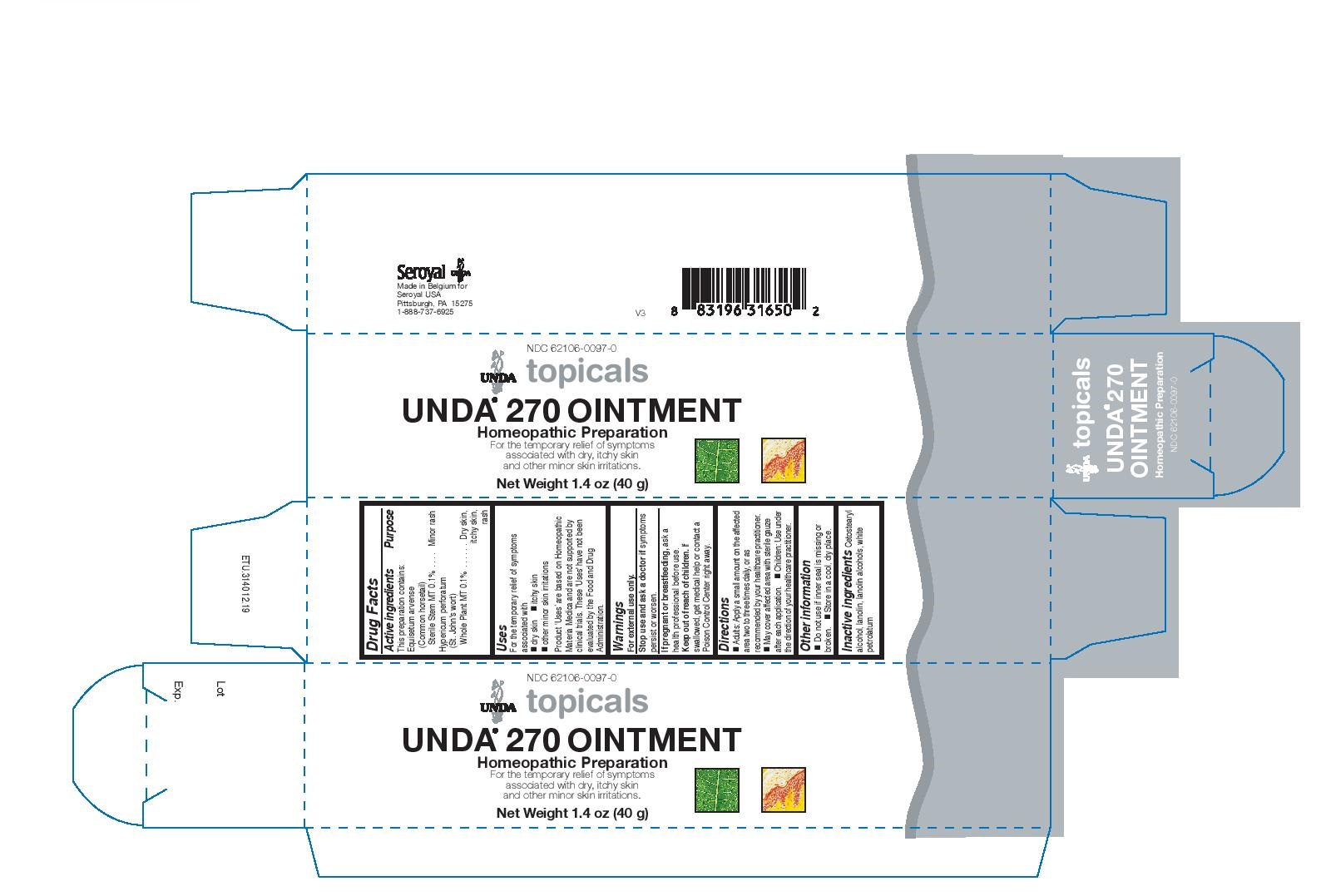
Label: UNDA 270- equisetum arvense, hypericum perforatum cream
- NDC Code(s): 62106-0097-0
- Packager: Seroyal USA
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated September 23, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- OVERDOSAGE
- INACTIVE INGREDIENT
- OTHER SAFETY INFORMATION
- DOSAGE & ADMINISTRATION
-
INDICATIONS & USAGE
Uses
For the temporary relief of symptoms associated withdry skin
itchy skin
other minor skin irritationsDirections
Adults: Apply a small amount on the affected area two to three times daily, or as recommended by your healthcare practitioner.May cover affected area with sterile gauze after each application.
Children: Use under the direction of your healthcare practitioner.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
UNDA 270
equisetum arvense, hypericum perforatum creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62106-0097 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYPERICUM PERFORATUM (UNII: XK4IUX8MNB) (HYPERICUM PERFORATUM - UNII:XK4IUX8MNB) HYPERICUM PERFORATUM 1 [hp_X] in 40 g EQUISETUM ARVENSE TOP (UNII: 1DP6Y6B65Z) (EQUISETUM ARVENSE TOP - UNII:1DP6Y6B65Z) EQUISETUM ARVENSE TOP 1 [hp_X] in 40 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) LANOLIN (UNII: 7EV65EAW6H) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62106-0097-0 1 in 1 CARTON 06/01/2015 1 40 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 06/01/2015 Labeler - Seroyal USA (018361118) Establishment Name Address ID/FEI Business Operations SAN’UP 401010287 manufacture(62106-0097)