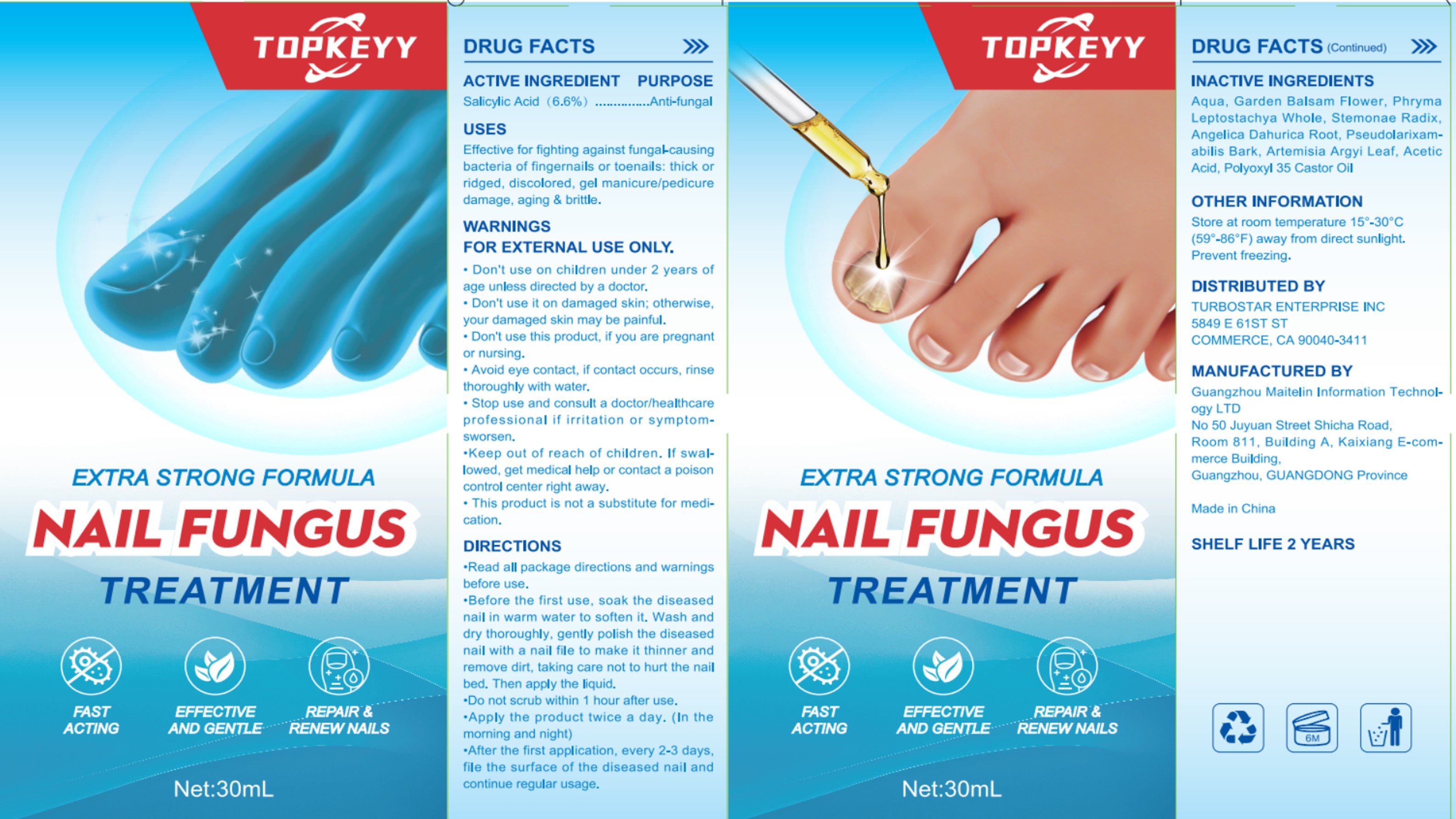
Label: NAIL FUNGUS TREATMENT- salicylic acid liquid
- NDC Code(s): 84257-101-01, 84257-101-02, 84257-101-03
- Packager: Guangzhou Maitelin Information Technology Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Read all package directions and warningsbefore use.
Before the first use, soak the diseasednail in warm water to soften it. Wash anddry thoroughly, gently polish the diseasednail with a nail file to make it thinner andremove dirt, taking care not to hurt the nailbed. Then apply the liquid.
Do not scrub within 1 hour after use.
Apply the product twice a day. (In themorning and night)
After the first application, every 2-3 daysfile the surface of the diseased nail andcontinue regular usage. - STORAGE AND HANDLING
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NAIL FUNGUS TREATMENT
salicylic acid liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84257-101 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 6.6 g in 100 mL Inactive Ingredients Ingredient Name Strength PHRYMA LEPTOSTACHYA WHOLE (UNII: 6G287VJM69) ANGELICA DAHURICA ROOT (UNII: 1V63N2S972) PSEUDOLARIX AMABILIS BARK (UNII: 49G13A93VE) WATER (UNII: 059QF0KO0R) STEMONA SESSILIFOLIA ROOT (UNII: EKG8030U4N) IMPATIENS BALSAMINA FLOWER (UNII: 3169PS5TJH) ARTEMISIA ARGYI LEAF (UNII: 2JYC99Q0WZ) ACETIC ACID (UNII: Q40Q9N063P) POLYOXYL 35 CASTOR OIL (UNII: 6D4M1DAL6O) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84257-101-01 20 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/11/2024 2 NDC:84257-101-02 25 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/11/2024 3 NDC:84257-101-03 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/11/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 04/11/2024 Labeler - Guangzhou Maitelin Information Technology Co., Ltd. (411899196) Establishment Name Address ID/FEI Business Operations Guangzhou Maitelin Information Technology Co., Ltd. 411899196 manufacture(84257-101)