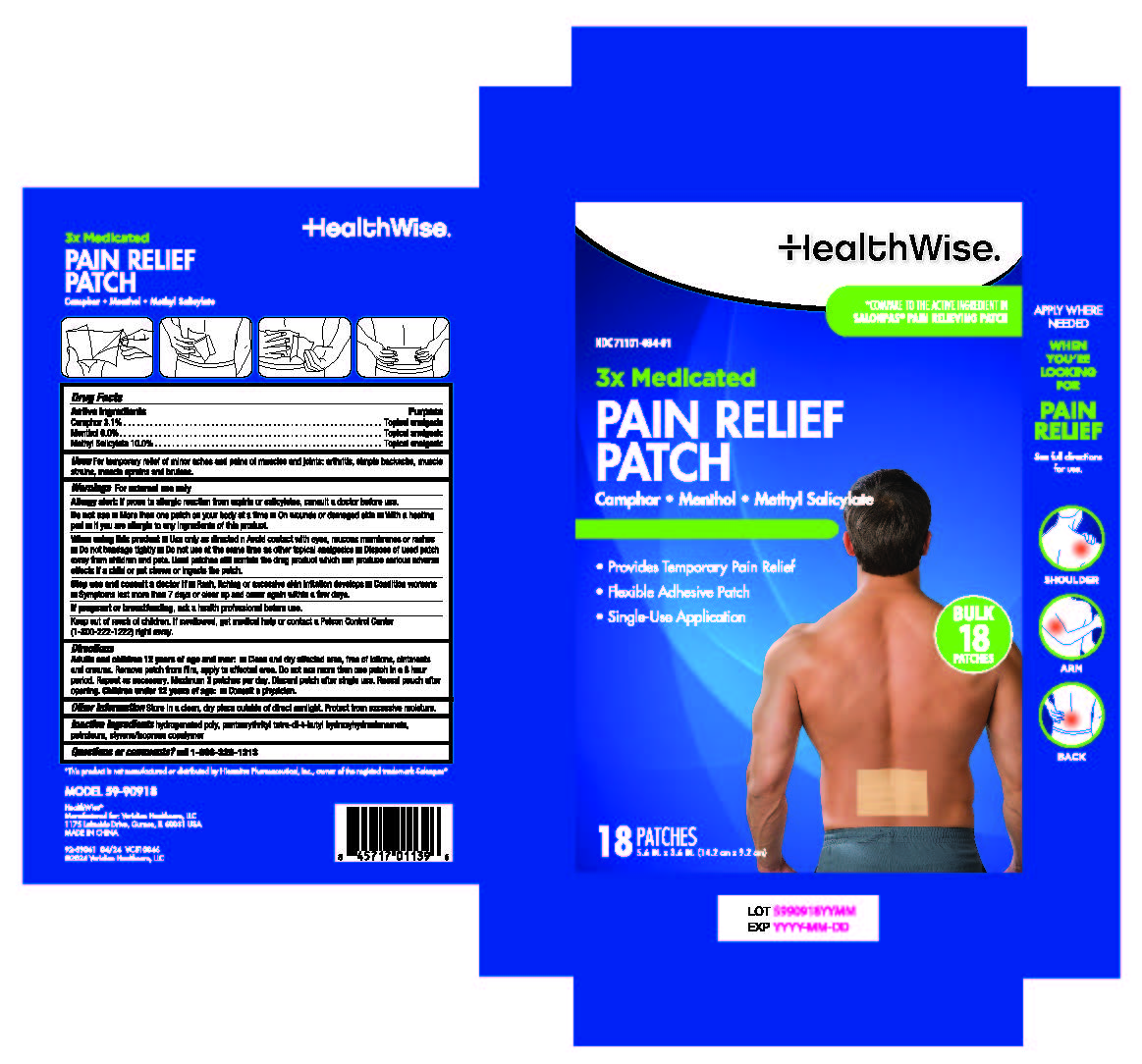
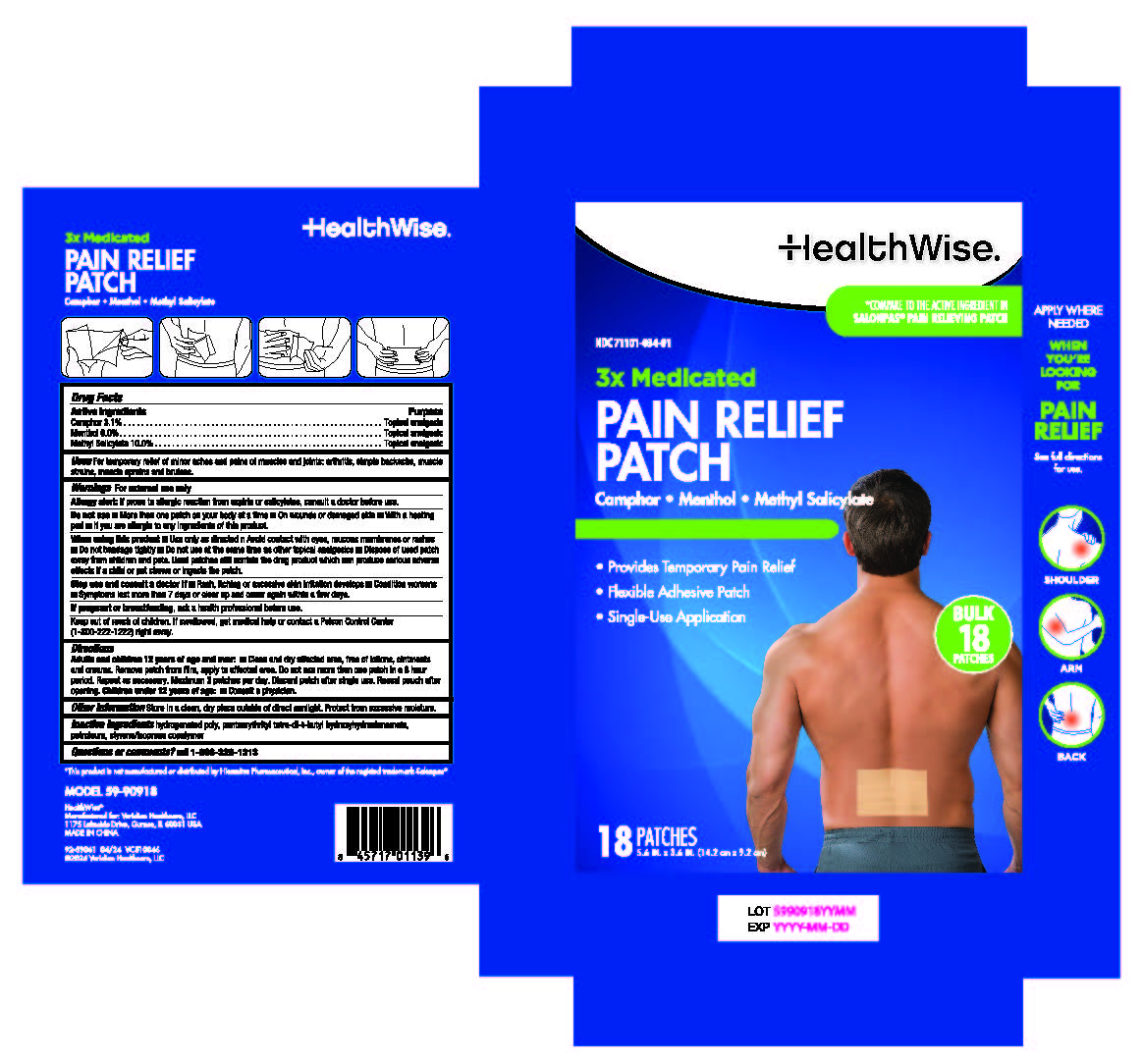
Label: HEALTHWISE 3X MEDICATED PAIN RELIEF PATCH- camphor,menthol, methyl salicylate patch
- NDC Code(s): 71101-034-01
- Packager: Veridian Healthcare
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 17, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purposes
- Use
-
Warnings
For external use only
Allergy alert: If prone to allergic reaction from aspirin or salicylates, consult a doctor before use.
Do not use
■More than one patch on your body at a time
■ On wounds or damaged skin
■ With a heating pad
■If you are allergic to any ingredients of this product.
When using this product
■Use only as directed and avoid contact with eyes, mucous membranes or rashes
■ Do not bandage tightly
■ Do not use at the same time as other topical analgesics
■ Dispose of used patch away from children and pets. Used patches still contain the drug product which can produce serious adverse effects if a child or pet chews or ingests the patch.
-
Directions
Adults and children 12 years of age and over:
■ Clean and dry affected area, free of lotions, ointments and creams. Remove patch from film, apply to affected area.Do not use more than one patch in a 8 hour period. Repeat as necessary. Maximum 3 patches per day. Discard patch after single use. Reseal pouch after opening.
