Label: DENTALCLEAN WHITENING MAX- sodium fluoride 0.24% paste, dentifrice
- NDC Code(s): 84035-004-03
- Packager: Rabbit Indústria e Comércio de Produtos de Higiene Pessoal Ltda
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 7, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
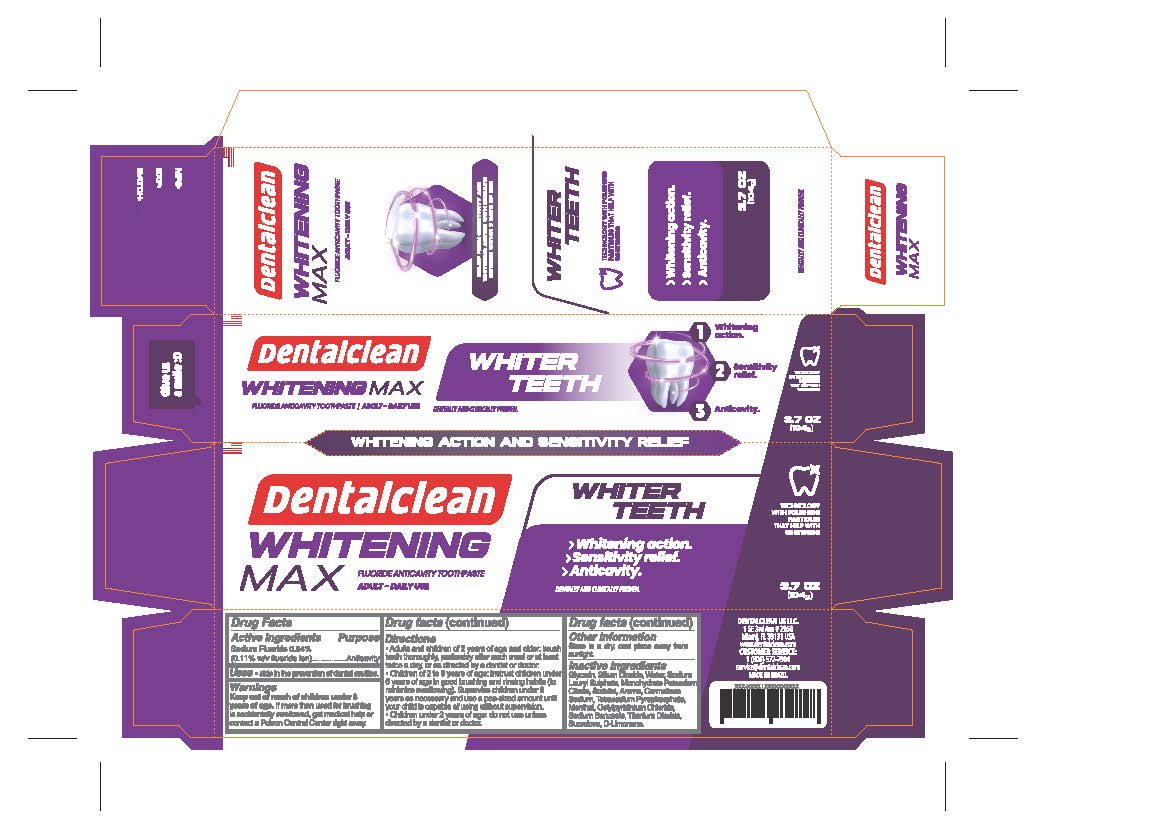
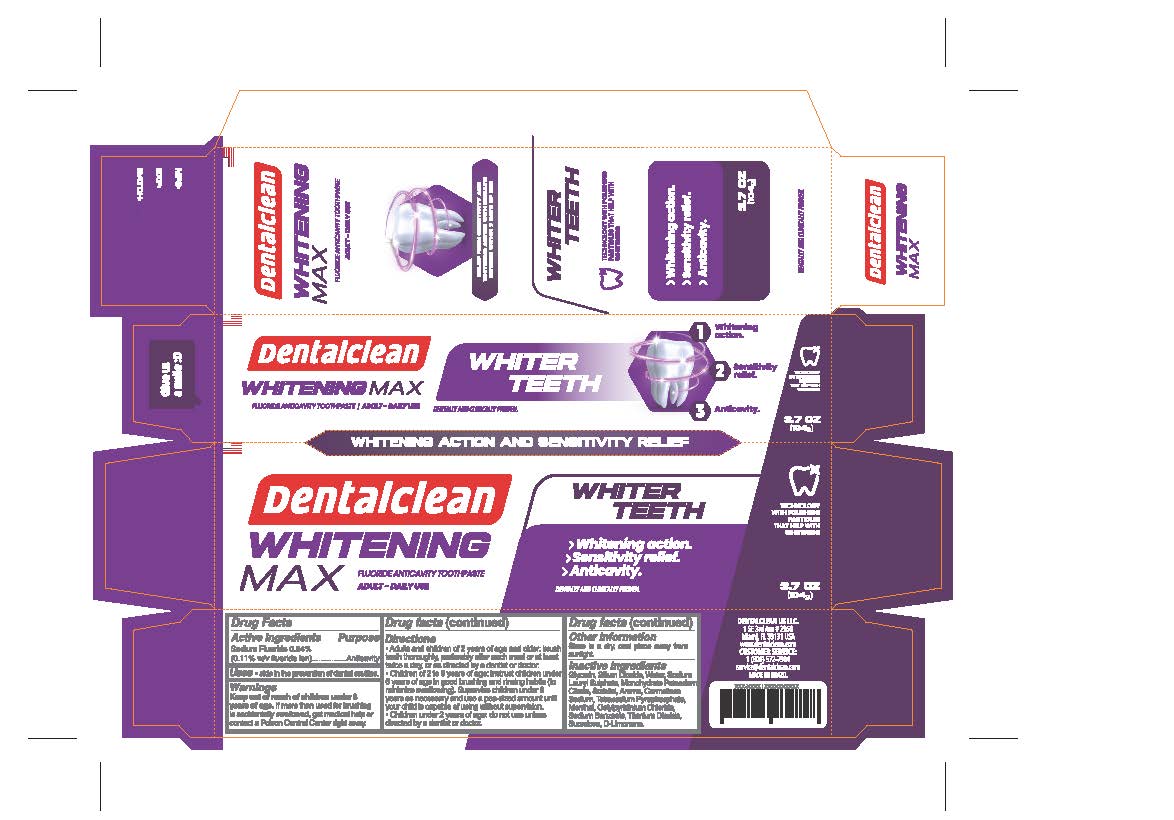
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
-
Directions
- Adults and children 2 years of age and older: brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or doctor.
• Children 2 to 6 years of age: instruct children under 6 years of age in good brushing and rinsing habits (to minimize swallowing). Supervise children under 6 years as necessary and use a pea-sized amount until your child is capable of using without supervision.
• Children under 2 years of age: do not use unless directed by a dentist or doctor or as directed by a dentist or doctor.
- OTHER SAFETY INFORMATION
- Inactive Ingredients
- KEEP OUT OF REACH OF CHILDREN
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DENTALCLEAN WHITENING MAX
