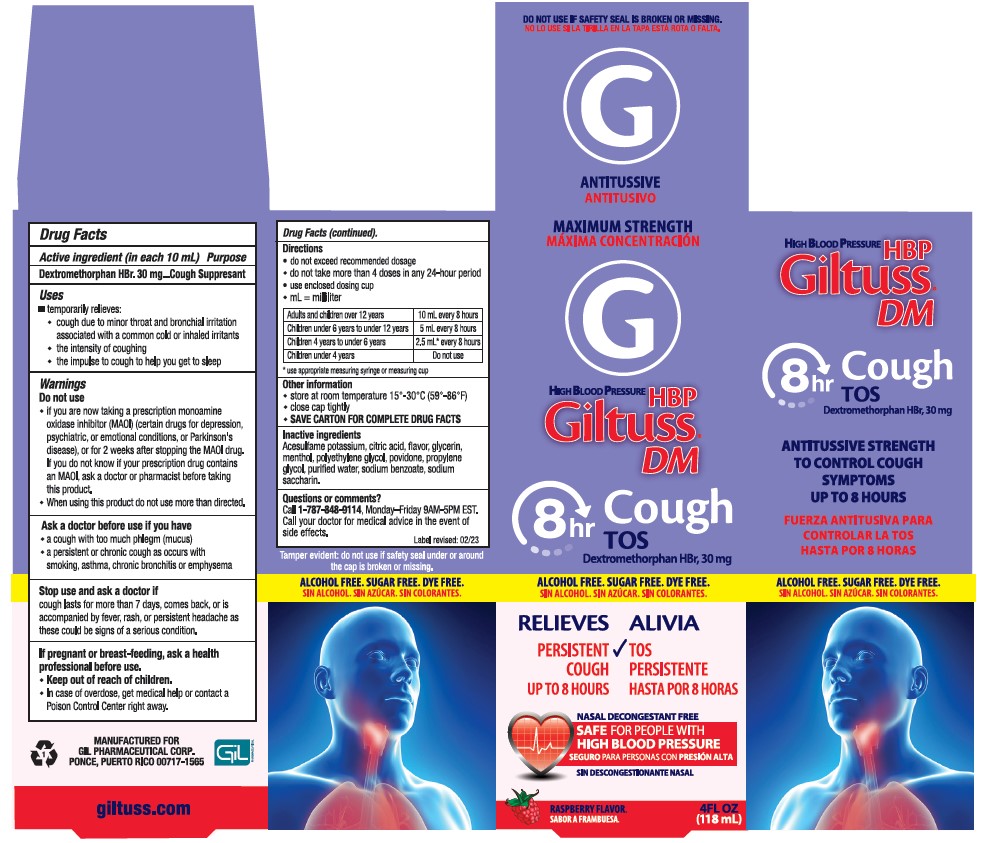
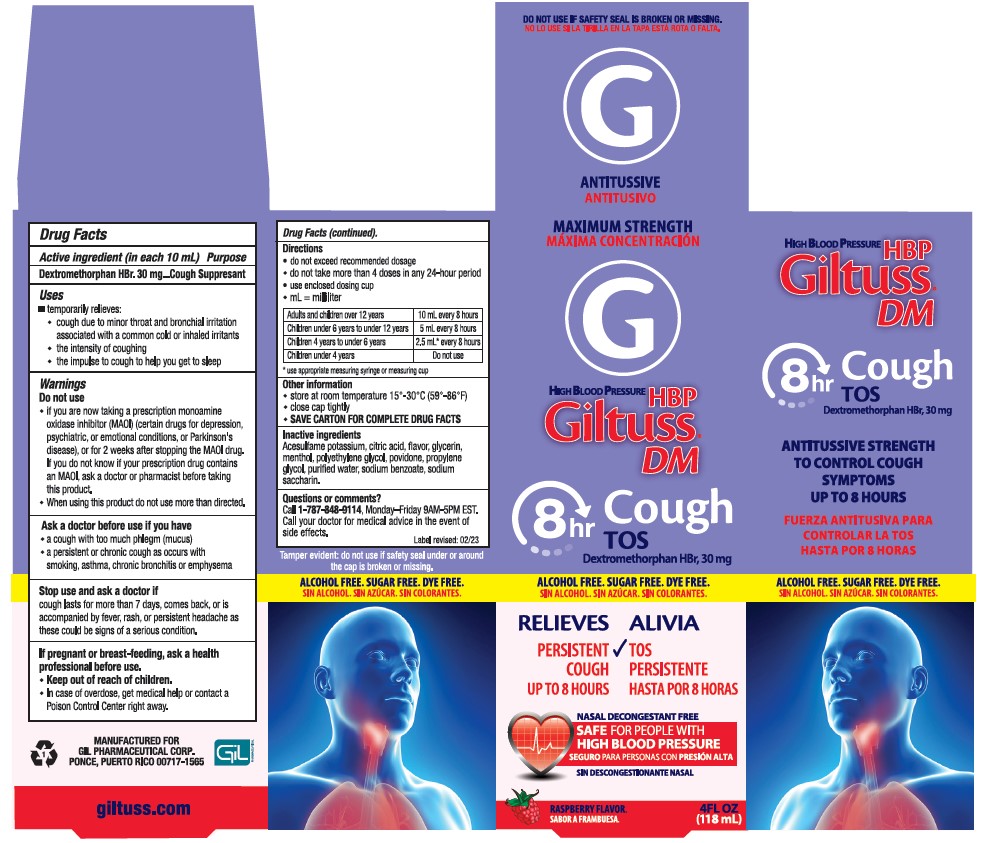
Label: GILTUSS DM- dextromethorphan hbr syrup
- NDC Code(s): 58552-150-04
- Packager: Gil Pharmaceutical Corp
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Do not use
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- When using this product do not use more than directed.
Ask a doctor before use if you have
- a cough with too much phlegm (mucus)
- a persistent or chronic cough as occurs with smoking, asthma, chronic bronchitis or emphysema
-
DOSAGE & ADMINISTRATION
Directions
- do not exceed recommended dosage
- do not take more than 4 doses in any 24-hour period
- use enclosed dosing cup
- ml= milliliter
Adults and children over 12 years
10 mL every 8 hours
Children under 6 years to under 12 years
5 mL every 8 hours
Children 4 years to under 6 years
2.5 mL* every 8 hours
Children under 4 years
Do not use
*use appropriate measuring syringe or measuring cup
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GILTUSS DM
dextromethorphan hbr syrupProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58552-150 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 30 mg in 10 mL Inactive Ingredients Ingredient Name Strength ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) RASPBERRY (UNII: 4N14V5R27W) GLYCERIN (UNII: PDC6A3C0OX) MENTHOL (UNII: L7T10EIP3A) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SACCHARIN SODIUM (UNII: SB8ZUX40TY) Product Characteristics Color white (clear color) Score Shape Size Flavor RASPBERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58552-150-04 1 in 1 CARTON 08/09/2023 1 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 06/27/2023 Labeler - Gil Pharmaceutical Corp (176826592)