Children under 12 years of age: ■ Consult a physician.
- Other information
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
HEALTHWISE 3X MEDICATED PAIN RELIEF PATCH
camphor,menthol, methyl salicylate patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71101-034 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYL SALICYLATE (UNII: LAV5U5022Y) (SALICYLIC ACID - UNII:O414PZ4LPZ) METHYL SALICYLATE 10 g CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 3.1 g LEVOMENTHOL (UNII: BZ1R15MTK7) (LEVOMENTHOL - UNII:BZ1R15MTK7) LEVOMENTHOL 6 g Inactive Ingredients Ingredient Name Strength PENTAERYTHRITOL TETRAKIS(3-(3,5-DI-TERT-BUTYL-4-HYDROXYPHENYL)PROPIONATE) (UNII: 255PIF62MS) HYDROGENATED POLYDECENE TYPE I (UNII: U333RI6EB7) STYRENE/ACRYLAMIDE COPOLYMER (MW 500000) (UNII: 5Z4DPO246A) LIQUID PETROLEUM (UNII: 6ZAE7X688J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71101-034-01 18 in 1 BOX 04/01/2024 1 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 04/01/2024 Labeler - Veridian Healthcare (830437997) Establishment Name Address ID/FEI Business Operations Foshan Aqua Gel Biotech Co., Ltd., 529128763 manufacture(71101-034)