sodium fluoride 0.24% paste, dentifriceProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84035-004 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) SODIUM FLUORIDE 2.4 mg in 1 g Inactive Ingredients Ingredient Name Strength MENTHOL (UNII: L7T10EIP3A) SUCRALOSE (UNII: 96K6UQ3ZD4) CETYLPYRIDINIUM CHLORIDE ANHYDROUS (UNII: 6BR7T22E2S) SODIUM LAURYL SULFATE (UNII: 368GB5141J) WATER (UNII: 059QF0KO0R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SORBITOL (UNII: 506T60A25R) POTASSIUM CITRATE (UNII: EE90ONI6FF) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) GLYCERIN (UNII: PDC6A3C0OX) SODIUM BENZOATE (UNII: OJ245FE5EU) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SODIUM PYROPHOSPHATE (UNII: O352864B8Z) LIMONENE, (+/-)- (UNII: 9MC3I34447) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84035-004-03 1 in 1 CARTON 06/01/2024 1 104 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M021 06/01/2024 Labeler - Rabbit Indústria e Comércio de Produtos de Higiene Pessoal Ltda (901719567) Establishment Name Address ID/FEI Business Operations Rabbit Indústria e Comércio de Produtos de Higiene Pessoal Ltda 901719567 manufacture(84035-004)