Label: BLISTEX CON MEDICACION CREMA LABIAL (camphor- synthetic and phenol stick
-
Contains inactivated NDC Code(s)
NDC Code(s): 10157-9926-1 - Packager: Blistex Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Export only
Drug Label Information
Updated June 23, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
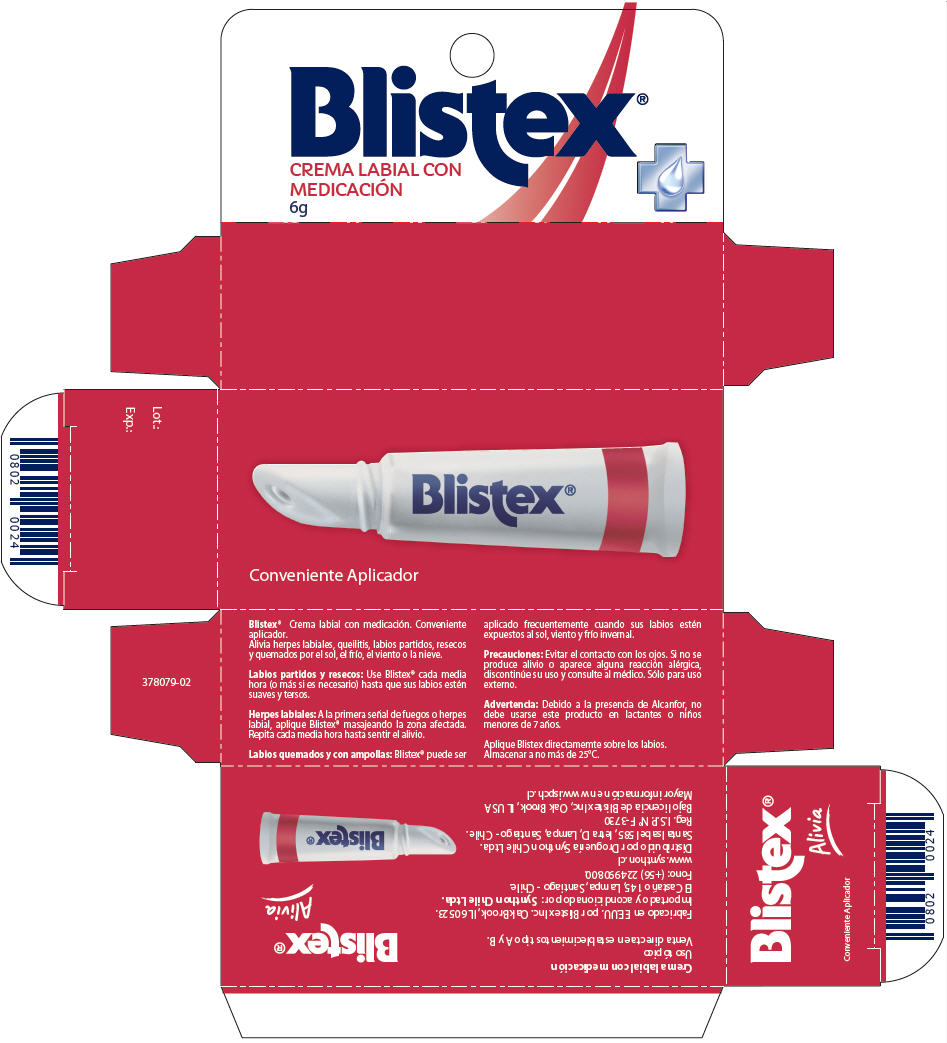
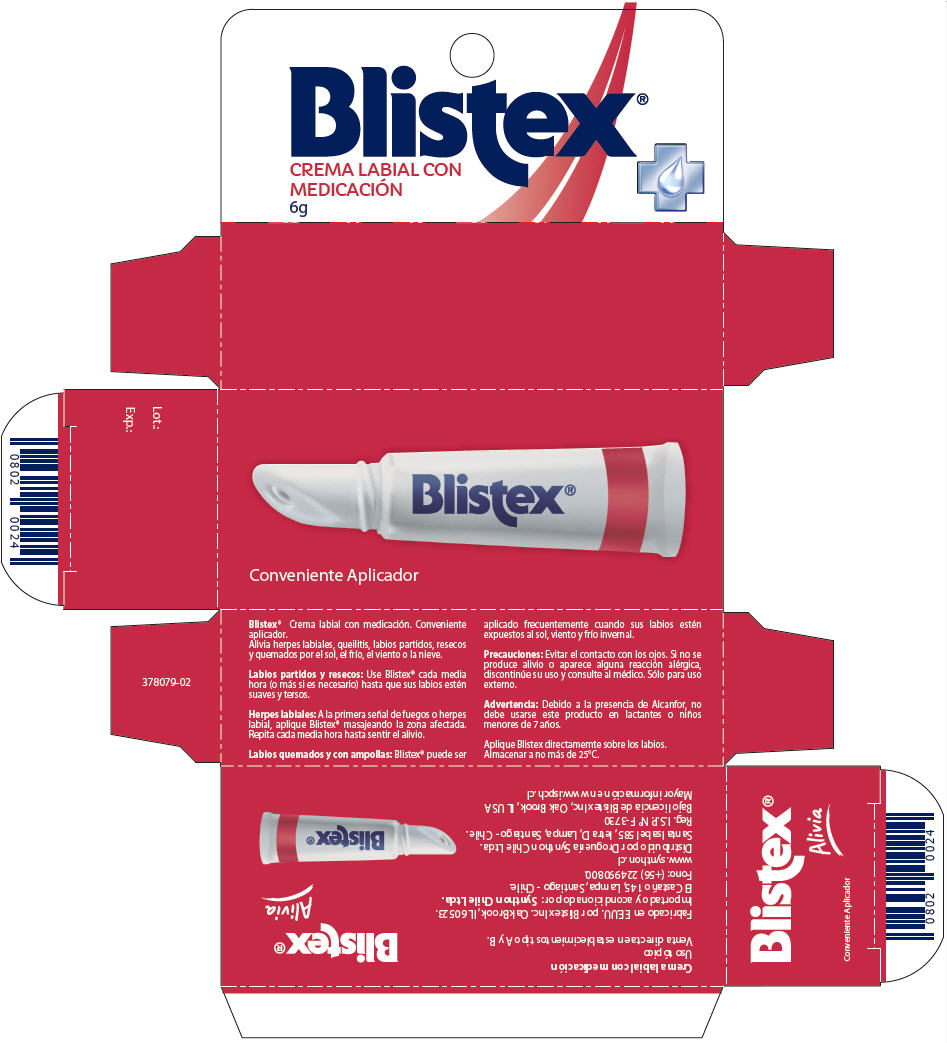
- PRINCIPAL DISPLAY PANEL - 6g Tube Carton
-
INGREDIENTS AND APPEARANCE
BLISTEX CON MEDICACION CREMA LABIAL
camphor (synthetic) and phenol stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10157-9926 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 1 g in 100 g PHENOL (UNII: 339NCG44TV) (PHENOL - UNII:339NCG44TV) PHENOL 0.45 g in 100 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) WATER (UNII: 059QF0KO0R) YELLOW WAX (UNII: 2ZA36H0S2V) PARAFFIN (UNII: I9O0E3H2ZE) PETROLATUM (UNII: 4T6H12BN9U) LANOLIN (UNII: 7EV65EAW6H) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) ALCOHOL (UNII: 3K9958V90M) GLYCERIN (UNII: PDC6A3C0OX) CALCIUM STEAROYL LACTYLATE (UNII: 30MXH4012A) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) METHYL SALICYLATE (UNII: LAV5U5022Y) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) LEVOMENTHOL (UNII: BZ1R15MTK7) CETYL ALCOHOL (UNII: 936JST6JCN) AMMONIA (UNII: 5138Q19F1X) CALCIUM HYDROXIDE (UNII: PF5DZW74VN) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10157-9926-1 6 g in 1 TUBE; Type 0: Not a Combination Product 10/06/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date EXPORT ONLY 10/06/2011 Labeler - Blistex Inc. (005126354) Establishment Name Address ID/FEI Business Operations Blistex Inc. 005126354 MANUFACTURE(10157-9926)