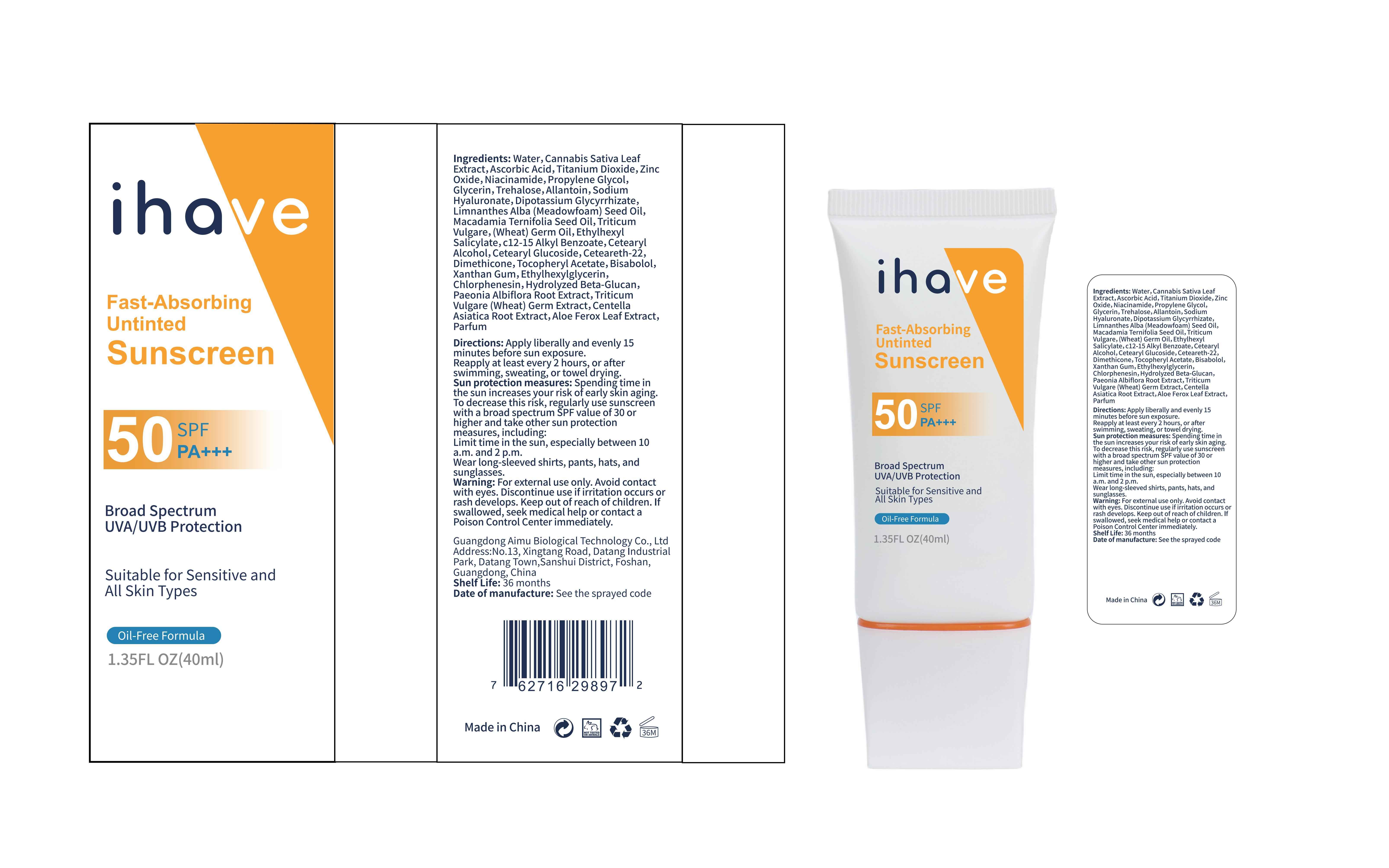
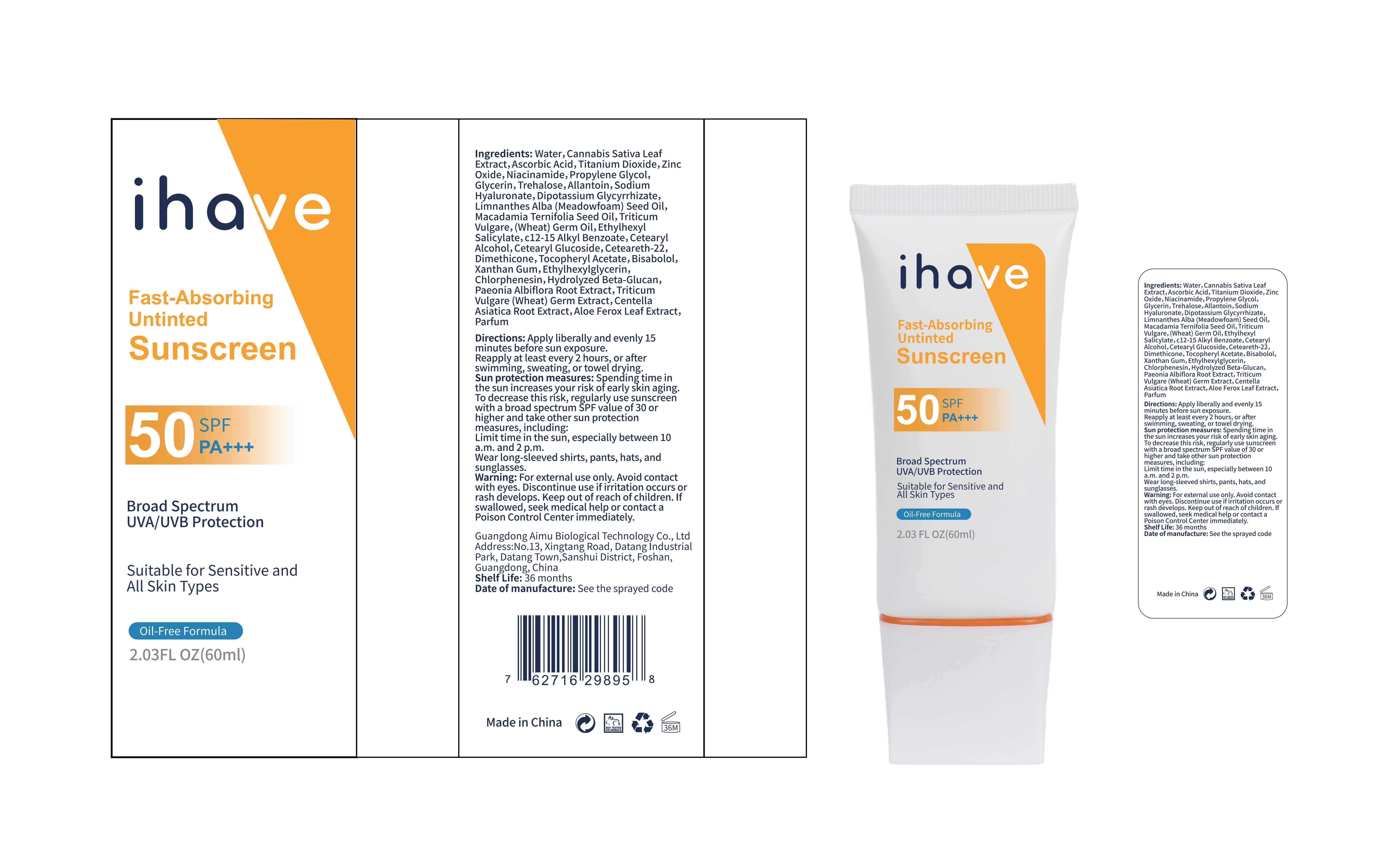
