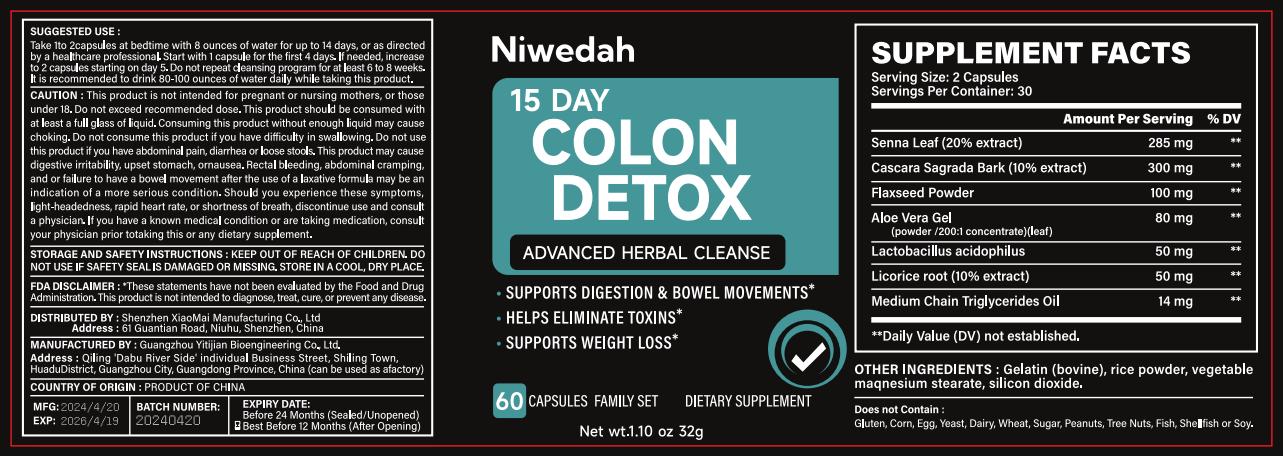
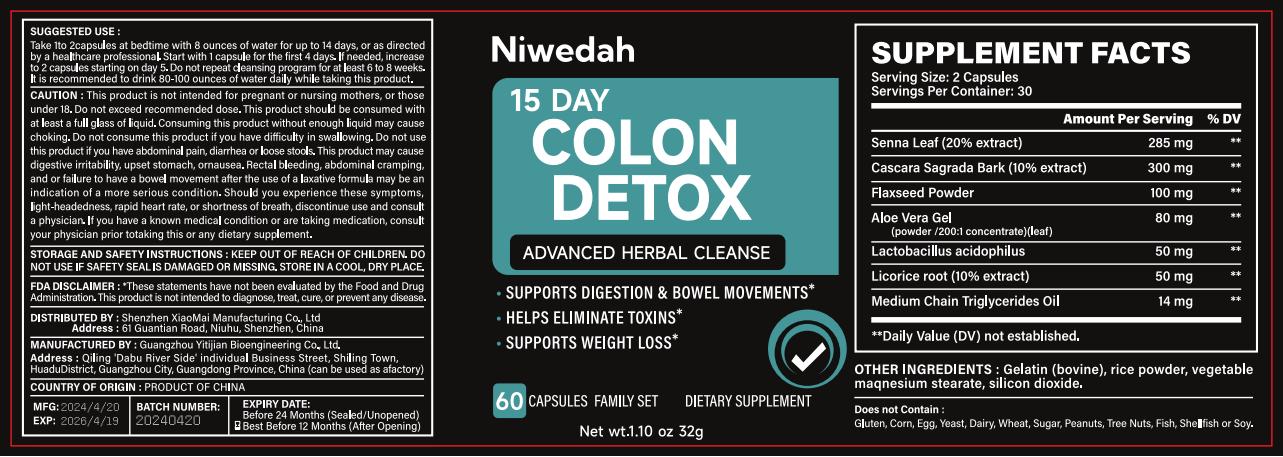
Label: 15 DAY COLON DETOX ADVANCED HERBAL CLEANSE capsule
- NDC Code(s): 83872-131-01
- Packager: Shenzhen XiaoMai Manufacturing Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 7, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- Uses
- WARNINGS
-
Do not use
CAUTlON : Do not consumethis productif you have difficulty in swallowing, This product is notintended for pregnantor nursing mothers or childrenunder the age of 18, Do not use this product if youhaveabdominal pain, diarrhea or loose stools, Rectalbleeding, abdominal crampingand or failure to have abowel.
- When using this product
- Stop use and ask a doctor
- Keep out of reach of children.
- Directions for use
- INACTIVE INGREDIENT
- Other Information
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
15 DAY COLON DETOX ADVANCED HERBAL CLEANSE
15 day colon detox advanced herbal cleanse capsuleProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83872-131 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LACTOBACILLUS ACIDOPHILUS (UNII: 1PRR1V42V5) (LACTOBACILLUS ACIDOPHILUS - UNII:1PRR1V42V5) LACTOBACILLUS ACIDOPHILUS 1.6 mg in 1 g LINSEED OIL (UNII: 84XB4DV00W) (LINSEED OIL - UNII:84XB4DV00W) LINSEED OIL 3.1 mg in 1 g SENNA LEAF (UNII: AK7JF626KX) (SENNA LEAF - UNII:AK7JF626KX) SENNA LEAF 8.9 mg in 1 g ALOE VERA LEAF (UNII: ZY81Z83H0X) (ALOE VERA LEAF - UNII:ZY81Z83H0X) ALOE VERA LEAF 2.5 mg in 1 g LICORICE (UNII: 61ZBX54883) (LICORICE - UNII:61ZBX54883) LICORICE 1.6 mg in 1 g Inactive Ingredients Ingredient Name Strength GELATIN (UNII: 2G86QN327L) 291.2 mg in 1 g MAGNESIUM STEARATE (UNII: 70097M6I30) 145.6 mg in 1 g SILICON DIOXIDE (UNII: ETJ7Z6XBU4) 48.5 mg in 1 g Product Characteristics Color white Score no score Shape CAPSULE Size 200mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83872-131-01 80 g in 1 BOTTLE; Type 0: Not a Combination Product 05/07/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 05/07/2024 Labeler - Shenzhen XiaoMai Manufacturing Co., Ltd. (712999147) Establishment Name Address ID/FEI Business Operations Shenzhen XiaoMai Manufacturing Co., Ltd. 712999147 manufacture(83872-131)