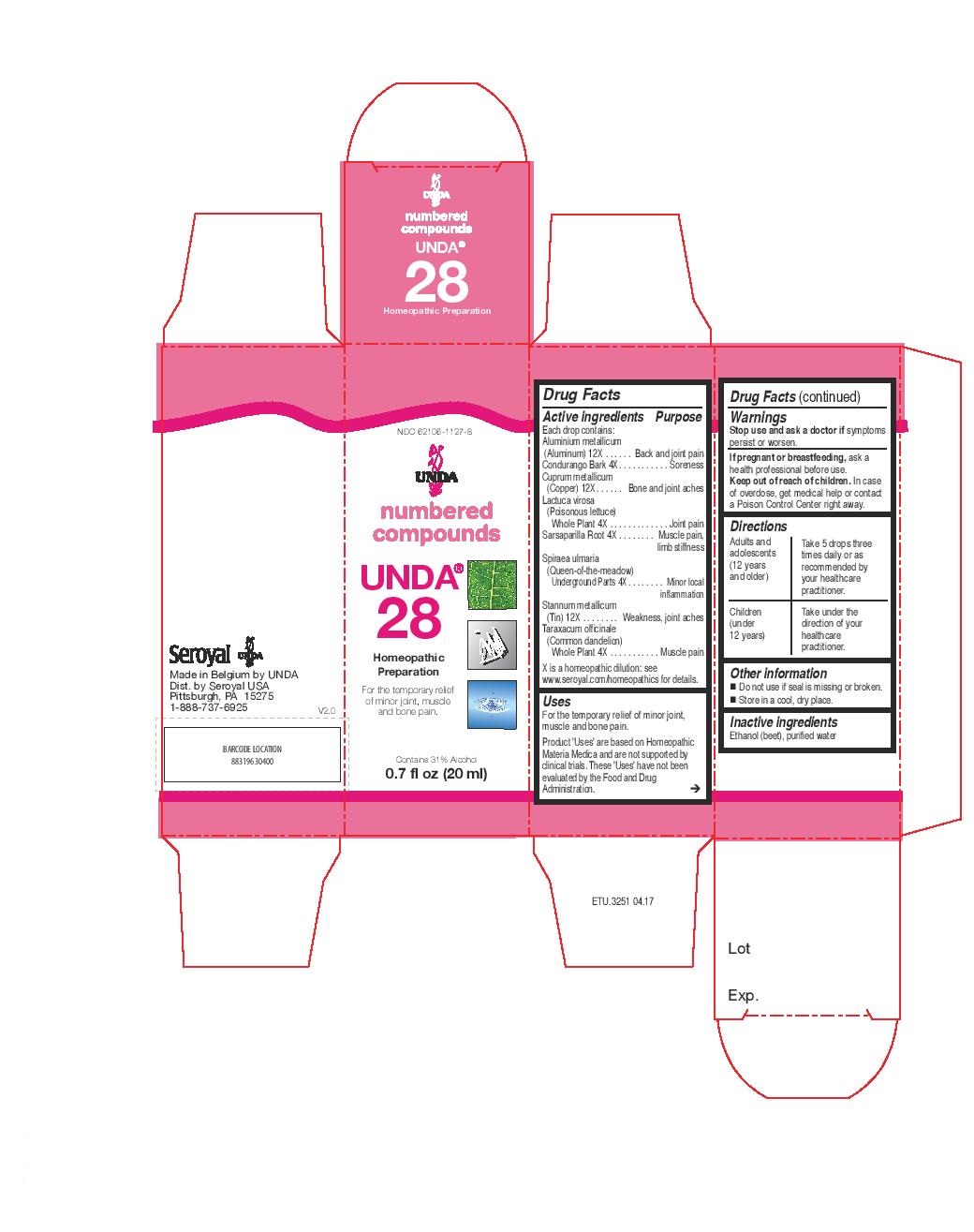
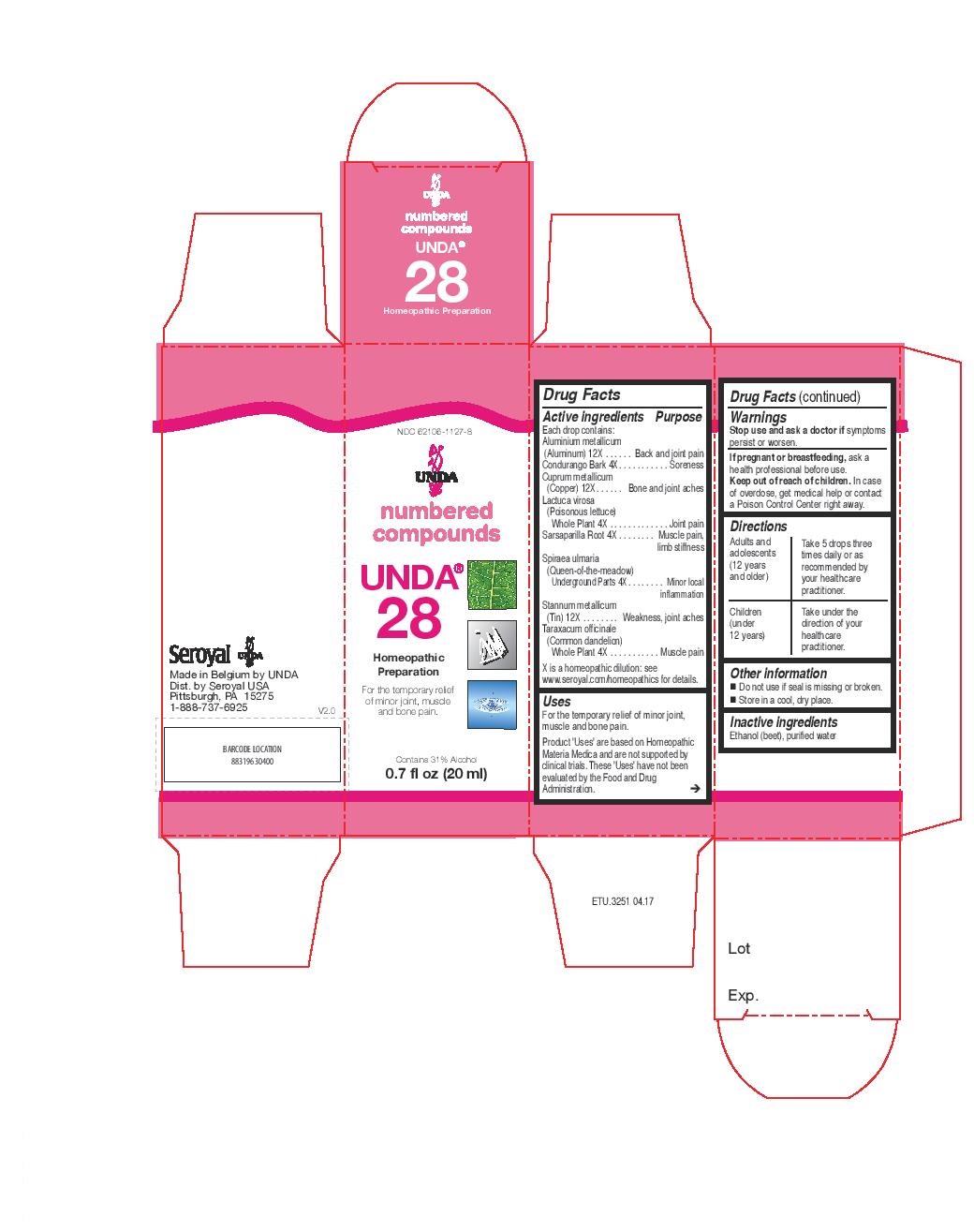
Label: UNDA 28- lactuca virosa, taraxacum officinale, sarsaparilla, spiraea ulmaria, condurango, aluminium metallicum, cuprum metallicum, stannum metallicum liquid
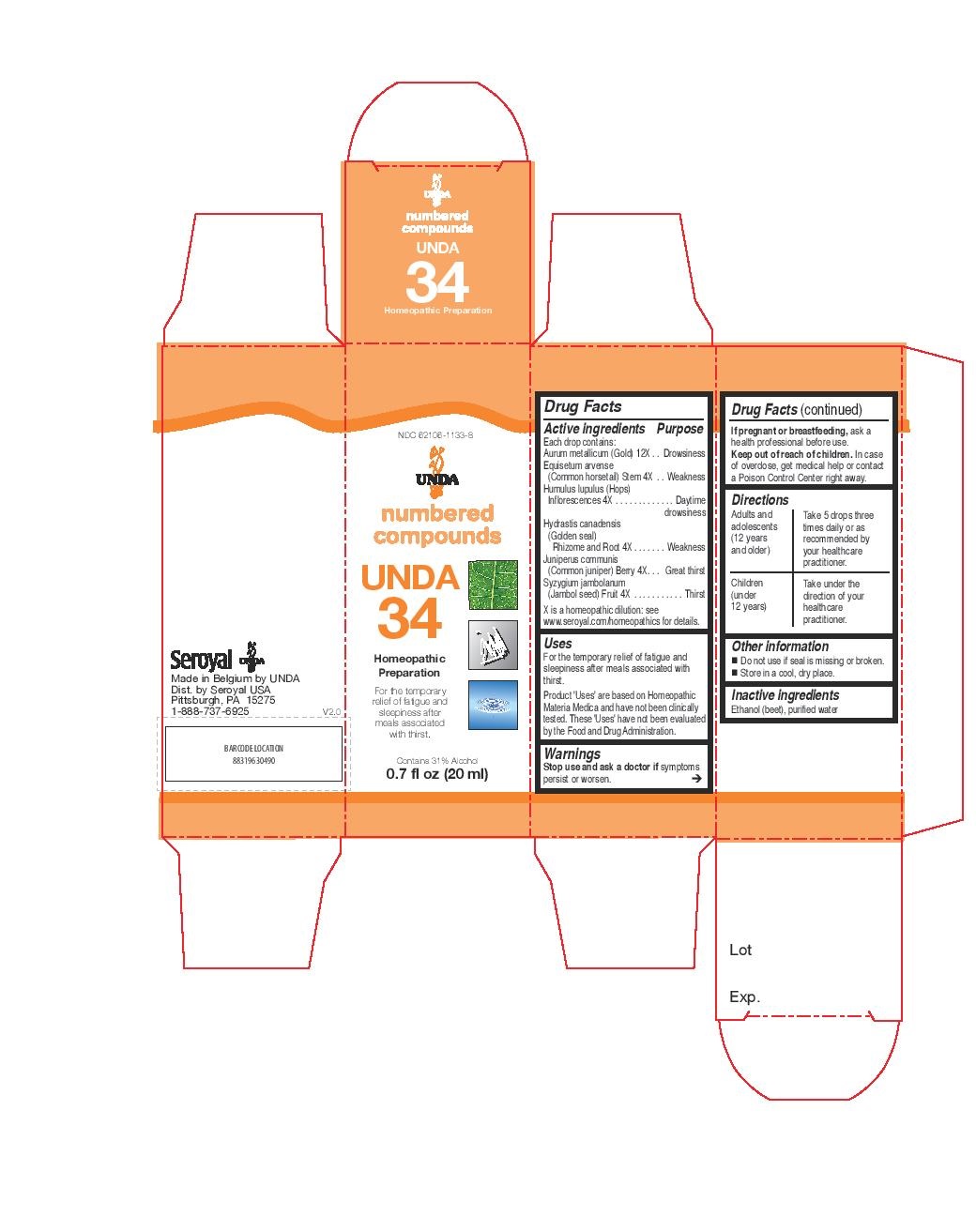
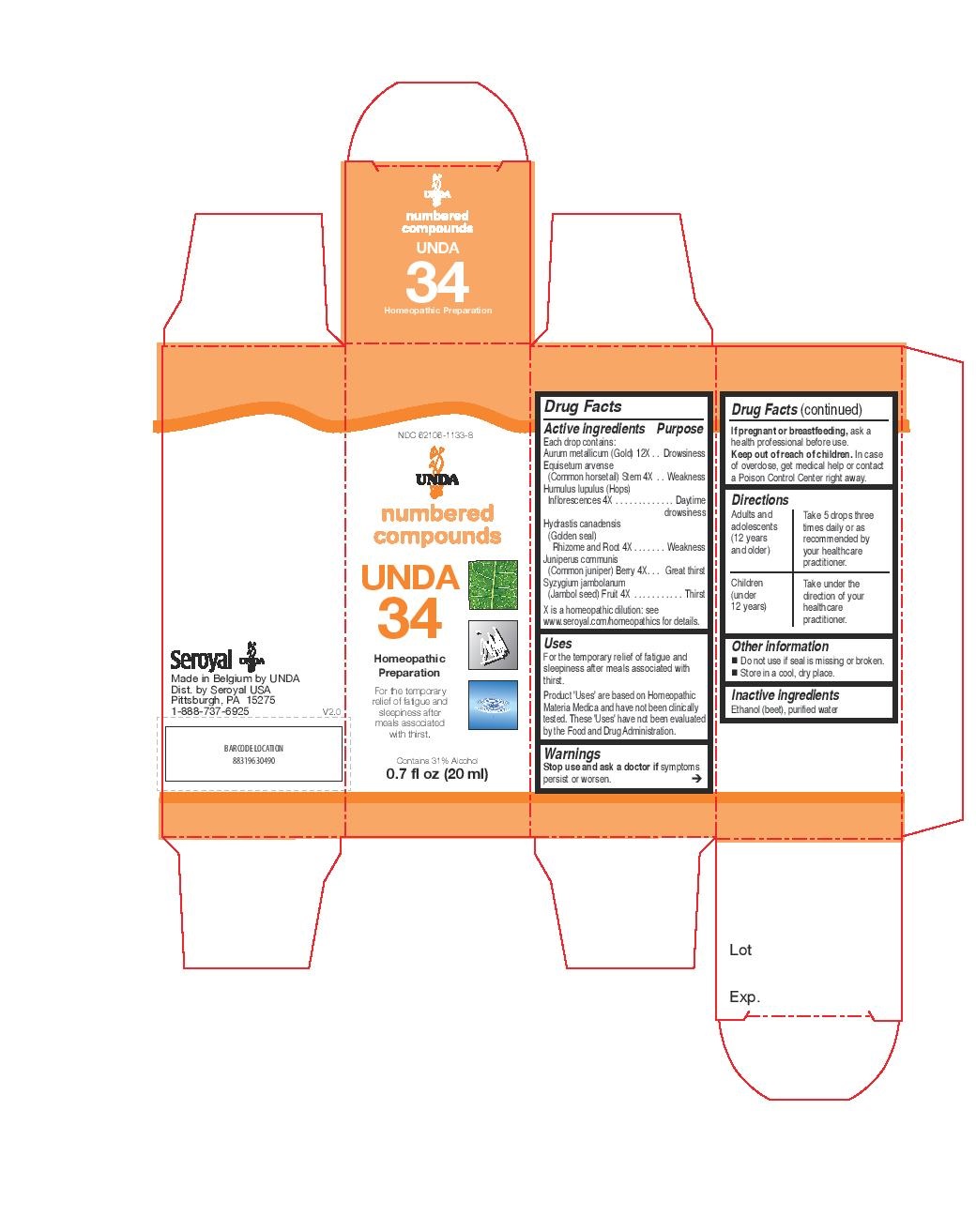
UNDA 34- humulus lupulus, juniperus communis, equisetum arvense, hydrastis canadensis, syzygium jambolanum, aurum metallicum liquid