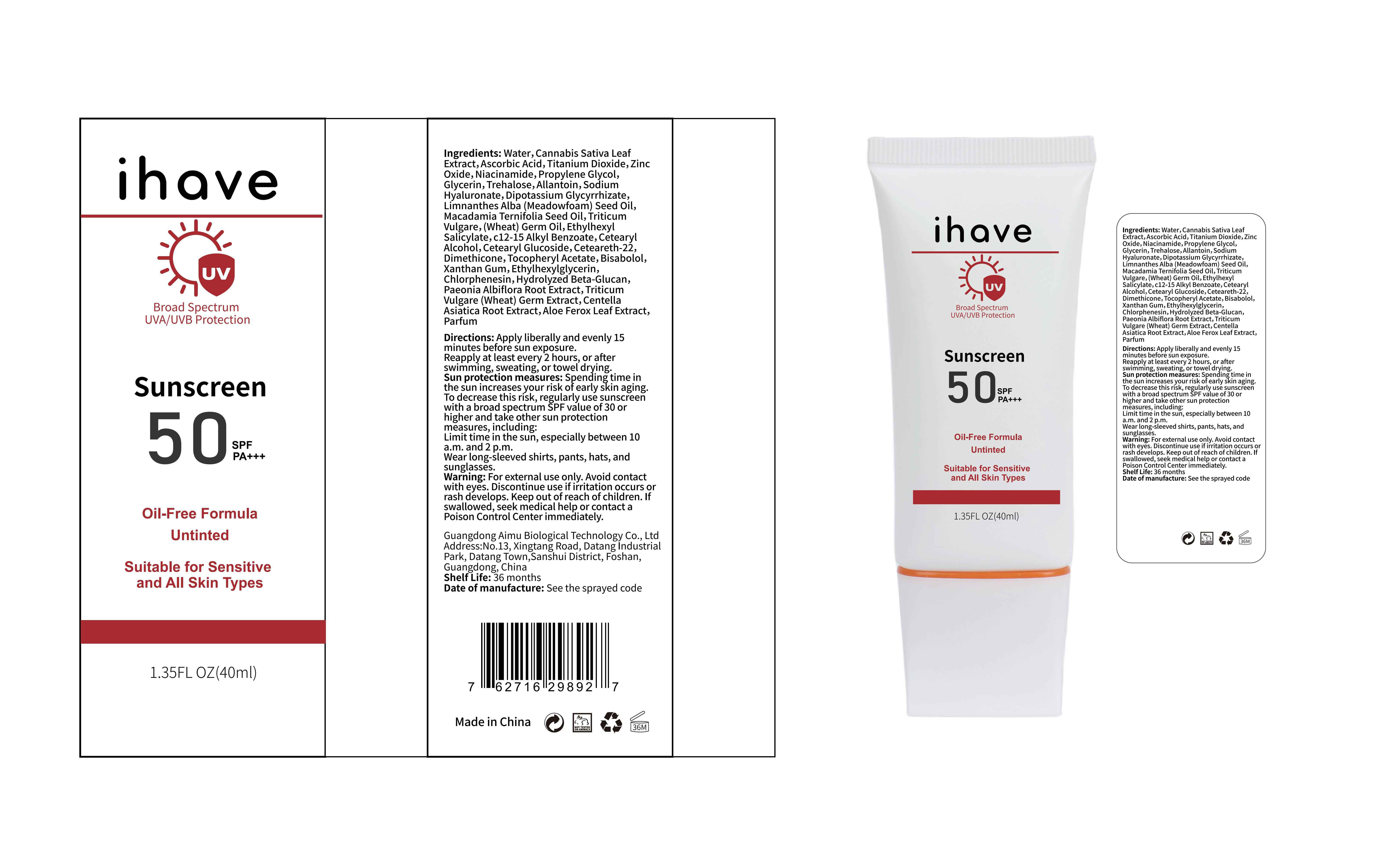
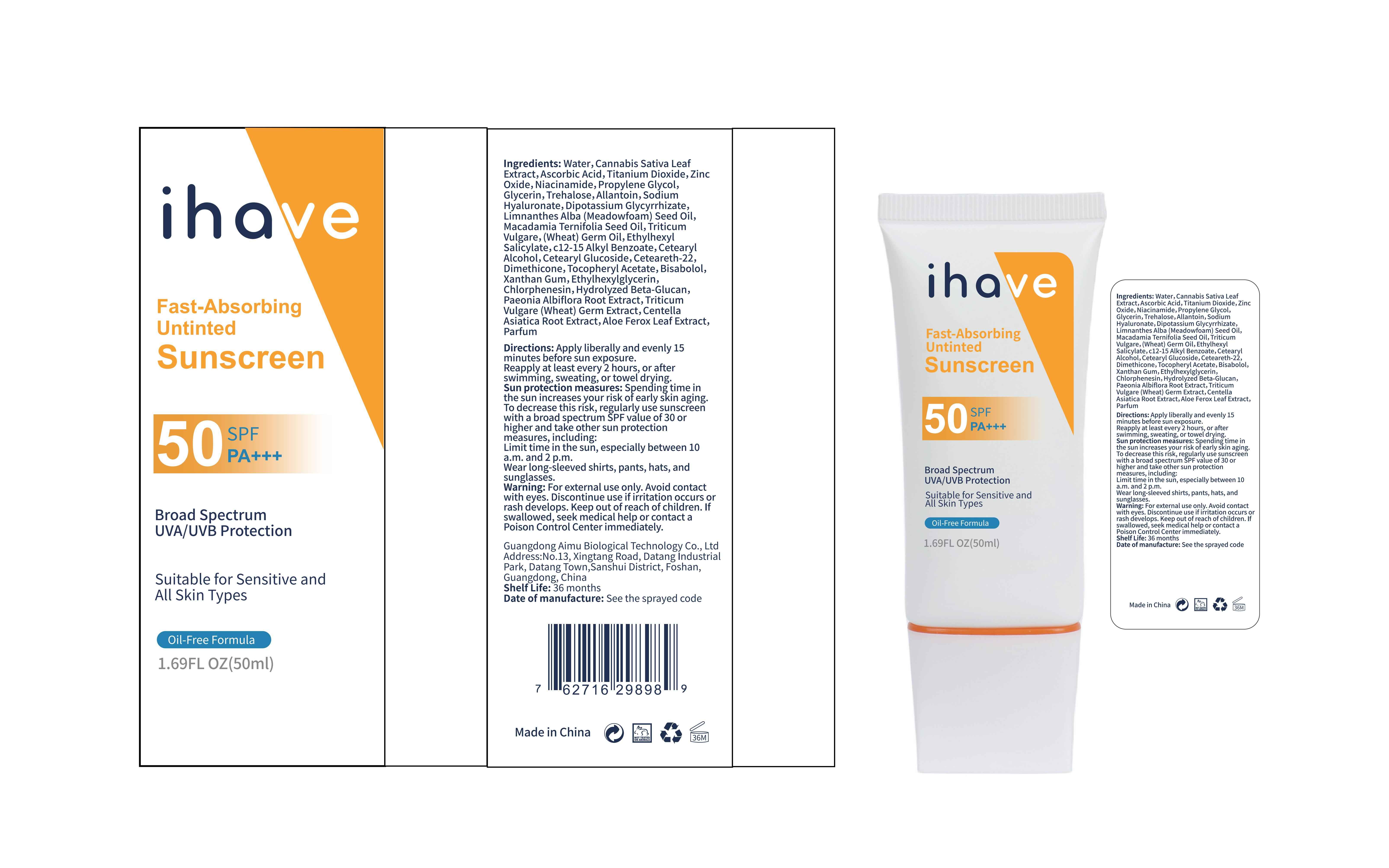
