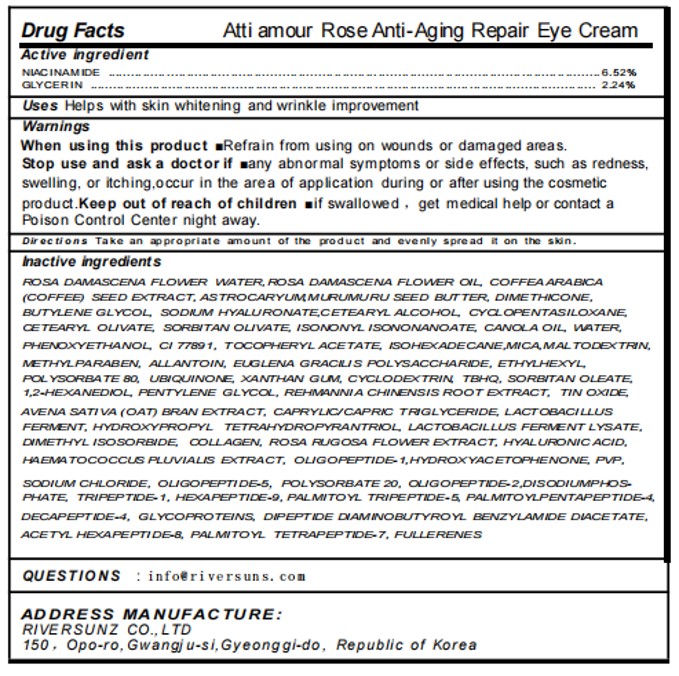
Label: ATTI AMOUR ROSE ANTI-AGING REPAIR EYE CREAM- niacinamide, glycerin cream
- NDC Code(s): 84601-003-01
- Packager: RIVER SUNZ co.,Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated August 5, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
When using this product mRefrain from using on wounds or damaged areas.Stop use and ask a doctorif many abnormal symptoms or side effects, such as redness.swvelling, or itching,occur in the area of applicatlon during or after using the cosmeticproduct.Keep out of reach of children if swallowed , get medical help or contact aPoison Control Center night away.
- DOSAGE & ADMINISTRATION
-
INACTIVE INGREDIENT
SODIUM HYALURONATE
ROSA DAMASCENA FLOWER WATER
ROSA DAMASCENA FLOWER OIL
COFFEA ARABICA (COFFEE) SEED EXTRACT
ASTROCARYUM MURUMURU SEED BUTTER
DIMETHICONE
BUTYLENE GLYCOL
CETEARYL OLIVATE
SORBITAN OLIVATE
CETEARYL ALCOHOL
CYCLOPENTASILOXANE
ISONONYL ISONONANOATE
MICA
CI 77891
TIN OXIDE
WATER
REHMANNIA CHINENSIS ROOT EXTRACT
CANOLA OIL
TBHQ
OLIGOPEPTIDE-1
OLIGOPEPTIDE-5
OLIGOPEPTIDE-2
TOCOPHERYL ACETATE
METHYLPARABEN
PENTYLENE GLYCOL
AVENA SATIVA (OAT) BRAN EXTRACT
ALLANTOIN
EUGLENA GRACILIS POLYSACCHARIDE
UBIQUINONE
LACTOBACILLUS FERMENT
LACTOBACILLUS FERMENT LYSATE
ROSA RUGOSA FLOWER EXTRACT
HYDROXYACETOPHENONE
COLLAGEN
HYALURONIC ACID
HAEMATOCOCCUS PLUVIALIS EXTRACT
CAPRYLIC/CAPRIC TRIGLYCERIDE
SODIUM CHLORIDE
POLYSORBATE 20
TRIPEPTIDE-1
HEXAPEPTIDE-9
PALMITOYL TRIPEPTIDE-5
PALMITOYL PENTAPEPTIDE-4
PALMITOYL TETRAPEPTIDE-7
HYDROXYPROPYL TETRAHYDROPYRANTRIOL
DECAPEPTIDE-4
GLYCOPROTEINS
DISODIUM PHOSPHATE
DIPEPTIDE DIAMINOBUTYROYL BENZYLAMIDE DIACETATE
ACETYL HEXAPEPTIDE-8
DIMETHYL ISOSORBIDE
FULLERENES
PVP
ISOHEXADECANE
POLYSORBATE 80
SORBITAN OLEATE
PHENOXYETHANOL
ETHYLHEXYLGLYCERIN
MALTODEXTRIN
XANTHAN GUM
CYCLODEXTRIN1,2-HEXANEDIOL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ATTI AMOUR ROSE ANTI-AGING REPAIR EYE CREAM
niacinamide, glycerin creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84601-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 6.52 g in 100 g GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 2.24 g in 100 g Inactive Ingredients Ingredient Name Strength CYCLODEXTRINS (UNII: 7E6SK9QDT8) ARABICA COFFEE BEAN (UNII: 3SW678MX72) CETEARYL OLIVATE (UNII: 58B69Q84JO) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) MICA (UNII: V8A1AW0880) DIMETHYL ISOSORBIDE (UNII: SA6A6V432S) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) DIPEPTIDE DIAMINOBUTYROYL BENZYLAMIDE DIACETATE (UNII: 38H206R00R) WATER (UNII: 059QF0KO0R) REHMANNIA GLUTINOSA ROOT (UNII: 1BEM3U6LQQ) HAEMATOCOCCUS PLUVIALIS (UNII: 31T0FF0472) BUCKMINSTERFULLERENE (UNII: NP9U26B839) ISOHEXADECANE (UNII: 918X1OUF1E) POLYSORBATE 80 (UNII: 6OZP39ZG8H) PHENOXYETHANOL (UNII: HIE492ZZ3T) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) HYDROXYPROPYL TETRAHYDROPYRANTRIOL (UNII: 4U3GMG1OT1) SORBITAN MONOOLEATE (UNII: 06XEA2VD56) HYALURONATE SODIUM (UNII: YSE9PPT4TH) HYDROLYZED WHEAT PROTEIN (ENZYMATIC, 3000 MW) (UNII: J2S07SB0YL) PENTYLENE GLYCOL (UNII: 50C1307PZG) SODIUM CHLORIDE (UNII: 451W47IQ8X) PALMITOYL PENTAPEPTIDE-4 (UNII: KK181SM5JG) PALMITOYL TETRAPEPTIDE-7 (UNII: Q41S464P1R) HEXAPEPTIDE-9 (UNII: L25414IE41) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ROSA DAMASCENA FLOWER OIL (UNII: 18920M3T13) DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (HARD PARTICLE) (UNII: H895X08VNQ) OAT BRAN (UNII: KQX236OK4U) COLLAGEN, SOLUBLE, FISH SKIN (UNII: 8JC99XGU4W) HYALURONIC ACID (UNII: S270N0TRQY) PALMITOYL TRIPEPTIDE-5 (UNII: 2A3916MQHO) ACETYL HEXAPEPTIDE-8 (UNII: L4EL31FWIL) POVIDONE (UNII: FZ989GH94E) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) METHYLPARABEN (UNII: A2I8C7HI9T) POLYSORBATE 20 (UNII: 7T1F30V5YH) PREZATIDE (UNII: 39TG2H631E) MALTODEXTRIN (UNII: 7CVR7L4A2D) XANTHAN GUM (UNII: TTV12P4NEE) SORBITAN OLIVATE (UNII: MDL271E3GR) ISONONYL ISONONANOATE (UNII: S4V5BS6GCX) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ALLANTOIN (UNII: 344S277G0Z) EUGLENA GRACILIS (UNII: T426JJ26WB) LACTOBACILLUS REUTERI F275 STRAIN (UNII: 3K209OP33H) TERT-BUTYLHYDROQUINONE (UNII: C12674942B) UBIDECARENONE (UNII: EJ27X76M46) ROSA RUGOSA FLOWER (UNII: 969IQC5YJU) ASTROCARYUM MURUMURU SEED BUTTER (UNII: 12V64UPU6R) STANNIC OXIDE (UNII: KM7N50LOS6) DIMETHICONE (UNII: 92RU3N3Y1O) CANOLA OIL (UNII: 331KBJ17RK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84601-003-01 20 g in 1 BOTTLE; Type 0: Not a Combination Product 08/05/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M016 08/05/2024 Labeler - RIVER SUNZ co.,Ltd (695457117) Establishment Name Address ID/FEI Business Operations RIVER SUNZ co.,Ltd 695457117 manufacture(84601-003)