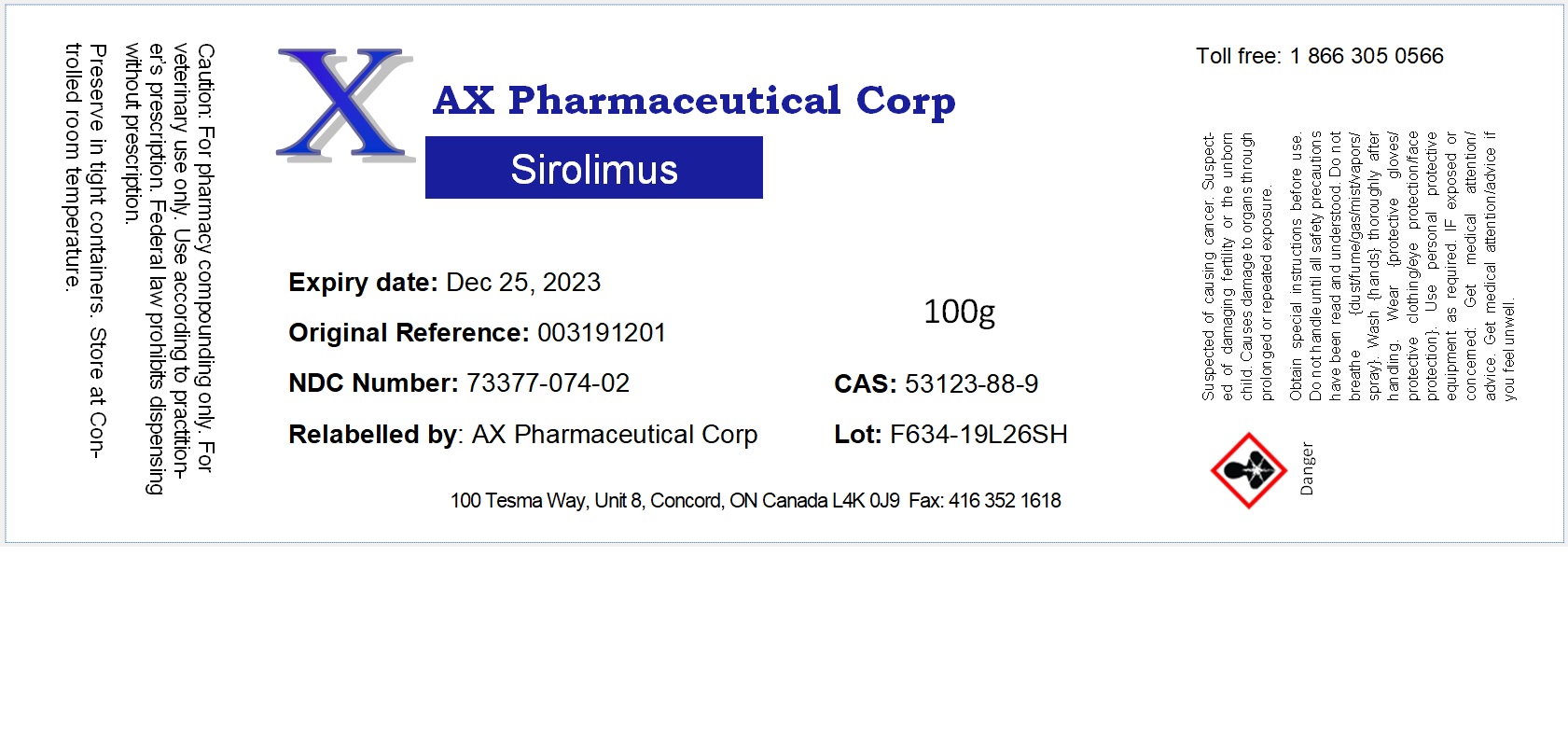
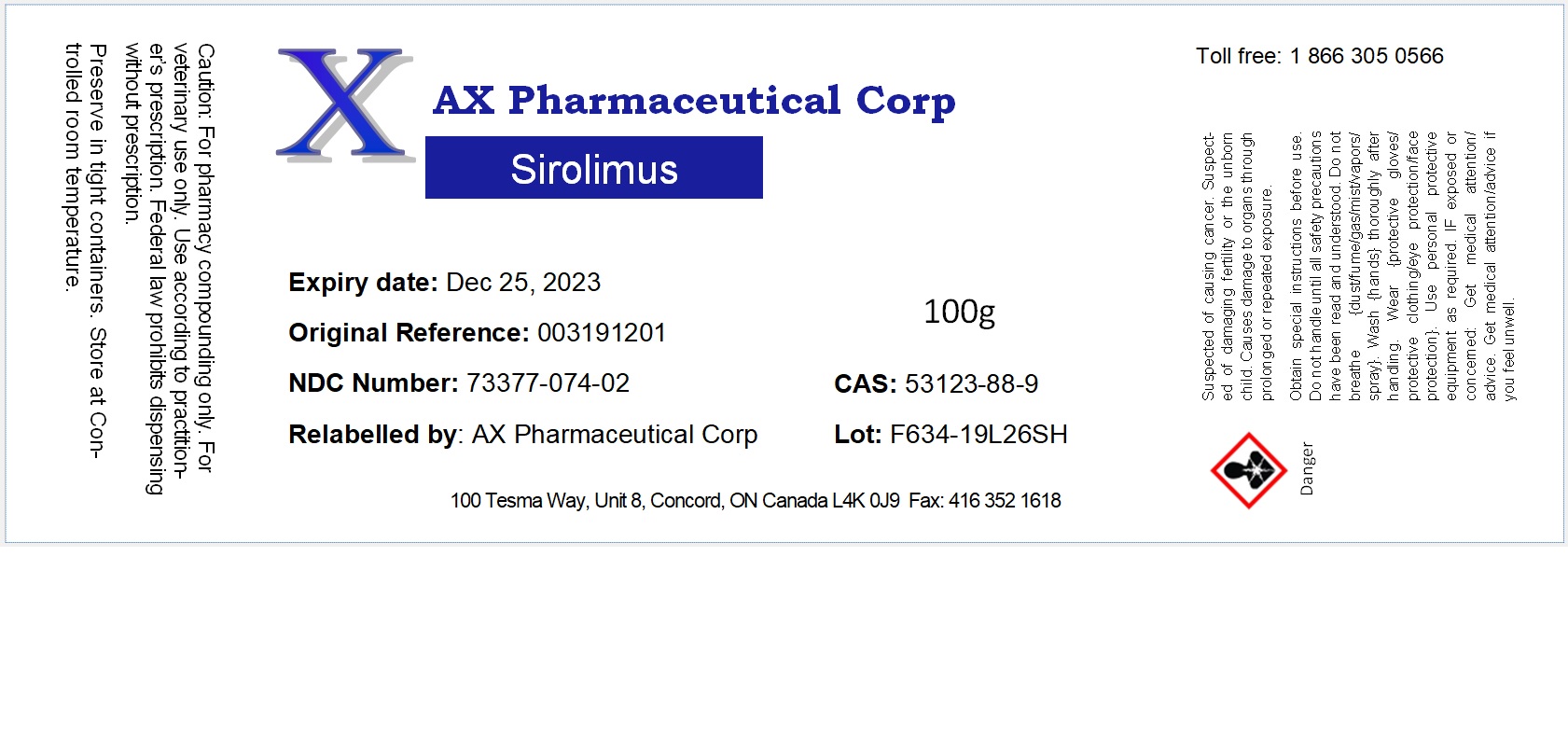
Label: SIROLIMUS powder
- NDC Code(s): 73377-074-01, 73377-074-02
- Packager: AX Pharmaceutical Corp
- Category: BULK INGREDIENT - ANIMAL DRUG
- DEA Schedule: None
- Marketing Status: Bulk Ingredient For Animal Drug Compounding
Drug Label Information
Updated April 21, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Sirolimus
-
INGREDIENTS AND APPEARANCE
SIROLIMUS
sirolimus powderProduct Information Product Type Item Code (Source) NDC:73377-074 Route of Administration NOT APPLICABLE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SIROLIMUS (UNII: W36ZG6FT64) (SIROLIMUS - UNII:W36ZG6FT64) SIROLIMUS 1 g in 1 g Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73377-074-01 10 g in 1 JAR 2 NDC:73377-074-02 100 g in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date bulk ingredient for animal drug compounding 09/29/2020 Labeler - AX Pharmaceutical Corp (204011316) Establishment Name Address ID/FEI Business Operations AX Pharmaceutical Corp 204011316 repack, relabel