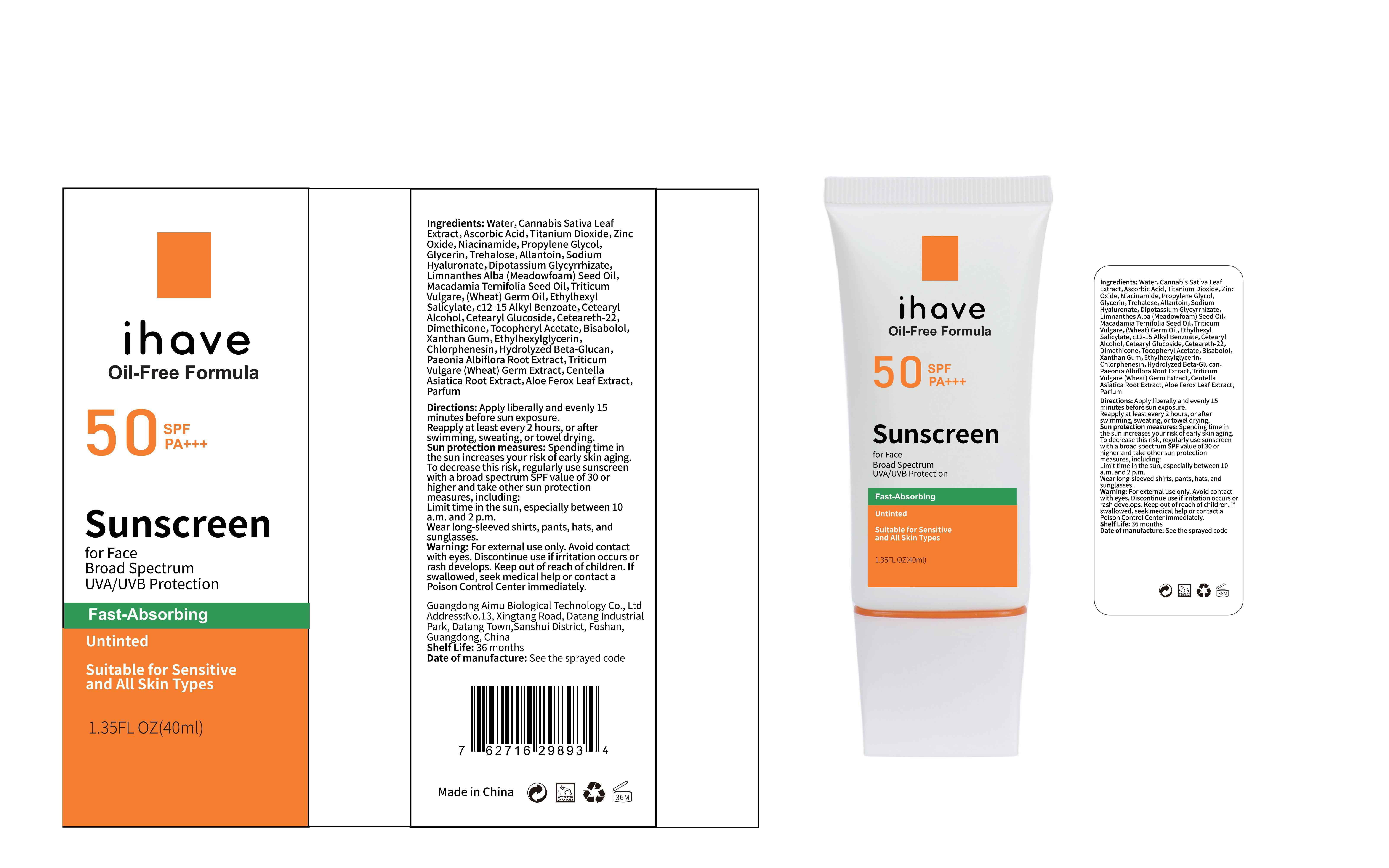