- NDC Code(s): 62106-1127-8, 62106-1133-8
- Packager: Seroyal USA
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated November 9, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active ingredients
Each drop contains:
Aluminium metallicum (Aluminum) 12X
Condurango Bark 4X
Cuprum metallicum (Copper) 12X
Lactuca virosa (Poisonous lettuce) Whole Plant 4X
Sarsaparilla Root 4X
Spiraea ulmaria (Queen-of-the-meadow) Underground Parts 4X
Stannum metallicum (Tin) 12X
Taraxacum officinale (Common dandelion) Whole Plant 4X - PURPOSE
- WARNINGS
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- OVERDOSAGE
- ASK DOCTOR
- INACTIVE INGREDIENT
- OTHER SAFETY INFORMATION
- DOSAGE & ADMINISTRATION
- INDICATIONS & USAGE
- ACTIVE INGREDIENT
- PURPOSE
- WARNINGS
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- OVERDOSAGE
- ASK DOCTOR
- INACTIVE INGREDIENT
- OTHER SAFETY INFORMATION
- DOSAGE & ADMINISTRATION
-
INDICATIONS & USAGE
Uses
For the temporary relief of fatigue and sleepiness after meals associated with thirst.Directions
Adults and adolescents (12 years and older)Take 5 drops three times daily or as recommended by your healthcare practitioner.
