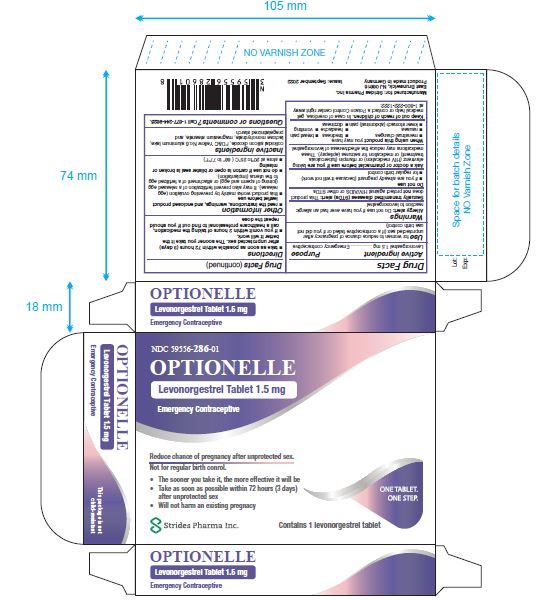
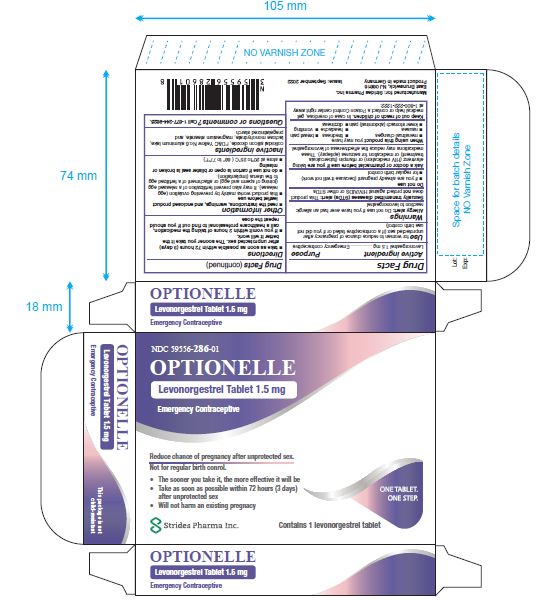
Label: OPTIONELLE- levonorgestrel tablet, 1.5 mg tablet
- NDC Code(s): 59556-286-01
- Packager: Strides Pharma Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated September 16, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- Purpose
- Use
- Warnings
- Do not use
- Ask a doctor or pharmacist before use if you are
- WHEN USING
- Keep out of reach of children.
- Directions
-
Other information
- read the instructions, warnings and enclosed product leaflet before use
- this product works mainly by preventing ovulation (egg release). It may also prevent fertilization of a released egg (joining of sperm and egg) or attachment of a fertilized egg to the uterus (implantation).
- do not use if carton is open or blister seal is broken or missing
- store at 20° to 25°C (68° to 77°F)
- Inactive ingredients
- Questions or comments?
-
INFORMATION FOR PATIENTS
Levonorgestrel Tablet 1.5 mg
- Emergency Contraceptive
- One Tablet. One Step.
- What You Need To Know
Optionelle is emergency contraception that helps prevent pregnancy after birth control failure or unprotected sex. It is a backup method of preventing pregnancy and should not be used as regular birth control.
What Optionelle is not.
Optionelle will not work if you are already pregnant and will not affect an existing pregnancy. Optionelle will not protect you from HIV infection (the virus that causes AIDS) and other sexually transmitted diseases (STDs).
When should I use Optionelle?
The sooner you take emergency contraception, the better it works. You should use Optionelle within 72 hours (3 days) after you have had unprotected sex.
Optionelle is a backup or emergency method of birth control you can use when:
- your regular birth control was used incorrectly or failed
- you did not use any birth control method
Optionelle should not be used:
- as a regular birth control method, because it's not as effective as regular birth control.
- if you are already pregnant, because it will not work.
- if you are allergic to levonorgestrel or any other ingredients in Optionelle.
When should I talk to a doctor or pharmacist?
Ask a doctor or pharmacist before use if you are taking efavirenz (HIV medication) or rifampin (tuberculosis treatment) or medication for seizures (epilepsy). These medications may reduce the effectiveness of Optionelle and increase your chance of becoming pregnant. Your doctor may prescribe another form of emergency contraception that may not be affected by these medications.
How does Optionelle work?
Optionelle is one tablet with levonorgestrel, a hormone that has been used in many birth control pills for several decades. Optionelle contains a higher dose of levonorgestrel than birth control pills, but works in a similar way to prevent pregnancy. It works mainly by stopping the release of an egg from the ovary. It is possible that Optionelle may also work by preventing fertilization of an egg (the uniting of sperm with the egg) or by preventing attachment (implantation) to the uterus (womb).
How can I get the best results from Optionelle?
You have 72 hours (3 days) to try to prevent pregnancy after birth control failure or unprotected sex. The sooner you take Optionelle, the better it works.
How effective is Optionelle?
If Optionelle is taken as directed, it can significantly decrease the chance that you will get pregnant. About 7 out of every 8 women who would have gotten pregnant will not become pregnant.
How will I know Optionelle worked?
You will know Optionelle has been effective when you get your next period, which should come at the expected time, or within a week of the expected time. If your period is delayed beyond 1 week, it is possible you may be pregnant. You should get a pregnancy test and follow up with your healthcare professional.
Will I experience any side effects?
- some women may have changes in their period, such as a period that is heavier or lighter or a period that is early or late. If your period is more than a week late, you may be pregnant.
- if you have severe abdominal pain, you may have an ectopic pregnancy, and should get immediate medical attention.
- when used as directed, Optionelle is safe and effective. Side effects may include changes in your period, nausea, lower stomach (abdominal) pain, tiredness, headache, dizziness, and breast tenderness.
- if you vomit within 2 hours of taking the medication, call a healthcare professional to find out if you should repeat the dose.
What if I still have questions about Optionelle?
If you have questions or need more information, call our toll free number, 1-877-244-9825.
Other Information
Keep out of reach of children:
In case of overdose, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
Do not use if the blister seal is opened.
Store at room temperature 20°-25°C (68° to 77°F).
Active ingredient: levonorgestrel 1.5 mg
Inactive ingredients: colloidal silicon dioxide, FD&C Yellow No. 6 aluminum lake, lactose monohydrate, magnesium stearate, and pregelatinized starch
If you are sexually active, you should see a healthcare provider for routine checkups. Your healthcare provider will talk to you about and, if necessary, test you for sexually transmitted diseases, teach you about effective methods of routine birth control, and answer any other questions you may have.
Manufactured for:
Strides Pharma Inc.
East Brunswick, NJ 08816
Product made in Germany.
NDC 59556-286-01 Rev. September 2022
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
OPTIONELLE
levonorgestrel tablet, 1.5 mg tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59556-286 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVONORGESTREL (UNII: 5W7SIA7YZW) (LEVONORGESTREL - UNII:5W7SIA7YZW) LEVONORGESTREL 1.5 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) Product Characteristics Color ORANGE (Light peach) Score no score Shape ROUND Size 8mm Flavor Imprint Code 17 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59556-286-01 1 in 1 CARTON 09/15/2022 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA202380 09/15/2022 Labeler - Strides Pharma Inc (078868278) Registrant - Naari Pte. Limited (659345996) Establishment Name Address ID/FEI Business Operations Haupt Pharma Muenster GmbH 344172564 ANALYSIS(59556-286) , MANUFACTURE(59556-286)