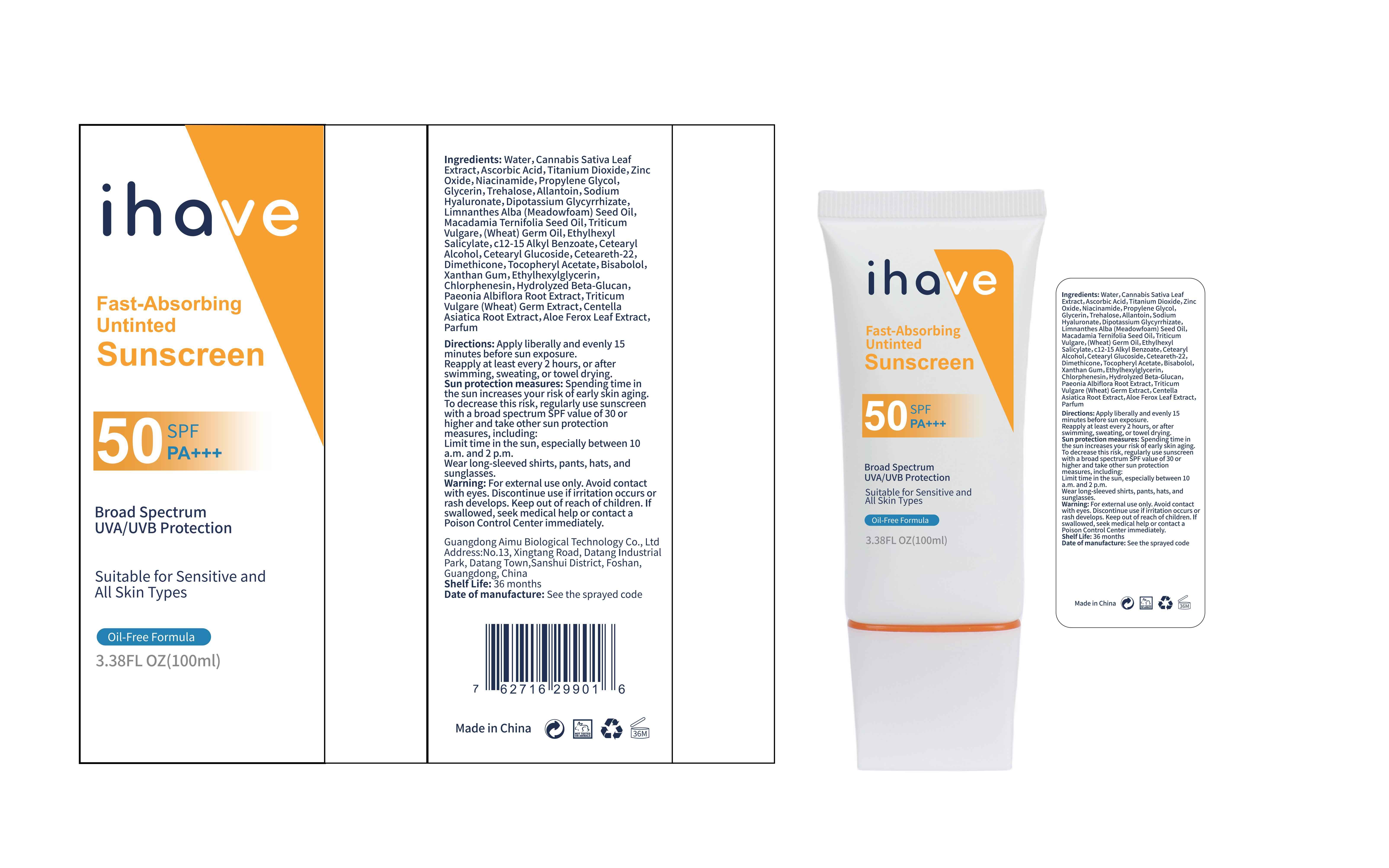
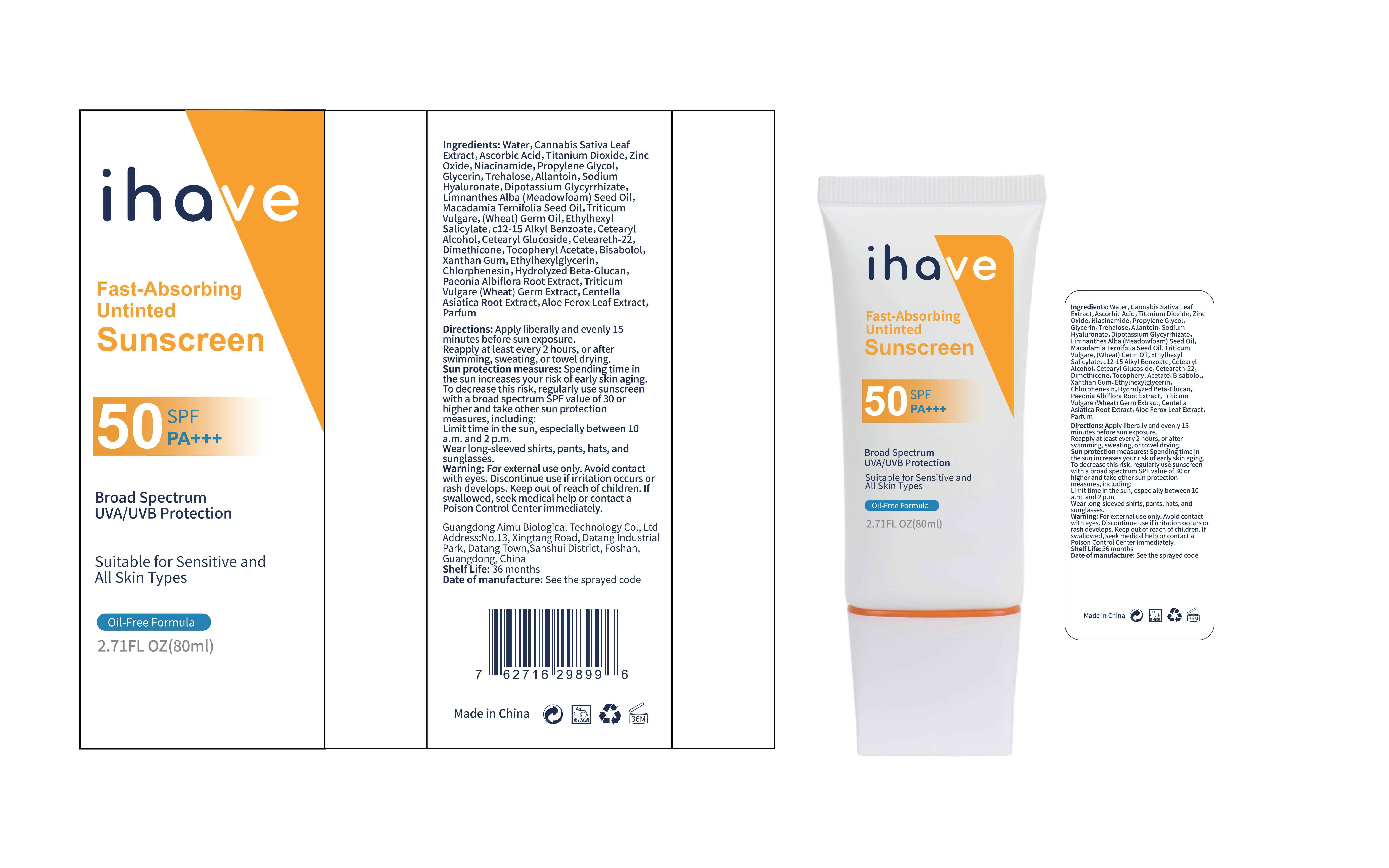
