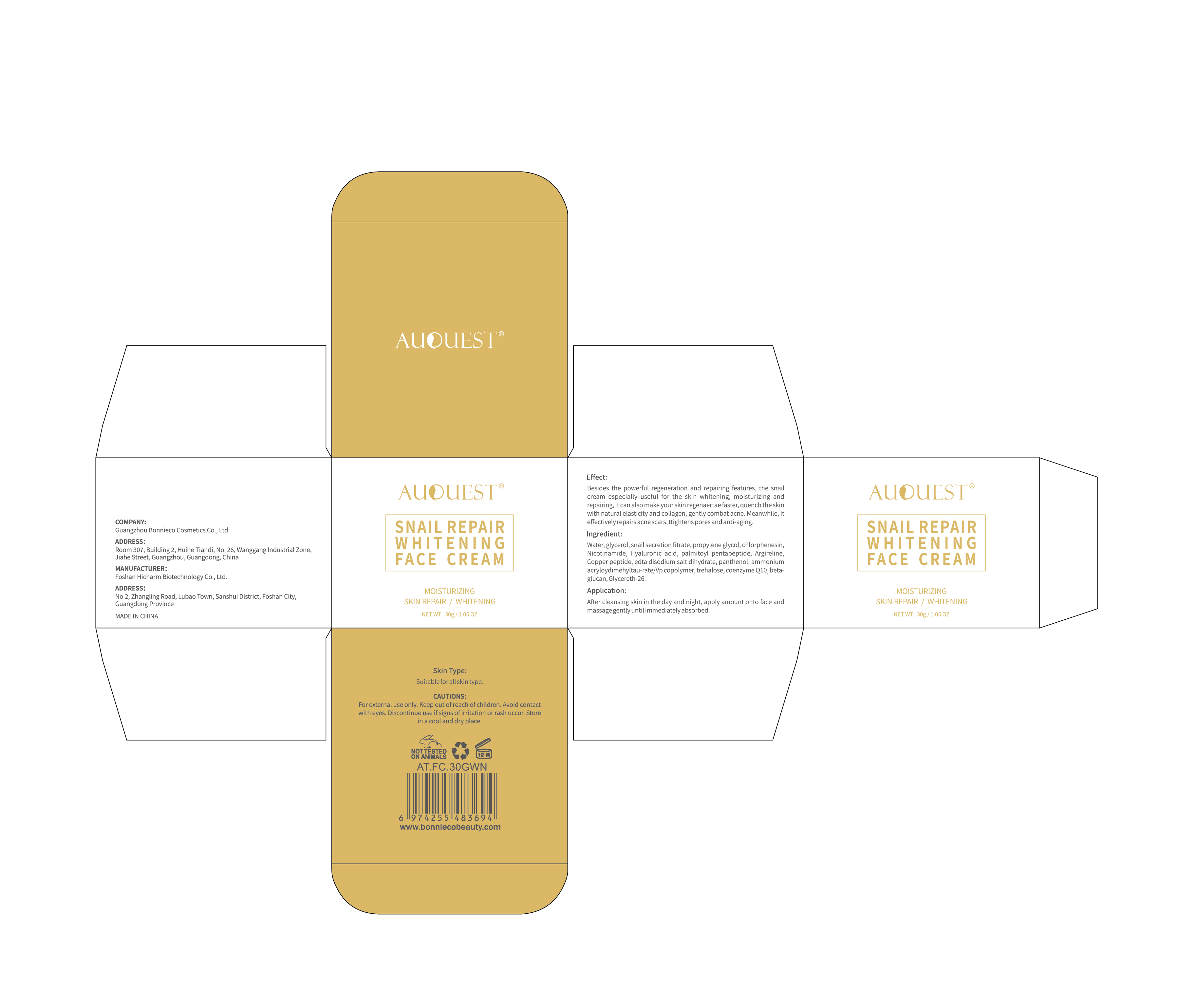
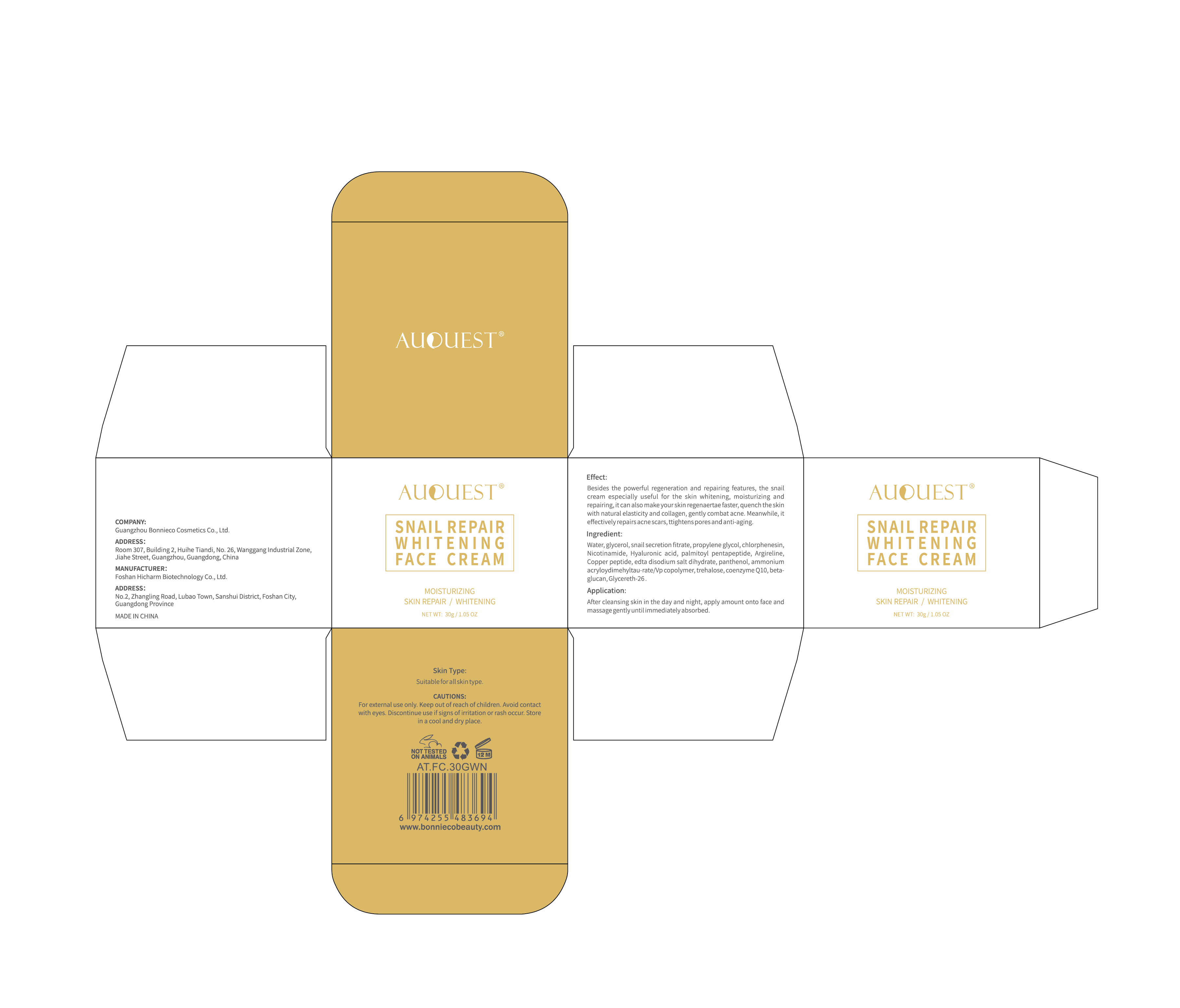
Label: AUQUEST SNAIL REPAIR WHITENING FACE CREAM- snail repair whitening face cream cream
- NDC Code(s): 84186-013-01
- Packager: Guangzhou Bonnieco Cosmetics Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 9, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
-
Do not use
Besides the powerful regeneration and repairing features, the snailcream especially useful for the skin whitening, moisturizing andrepairing,it can also make yourskin regenaertae faster, quench the skinwith natural elasticity and collagen, gently combat acne.Meanwhile, iteffectively repairs acne scars, ttightens pores and anti-aging.
- When Using
- Stop Use
- Ask Doctor
- Keep Oot Of Reach Of Children
- Directions
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
AUQUEST SNAIL REPAIR WHITENING FACE CREAM
snail repair whitening face cream creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84186-013 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 8 g in 100 