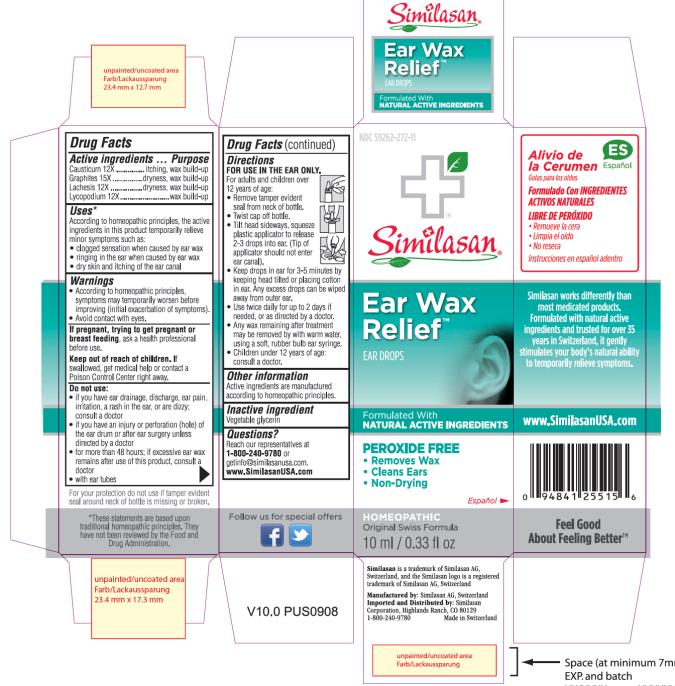
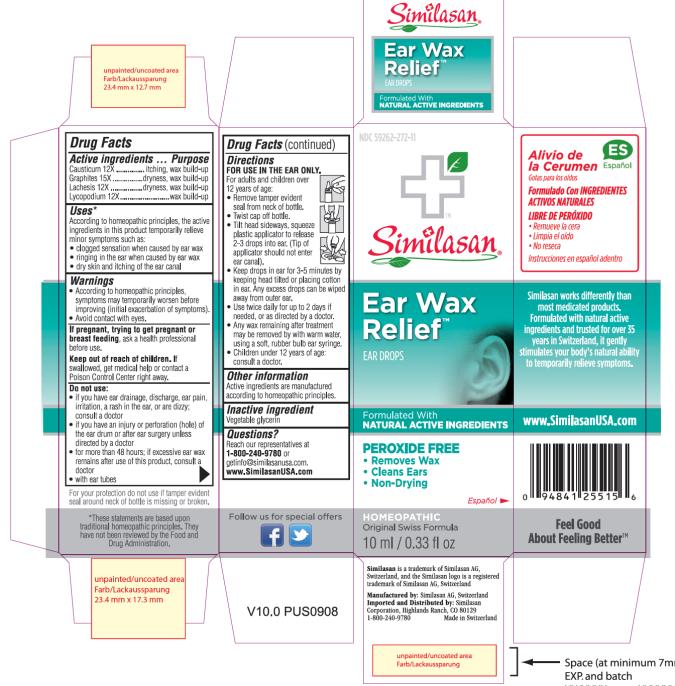
Label: EAR WAX RELIEF- causticum, graphite, lachesis muta venom and lycopodium clavatum spore solution
- NDC Code(s): 59262-272-11
- Packager: Similasan Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated April 16, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Active Ingredients
- Purpose
- Active Ingredient
- Purpose
- Active Ingredient
- Purpose
- Uses:
-
Warnings:
- According to homeopathic principles, symptoms may temporarily worsen before improving (initial exacerbation of symptoms).
- Avoid contact with eyes.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Do not use:
- if you have ear drainage, discharge, ear pain, irritation, a rash in the ear, or are dizzy; consult a doctor
- if you have an injury or perforation (hole) of the ear drum or after ear surgery unless directed by a doctor
- for more than 48 hours; if excessive ear wax remains after the use of this product, consult a doctor
- with ear tubes
- According to homeopathic principles, symptoms may temporarily worsen before improving (initial exacerbation of symptoms).
-
Directions:
FOR USE IN THE EAR ONLY.
For adults and children, over 12 years of age:
- Remove tamper evident seal from neck of bottle.
- Twist cap off bottle.
- Tilt head sideways, squeeze plastic applicator to release 2-3 drops into ear. (Tip of applicator should not enter ear canal).
- Keep drops in ear for 3-5 minutes by keeping head tilted or placing cotton in ear. Any excess drops can be wiped away from outer ear.
- Use twice daily for up to 2 days if needed, or as directed by a doctor.
- Any wax remaining after treatment may be removed by with warm water, using a soft, rubber bulb ear syringe.
- Children under 12 years of age: consult a doctor.
- Remove tamper evident seal from neck of bottle.
- Other information:
- Inactive ingredient:
- Questions?
- Principle Display Panel
-
INGREDIENTS AND APPEARANCE
EAR WAX RELIEF
causticum, graphite, lachesis muta venom and lycopodium clavatum spore solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59262-272 Route of Administration AURICULAR (OTIC) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAUSTICUM (UNII: DD5FO1WKFU) (CAUSTICUM - UNII:DD5FO1WKFU) CAUSTICUM 12 [hp_X] in 10 mL GRAPHITE (UNII: 4QQN74LH4O) (GRAPHITE - UNII:4QQN74LH4O) GRAPHITE 15 [hp_X] in 10 mL LACHESIS MUTA VENOM (UNII: VSW71SS07I) (LACHESIS MUTA VENOM - UNII:VSW71SS07I) LACHESIS MUTA VENOM 12 [hp_X] in 10 mL LYCOPODIUM CLAVATUM SPORE (UNII: C88X29Y479) (LYCOPODIUM CLAVATUM SPORE - UNII:C88X29Y479) LYCOPODIUM CLAVATUM SPORE 12 [hp_X] in 10 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59262-272-11 10 mL in 1 BOTTLE, DROPPER; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 06/01/2003 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 06/01/2003 Labeler - Similasan Corporation (111566530) Registrant - Similasan AG (481545754)