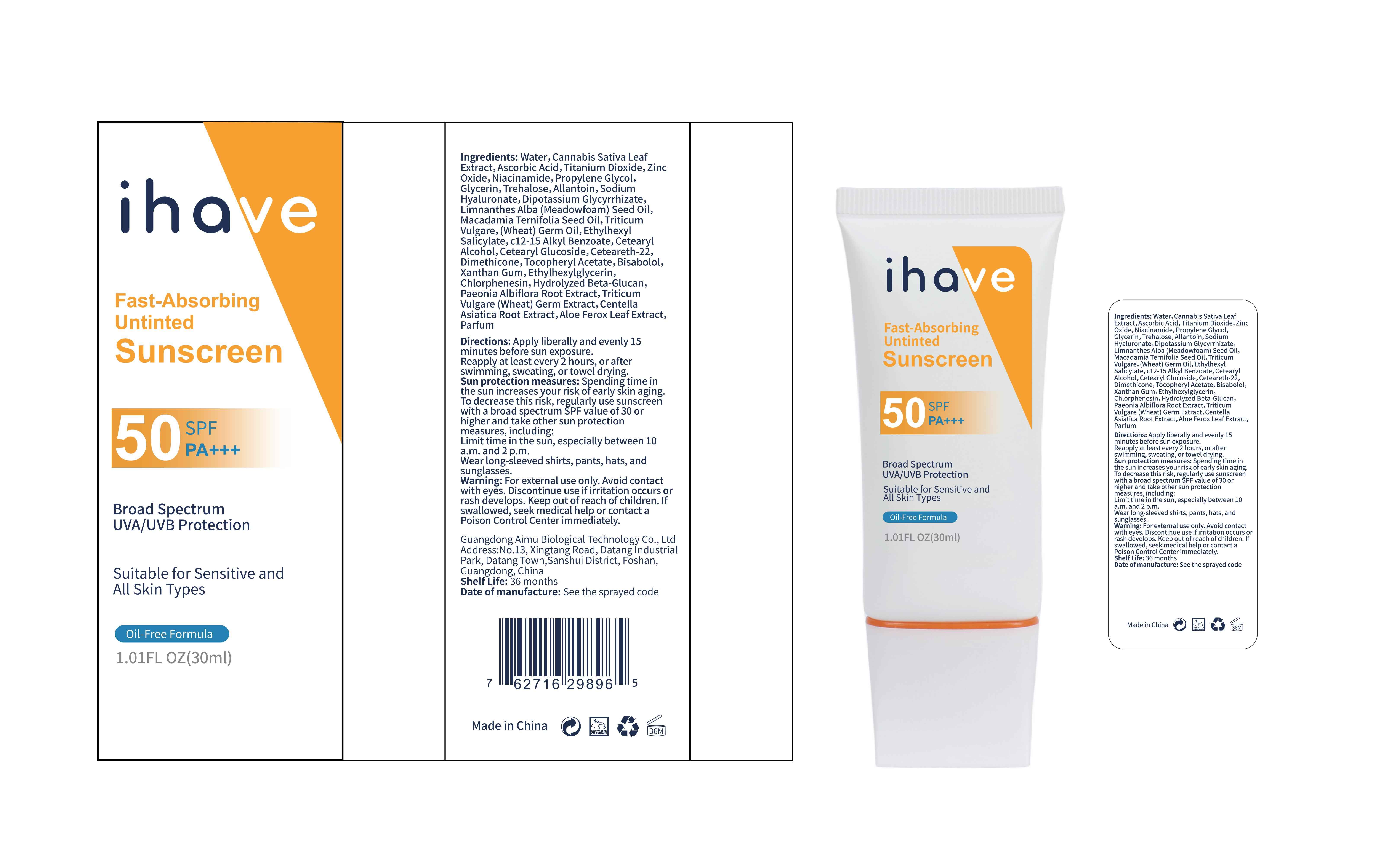
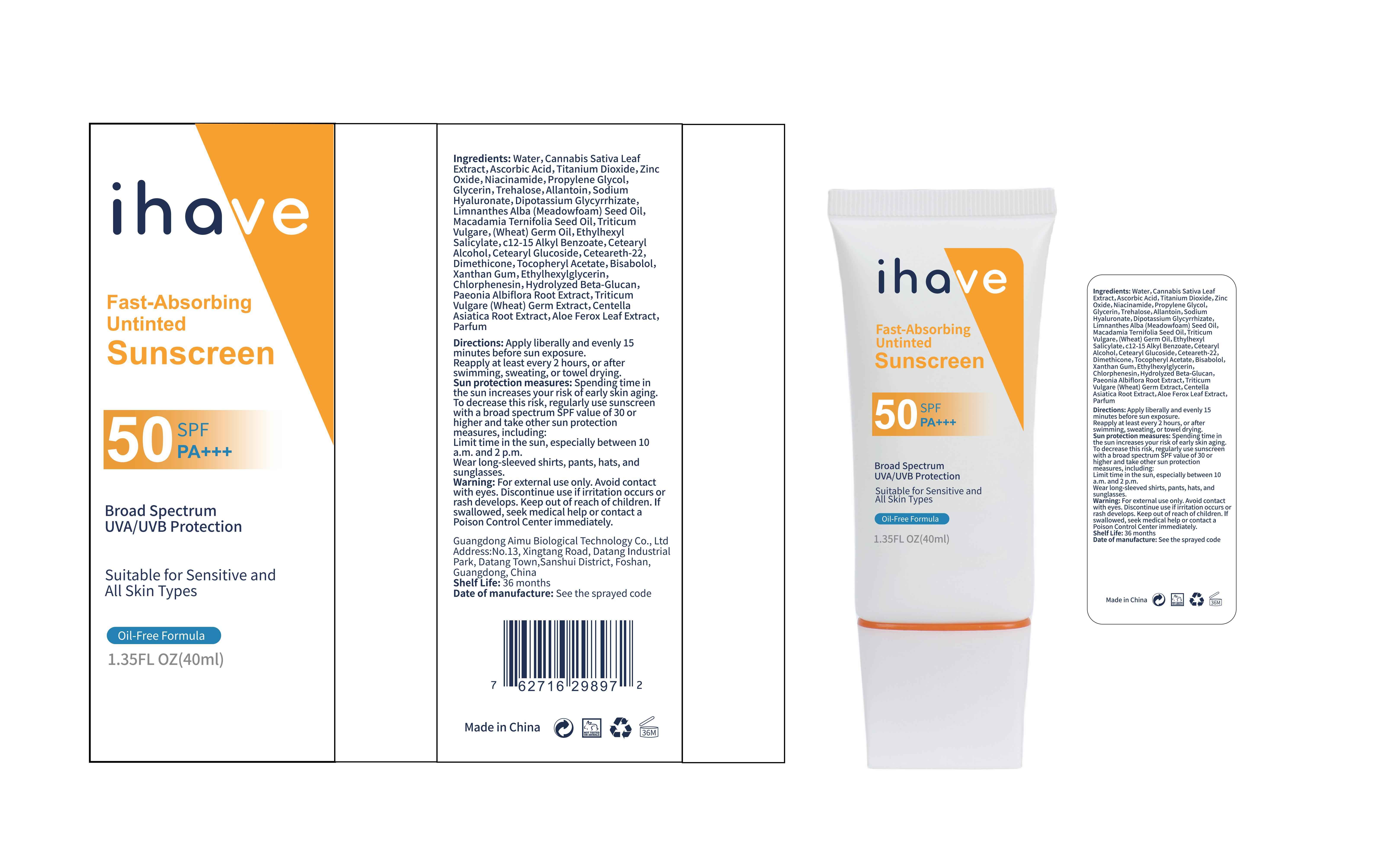