Children (under 12 years)Take under the direction of your healthcare practitioner.
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
UNDA 28
lactuca virosa, taraxacum officinale, sarsaparilla, spiraea ulmaria, condurango, aluminium metallicum, cuprum metallicum, stannum metallicum liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62106-1127 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SARSAPARILLA (UNII: 2H1576D5WG) (SARSAPARILLA - UNII:2H1576D5WG) SARSAPARILLA 4 [hp_X] in 20 mL FILIPENDULA ULMARIA ROOT (UNII: 997724QNDS) (FILIPENDULA ULMARIA ROOT - UNII:997724QNDS) FILIPENDULA ULMARIA ROOT 4 [hp_X] in 20 mL ALUMINUM (UNII: CPD4NFA903) (ALUMINUM - UNII:CPD4NFA903) ALUMINUM 12 [hp_X] in 20 mL COPPER (UNII: 789U1901C5) (COPPER - UNII:789U1901C5) COPPER 12 [hp_X] in 20 mL TIN (UNII: 387GMG9FH5) (TIN - UNII:387GMG9FH5) TIN 12 [hp_X] in 20 mL MARSDENIA CONDURANGO BARK (UNII: R23QIR6YBA) (MARSDENIA CONDURANGO BARK - UNII:R23QIR6YBA) MARSDENIA CONDURANGO BARK 4 [hp_X] in 20 mL TARAXACUM OFFICINALE WHOLE (UNII: 7C42E2D7B0) (TARAXACUM OFFICINALE WHOLE - UNII:7C42E2D7B0) TARAXACUM OFFICINALE WHOLE 4 [hp_X] in 20 mL LACTUCA VIROSA (UNII: 6D74QW4H67) (LACTUCA VIROSA - UNII:6D74QW4H67) LACTUCA VIROSA 4 [hp_X] in 20 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62106-1127-8 1 in 1 CARTON 06/10/2015 1 20 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 06/10/2015 UNDA 34
humulus lupulus, juniperus communis, equisetum arvense, hydrastis canadensis, syzygium jambolanum, aurum metallicum liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62106-1133 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOPS (UNII: 01G73H6H83) (HOPS - UNII:01G73H6H83) HOPS 4 [hp_X] in 20 mL JUNIPERUS COMMUNIS WHOLE (UNII: 464910T5N9) (JUNIPERUS COMMUNIS WHOLE - UNII:464910T5N9) JUNIPERUS COMMUNIS WHOLE 4 [hp_X] in 20 mL EQUISETUM ARVENSE BRANCH (UNII: 1L0VKZ185E) (EQUISETUM ARVENSE BRANCH - UNII:1L0VKZ185E) EQUISETUM ARVENSE BRANCH 4 [hp_X] in 20 mL GOLDENSEAL (UNII: ZW3Z11D0JV) (GOLDENSEAL - UNII:ZW3Z11D0JV) GOLDENSEAL 4 [hp_X] in 20 mL SYZYGIUM CUMINI SEED (UNII: 820LSF646I) (SYZYGIUM CUMINI SEED - UNII:820LSF646I) SYZYGIUM CUMINI SEED 4 [hp_X] in 20 mL GOLD (UNII: 79Y1949PYO) (GOLD - UNII:79Y1949PYO) GOLD 12 [hp_X] in 20 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62106-1133-8 1 in 1 CARTON 06/10/2015 1 20 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 06/10/2015 Labeler - Seroyal USA (018361118) Establishment Name Address ID/FEI Business Operations SAN’UP 401010287 manufacture(62106-1127, 62106-1133)