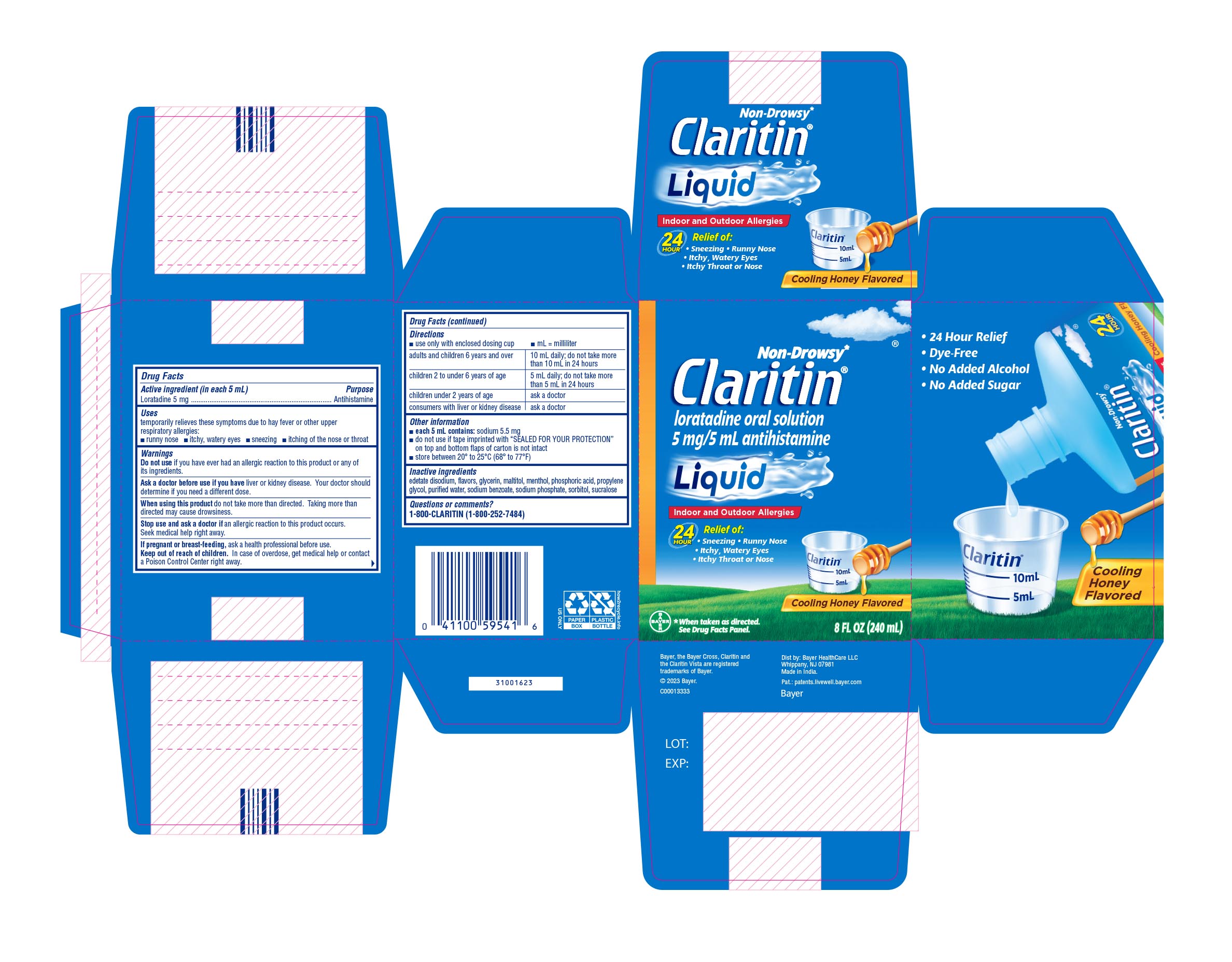
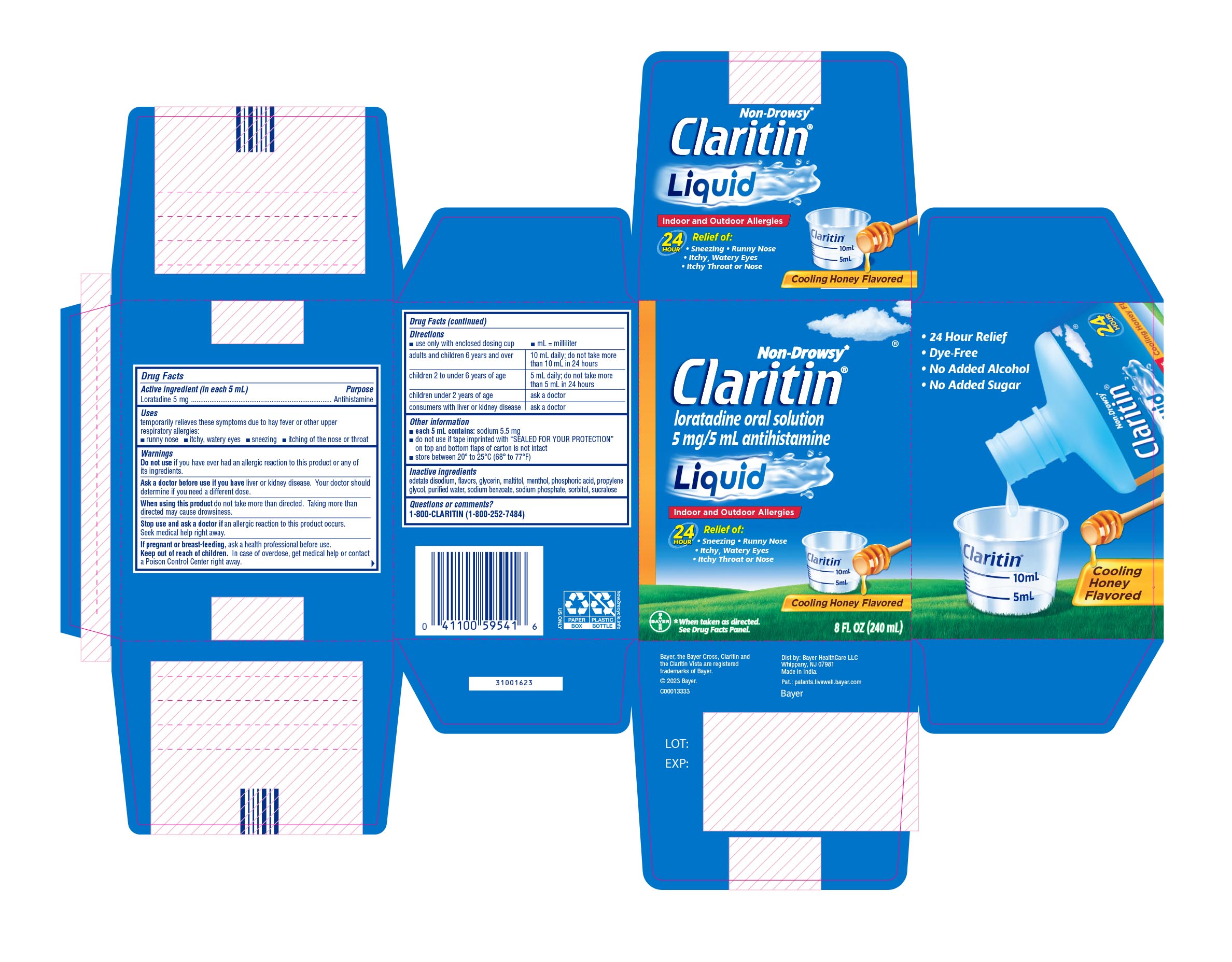
Label: CLARITIN ALLERGY- loratadine solution
- NDC Code(s): 11523-0101-1, 11523-0101-2, 11523-0101-3
- Packager: Bayer HealthCare LLC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated May 30, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient (in each 5 mL teaspoonful)
- Purpose
- Uses
- Warnings
- DO NOT USE
- ASK DOCTOR
- WHEN USING
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other Information
- INACTIVE INGREDIENT
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CLARITIN ALLERGY
loratadine solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11523-0101 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength EDETATE DISODIUM (UNII: 7FLD91C86K) SORBITOL (UNII: 506T60A25R) MALTITOL (UNII: D65DG142WK) GLYCERIN (UNII: PDC6A3C0OX) PHOSPHORIC ACID (UNII: E4GA8884NN) SUCRALOSE (UNII: 96K6UQ3ZD4) WATER (UNII: 059QF0KO0R) SODIUM PHOSPHATE, MONOBASIC (UNII: 3980JIH2SW) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM BENZOATE (UNII: OJ245FE5EU) Product Characteristics Color yellow (Clear, colorless to light yellow) Score Shape Size Flavor HONEY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11523-0101-1 1 in 1 CARTON 06/28/2024 1 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:11523-0101-2 1 in 1 CARTON 06/28/2024 2 80 mL in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:11523-0101-3 1 in 1 CARTON 06/28/2024 3 240 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020641 05/22/2024 Labeler - Bayer HealthCare LLC. (112117283)