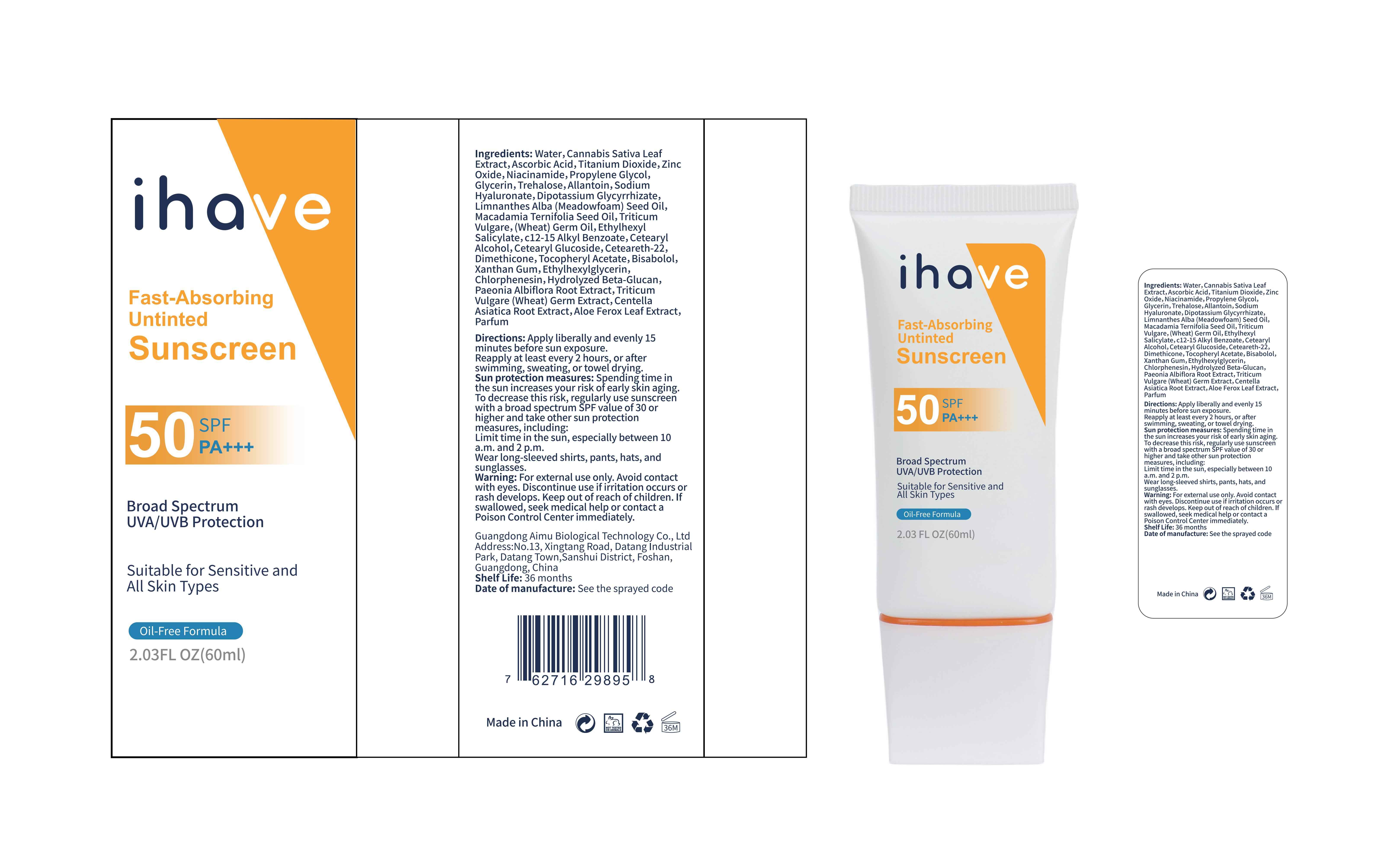
