Label: CKP SAN-4I LIQ- alcohol solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 78235-996-31, 78235-996-51, 78235-996-61, 78235-996-71 - Packager: CK Polymers
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 2, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTION
This is a hand sanitizer manufactured according to the Temporary Policy for Preparation of Certain Alcohol-Based Hand Sanitizer Products During the Public Health Emergency (CoViD-19); Guidance for Industry.
The hand sanitizer is manufactured using only the following United States Pharmacopoeia (USP) grade ingredients in the preparation of the product (percentage in final product formulation) consistent with World Health Organization (WHO) recommendations:
- Alcohol (ethanol) (USP or Food Chemical Codex (FCC) grade) (80%, volume/volume (v/v)) in an aqueous solution denatured according to Alcohol and Tobacco Tax and Trade Bureau regulations in 27 CFR part 20.
- Glycerol (1.45% v/v).
- Hydrogen peroxide (0.125% v/v).
- Sterile distilled water or boiled cold water.
The firm does not add other active or inactive ingredients. Different or additional ingredients may impact the quality and potency of the product.
- Active Ingredient(s)
- Purpose
- Use
- Warnings
- Do not use
-
WHEN USING
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away. - STOP USE
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
-
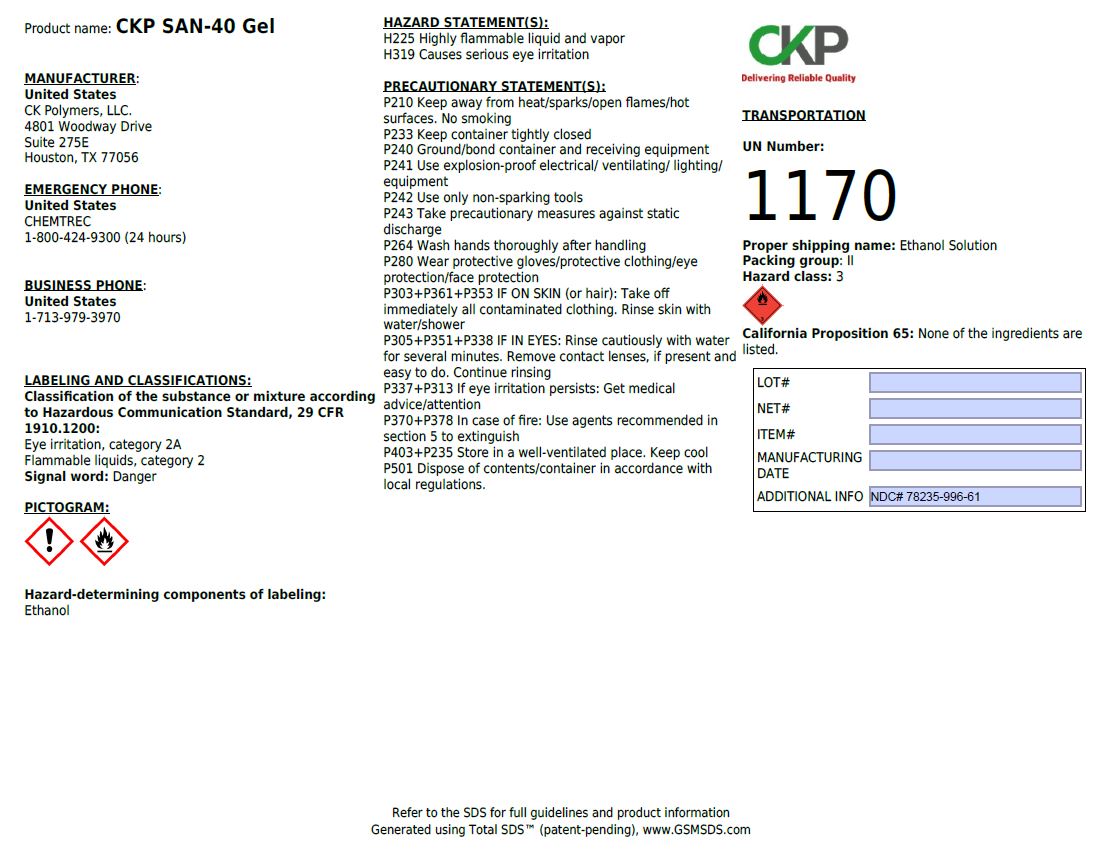
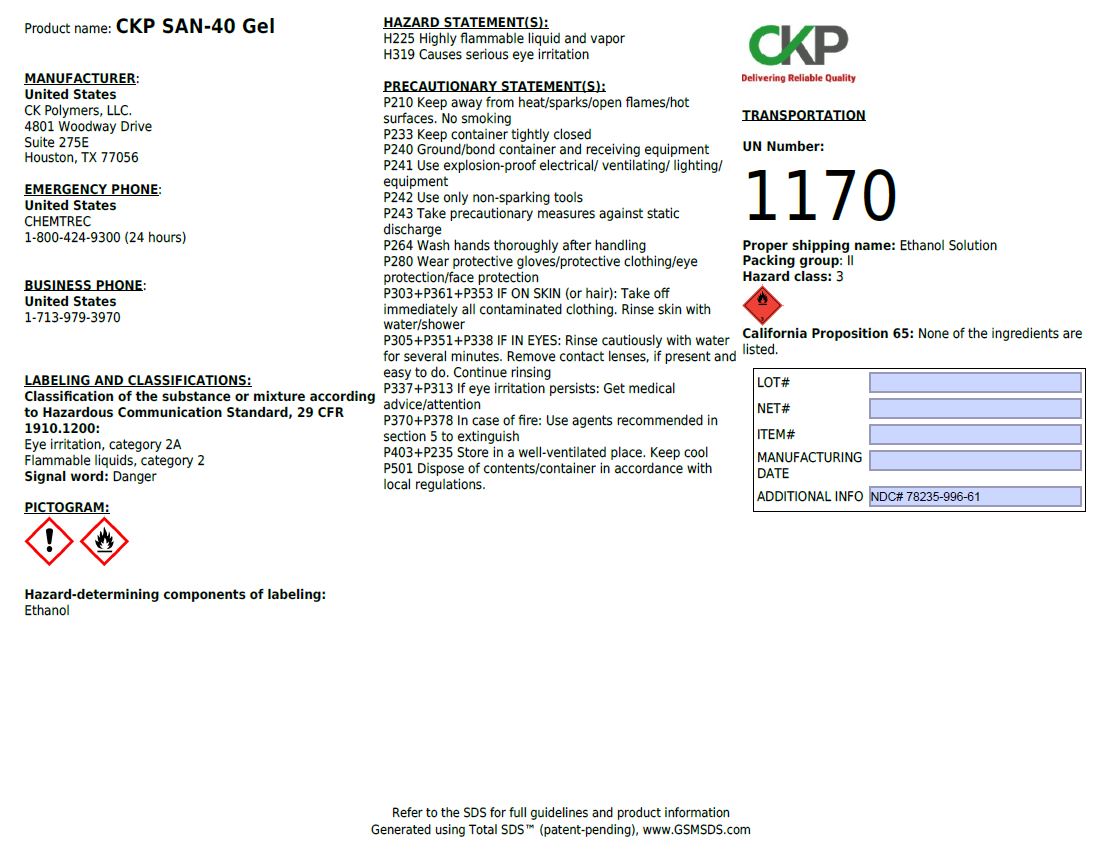
Package Label - Principal Display Panel
1 L (1000 mL) NDC: 78235-996-41

