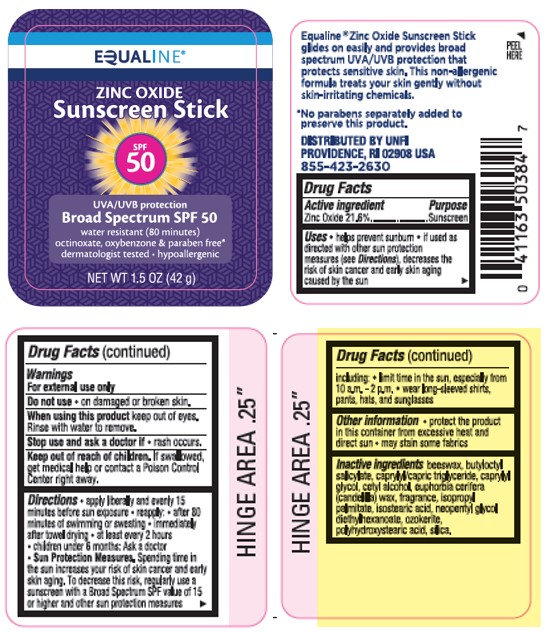
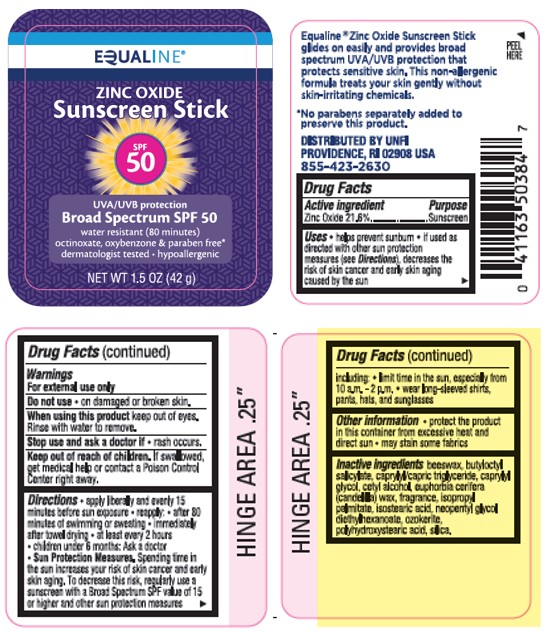
Label: EQUALINE ZINC OXIDE SUNSCREEN STICK SPF 50- zinc oxide stick
- NDC Code(s): 41163-969-28
- Packager: United Natural Foods, Inc. dba UNFI
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated July 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children.
-
Directions
- apply liberally 15 minutes before sun exposure
- reapply:
• after 80 minutes of swimming or sweating
• immediately after towel drying
• at least every 2 hours
- children under 6 months of age: Ask a doctor
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m. - 2 p.m.
• wear long-sleeved shirts, pants, hats, and sunglasses
- Other Information
- Inactive Ingredient(s)
- Label
-
INGREDIENTS AND APPEARANCE
EQUALINE ZINC OXIDE SUNSCREEN STICK SPF 50
zinc oxide stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41163-969 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 216 mg in 1 g Inactive Ingredients Ingredient Name Strength POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) CANDELILLA WAX (UNII: WL0328HX19) SUNFLOWER OIL (UNII: 3W1JG795YI) WHITE WAX (UNII: 7G1J5DA97F) NEOPENTYL GLYCOL DIETHYLHEXANOATE (UNII: U68ZV6W62C) ISOSTEARIC ACID (UNII: X33R8U0062) CETYL ALCOHOL (UNII: 936JST6JCN) COCOA BUTTER (UNII: 512OYT1CRR) CERESIN (UNII: Q1LS2UJO3A) CAPRYLYL GLYCOL (UNII: 00YIU5438U) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41163-969-28 42 g in 1 CONTAINER; Type 0: Not a Combination Product 09/14/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 09/14/2023 Labeler - United Natural Foods, Inc. dba UNFI (943556183)