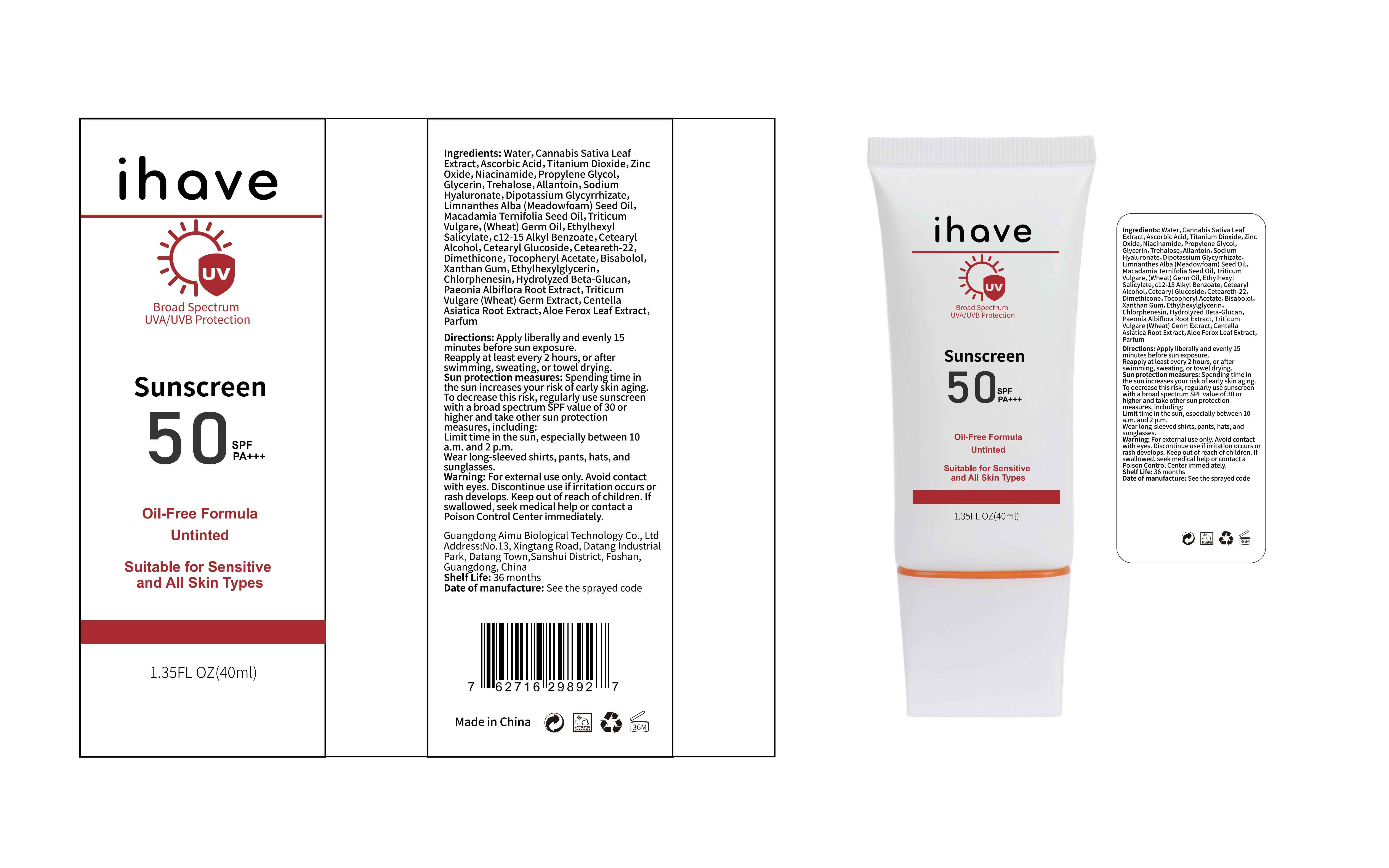
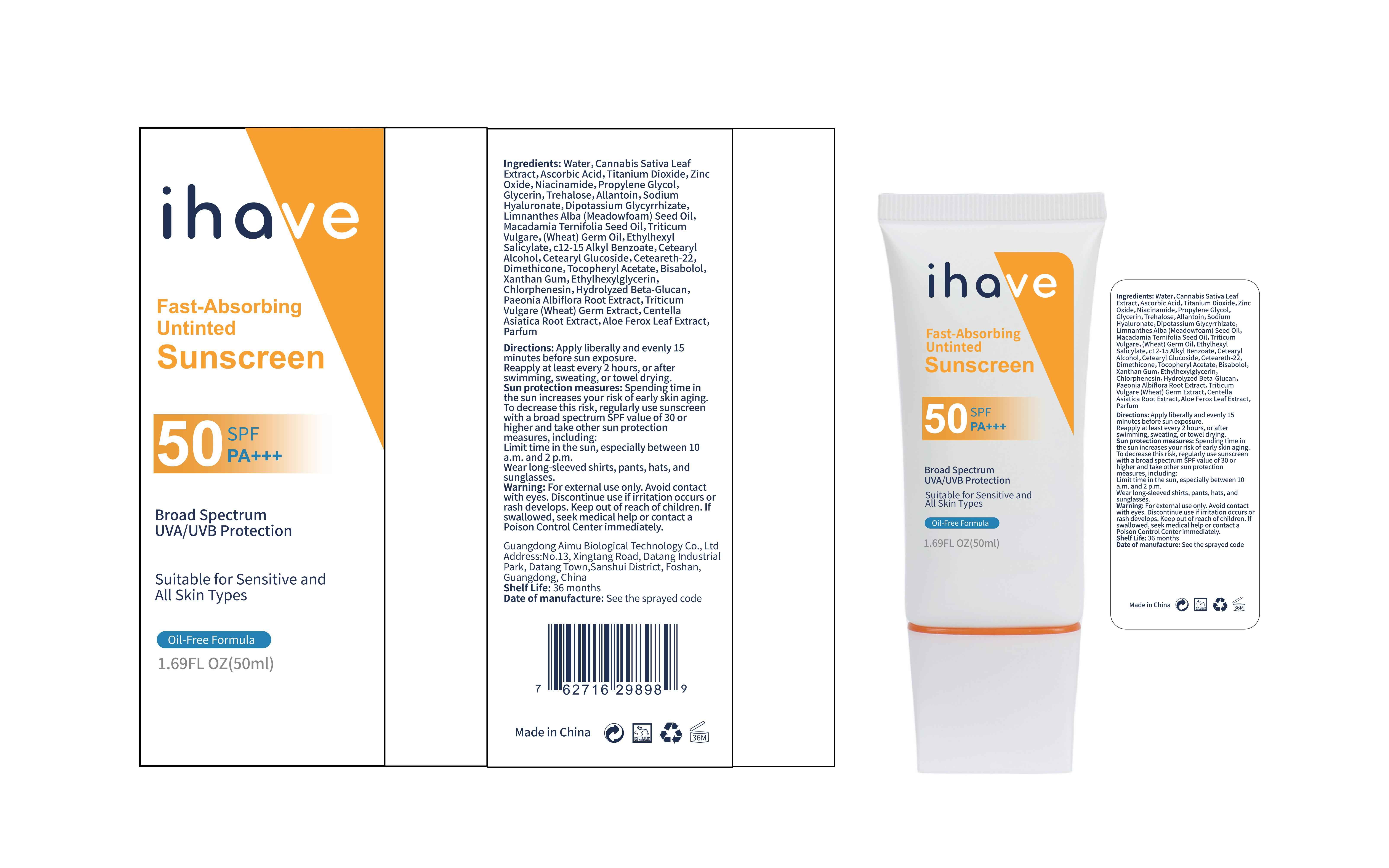