g Inactive Ingredients Ingredient Name Strength PREZATIDE COPPER (UNII: 6BJQ43T1I9) CHLORPHENESIN (UNII: I670DAL4SZ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) NIACINAMIDE (UNII: 25X51I8RD4) HYALURONIC ACID (UNII: S270N0TRQY) SNAIL, UNSPECIFIED (UNII: 2VIO5GL90I) ACETYL HEXAPEPTIDE-8 (UNII: L4EL31FWIL) AMMONIUM CATION (UNII: 54S68520I4) TREHALOSE (UNII: B8WCK70T7I) TRANSFORMING GROWTH FACTOR BETA RECEPTOR TYPE 3 (UNII: 18YWT2KYS8) GLYCERETH-26 (UNII: NNE56F2N14) PANTHENOL (UNII: WV9CM0O67Z) WATER (UNII: 059QF0KO0R) PALMITOYL PENTAPEPTIDE-4 (UNII: KK181SM5JG) EDETATE DISODIUM (UNII: 7FLD91C86K) UBIDECARENONE (UNII: EJ27X76M46) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84186-013-01 30 g in 1 CANISTER; Type 0: Not a Combination Product 04/09/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M016 04/09/2024 Labeler - Guangzhou Bonnieco Cosmetics Co., Ltd (412244189) Establishment Name Address ID/FEI Business Operations Guangzhou Bonnieco Cosmetics Co., Ltd 412244189 label(84186-013) , manufacture(84186-013)