MANUFACTURER:
United States
CK Polymers, LLC
4801 Woodway Drive
Suite 275E
Houston, TX 77056
EMERGENCY PHONE:
United States
CHEMTREC
1-800-424-9300 (24 hours)
BUSINESS PHONE:
United States
1-713-979-3970
LABELING AND CLASSIFICATIONS:
Classification of the substance or mixture according to Hazardous Communication Standard, 29 CFR 1910.1200:
Eye irritation, category 2A
Flammable liquids, category 2
Signal word: Danger
Hazard-determining components of labeling:
Ethanol
HAZARD STATEMENTS:
H224 Highly flammable liquid and vapor
H319 Causes serious eye irritation
PRECAUTIONARY STATEMENTS:
P210 Keep away from heat/sparks/open flames/hot surfaces. No smoking
P233 Keep container tightly closed
P240 Ground/bond container and receiving equipment
P241 Use explosion-proof electrical/ventilating/lighting/equipment
P242 Use only non-sparking tools
P243 Take precautionary measures against static discharge
P264 Wash hands thoroughly after handling
P280 Wear protective gloves/protective clothing/eye protection/face protection
P303+P361+P353 IF ON SKIN (or hair): Take off immediately all contaminated clothing. Rinse skin with water/shower.
P305+P351+P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing
P337+P313 If eye irritation persists: Get medical advice/attention
P370+P378 In case of fire: Use agents recommended in section 5 to extinguish
P403+P235 Store in a well-ventilated place. Keep cool
P501 Dispose of contents/container in accordance with local regulations.
TRANSPORTATION:
UN Number: 1170
Proper shipping name: Ethanol Solution
Packing group: II
Hazard class: 3
California Proposition 65: None of the ingredients are listed.
Refer to the SDS for full guidelines and product information
Generated using Total SDS (patent-pending), www.GSMSDS.com
NDC: 78235-996-51

NDC: 78235-996-61
NDC: 78235-996-71

-
INGREDIENTS AND APPEARANCE
CKP SAN-4I LIQ
alcohol solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:78235-996 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 80 L in 100 L Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) 1.45 L in 100 L HYDROGEN PEROXIDE (UNII: BBX060AN9V) 0.125 L in 100 L WATER (UNII: 059QF0KO0R) 18.425 L in 100 L Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:78235-996-31 1249.19 L in 1 BOTTLE; Type 0: Not a Combination Product 05/29/2020 2 NDC:78235-996-51 3.78541 L in 1 BOTTLE; Type 0: Not a Combination Product 06/25/2020 3 NDC:78235-996-61 18.9271 L in 1 BOTTLE; Type 0: Not a Combination Product 06/25/2020 4 NDC:78235-996-71 208.198 L in 1 BOTTLE; Type 0: Not a Combination Product 06/25/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 05/29/2020 Labeler - CK Polymers (079790285) Establishment Name Address ID/FEI Business Operations CK Polymers 079790285 manufacture(78235-996)