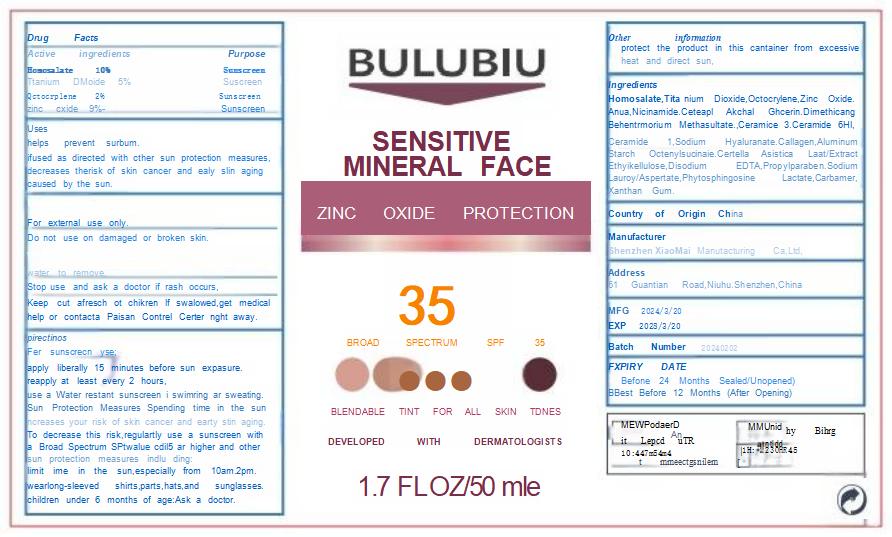
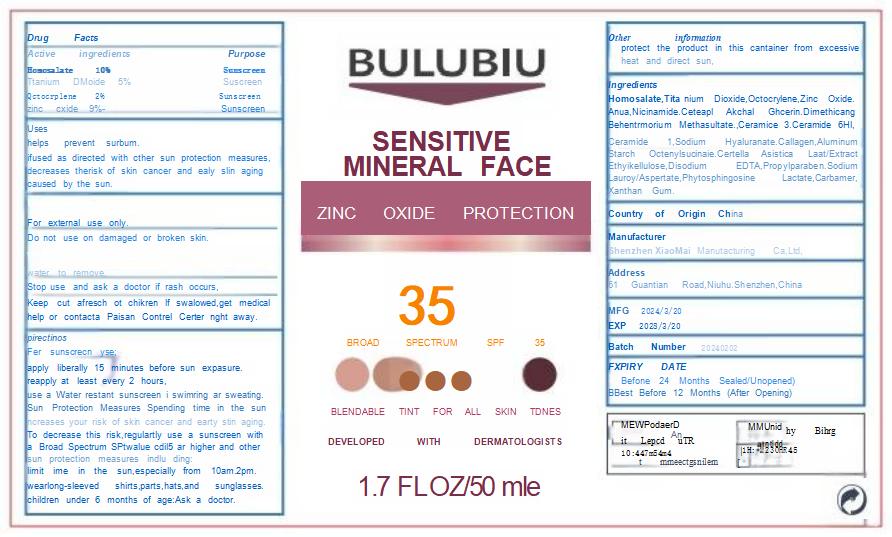
Label: BBULUBIU SENSITIVE MINERAL FACE ZINC OXIDE PROTECTION 35ULUBIU SENSITIVE MINERAL FACE ZINC OXIDE PROTECTION 35- sensitive mineral face zinc oxide protection 35 cream
- NDC Code(s): 83872-101-01
- Packager: Shenzhen XiaoMai Manufacturing Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- Uses
- WARNINGS
- Do not use
- When using this product
- Stop use and ask a doctor
- Keep out of reach of children.
- Directions for use
-
INACTIVE INGREDIENT
Cetearyl Alcohol: 4.5%
Ethylcellulose: 1%
Carbomer: 0.2%
Disodium EDTA: 0.6%
Glycerin: 5.5%
Dimethicone: 3.5%
Behentrimonium Methosulfate: 1.5%
Aluminum Starch Octenylsuccinate: 2%
Propylparaben: 0.6%
Sodium Lauroyl Aspartate: 0.3%
Phytosphingosine: 0.2%
Lactic Acid: 0.2%
Xanthan Gum: 0.2%
Aqua: 42.7% - Other Information
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BBULUBIU SENSITIVE MINERAL FACE ZINC OXIDE PROTECTION 35ULUBIU SENSITIVE MINERAL FACE ZINC OXIDE PROTECTION 35
sensitive mineral face zinc oxide protection 35 creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83872-101 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 20 mg in 1 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 50 mg in 1 g NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 40 mg in 1 g CERAMIDE 1 (UNII: 5THT33P7X7) (CERAMIDE 1 - UNII:5THT33P7X7) CERAMIDE 1 5 mg in 1 g CERAMIDE 3 (UNII: 4370DF050B) (CERAMIDE 3 - UNII:4370DF050B) CERAMIDE 3 5 mg in 1 g HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 100 mg in 1 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 90 mg in 1 g CERAMIDE 6 II (UNII: F1X8L2B00J) (CERAMIDE 6 II - UNII:F1X8L2B00J) CERAMIDE 6 II 5 mg in 1 g Inactive Ingredients Ingredient Name Strength BEHENTRIMONIUM METHOSULFATE (UNII: 5SHP745C61) 15 mg in 1 g GLYCERIN (UNII: PDC6A3C0OX) 55 mg in 1 g XANTHAN GUM (UNII: TTV12P4NEE) 2 mg in 1 g DIMETHICONE (UNII: 92RU3N3Y1O) 35 mg in 1 g ALUMINUM STARCH OCTENYLSUCCINATE (UNII: I9PJ0O6294) 20 mg in 1 g PROPYLPARABEN (UNII: Z8IX2SC1OH) 6 mg in 1 g WATER (UNII: 059QF0KO0R) 427 mg in 1 g SODIUM LAUROYL ASPARTATE (UNII: 328G13N8ZR) 3 mg in 1 g LACTIC ACID (UNII: 33X04XA5AT) 2 mg in 1 g PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) 2 mg in 1 g Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83872-101-01 50 g in 1 BOTTLE; Type 0: Not a Combination Product 04/10/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 04/10/2024 Labeler - Shenzhen XiaoMai Manufacturing Co., Ltd. (712999147) Establishment Name Address ID/FEI Business Operations Shenzhen XiaoMai Manufacturing Co., Ltd. 712999147 manufacture(83872-101)