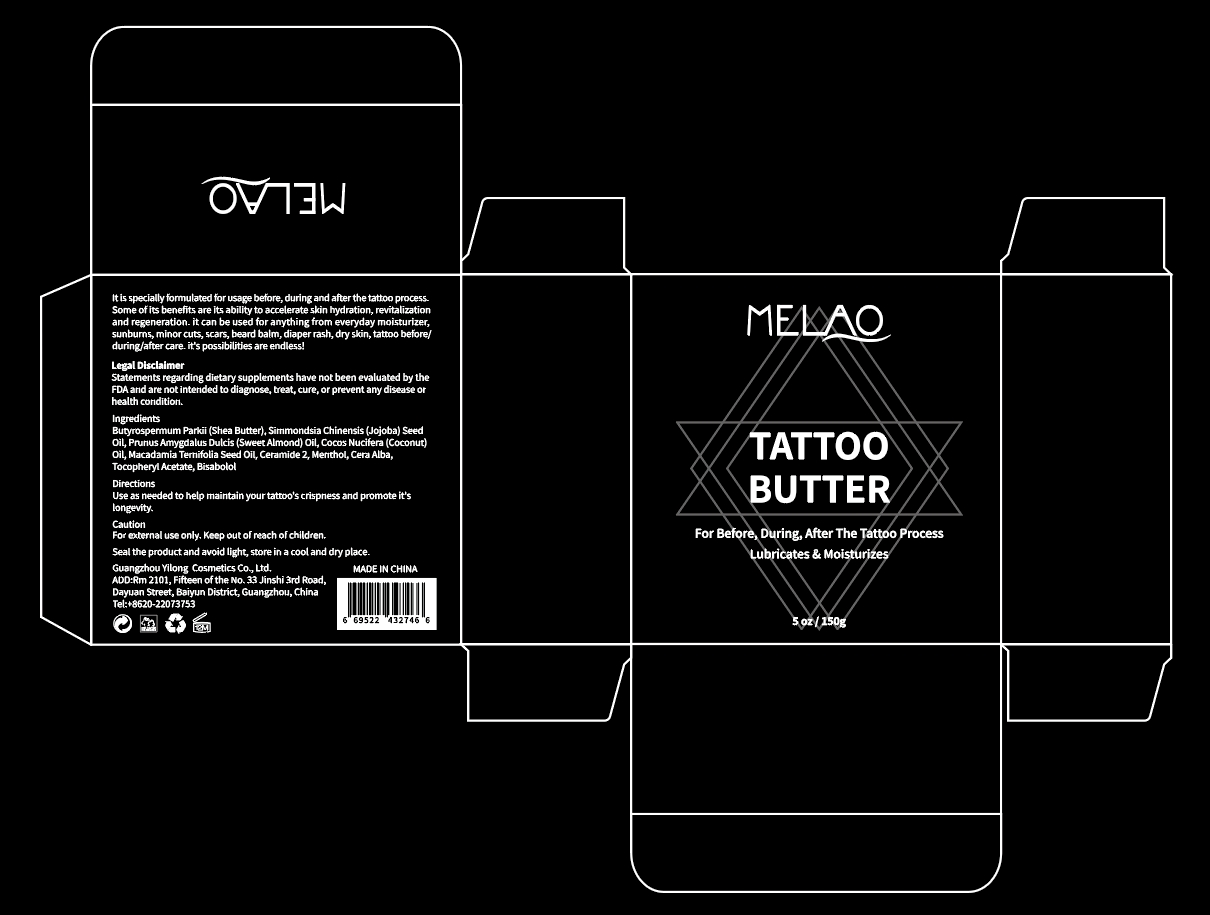
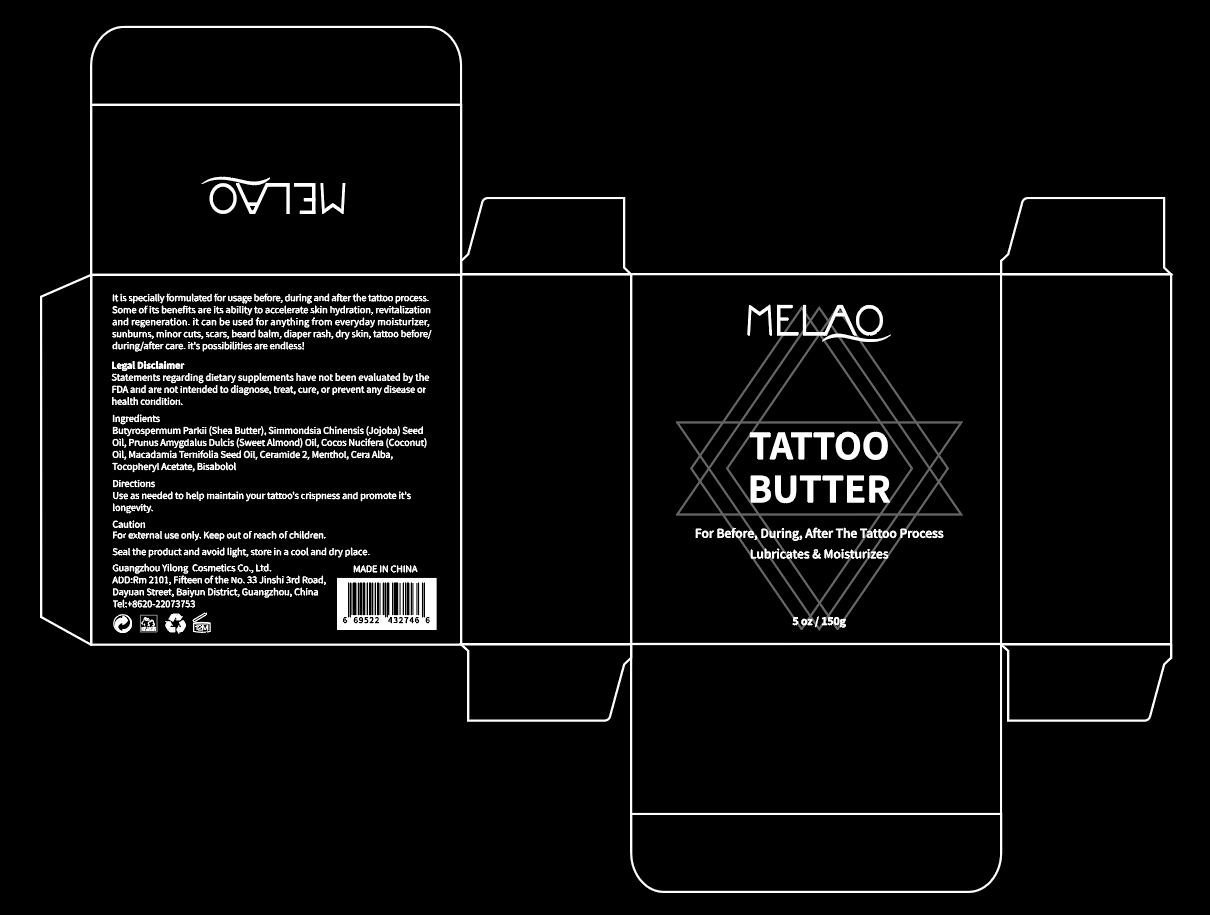
Label: MELAO TATTOO BUTTER- tocopheryl acetate jelly
- NDC Code(s): 83566-110-01
- Packager: Guangzhou Yilong Cosmetics Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 20, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- STOP USE
- DO NOT USE
- WHEN USING
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MELAO TATTOO BUTTER
tocopheryl acetate jellyProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83566-110 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BUTYROSPERMUM PARKII (SHEA) BUTTER UNSAPONIFIABLES (UNII: 0C9AC7D6XU) (BUTYROSPERMUM PARKII (SHEA) BUTTER UNSAPONIFIABLES - UNII:0C9AC7D6XU) BUTYROSPERMUM PARKII (SHEA) BUTTER UNSAPONIFIABLES 56.74 g in 100 g Inactive Ingredients Ingredient Name Strength SUNFLOWER SEED (UNII: R9N3379M4Z) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) SIMMONDSIA CHINENSIS SEED (UNII: D24K2Q1F6H) MACADAMIA TERNIFOLIA WHOLE (UNII: 480C47LI24) SYNTHETIC BEESWAX (UNII: 08MNR5YE2R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83566-110-01 150 g in 1 BOX; Type 0: Not a Combination Product 03/09/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 03/09/2024 Labeler - Guangzhou Yilong Cosmetics Co., Ltd (712647107) Establishment Name Address ID/FEI Business Operations Guangzhou Yilong Cosmetics Co., Ltd 712647107 manufacture(83566-110)