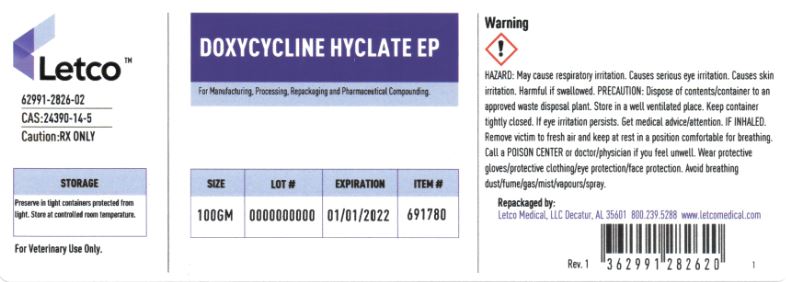
Label: DOXYCYCLINE HYCLATE powder
-
NDC Code(s):
62991-2826-1,
62991-2826-2,
62991-2826-3,
62991-2826-4, view more62991-2826-5
- Packager: LETCO MEDICAL, LLC
- Category: BULK INGREDIENT - ANIMAL DRUG
- DEA Schedule: None
- Marketing Status: Bulk Ingredient For Animal Drug Compounding
Drug Label Information
Updated October 17, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DOXYCYCLINE HYCLATE
doxycycline hyclate powderProduct Information Product Type Item Code (Source) NDC:62991-2826 Route of Administration NOT APPLICABLE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOXYCYCLINE HYCLATE (UNII: 19XTS3T51U) (DOXYCYCLINE - UNII:N12000U13O) DOXYCYCLINE HYCLATE 1 g in 1 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62991-2826-1 25 g in 1 JAR 2 NDC:62991-2826-2 100 g in 1 JAR 3 NDC:62991-2826-3 1000 g in 1 JAR 4 NDC:62991-2826-4 25000 g in 1 DRUM 5 NDC:62991-2826-5 5000 g in 1 PAIL Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BULK INGREDIENT FOR ANIMAL DRUG COMPOUNDING 08/19/2010 05/31/2024 Labeler - LETCO MEDICAL, LLC (079891426)