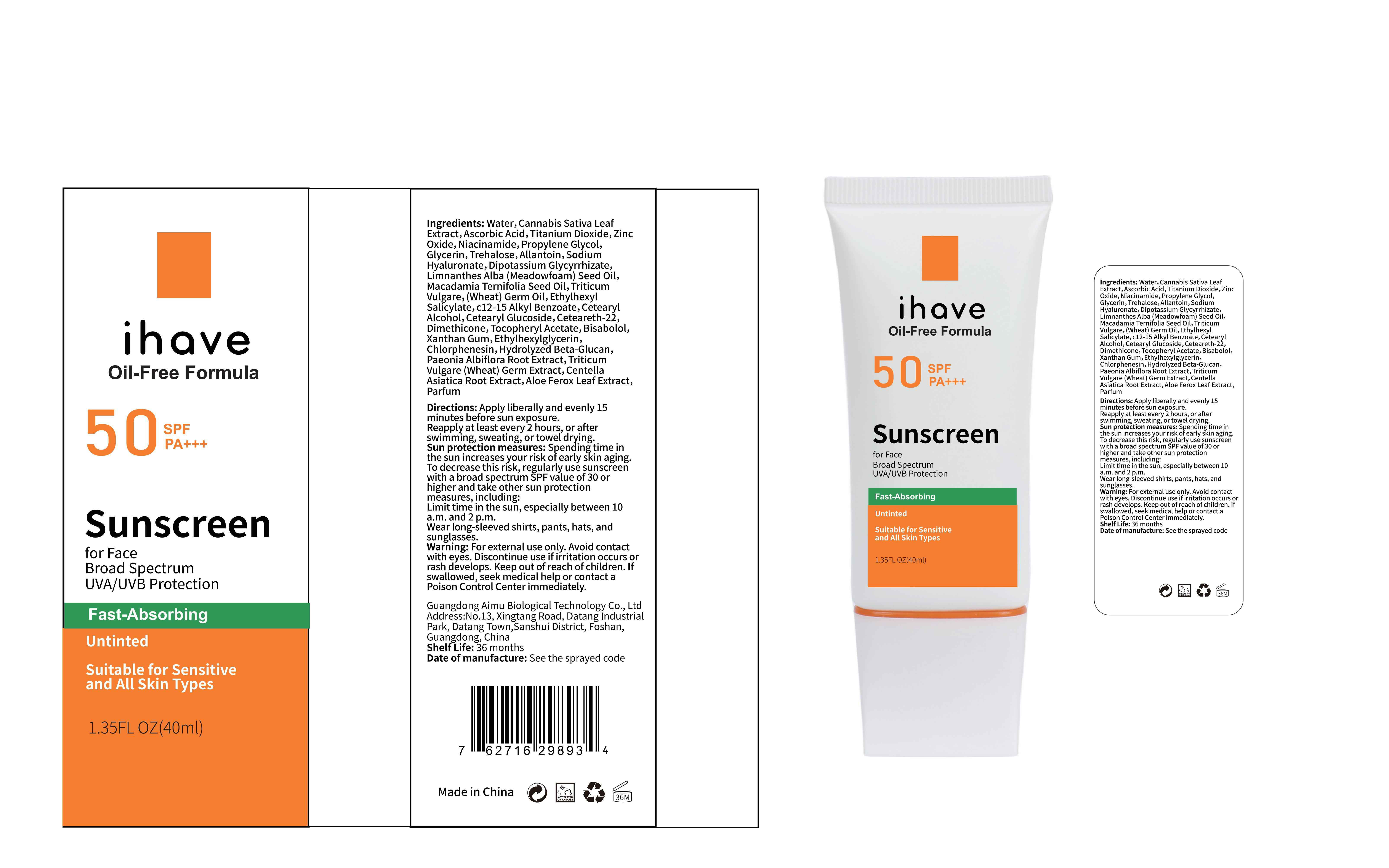
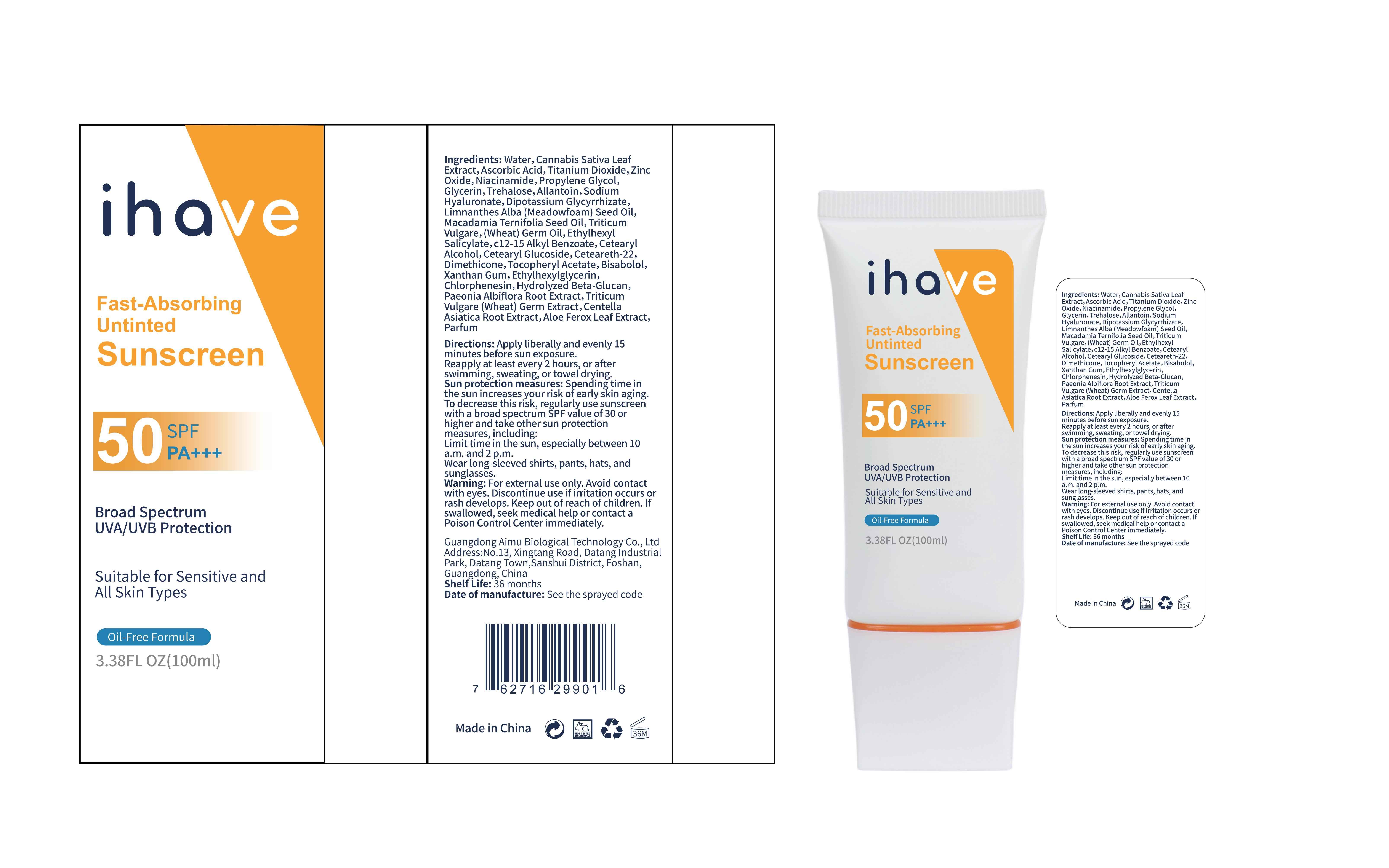
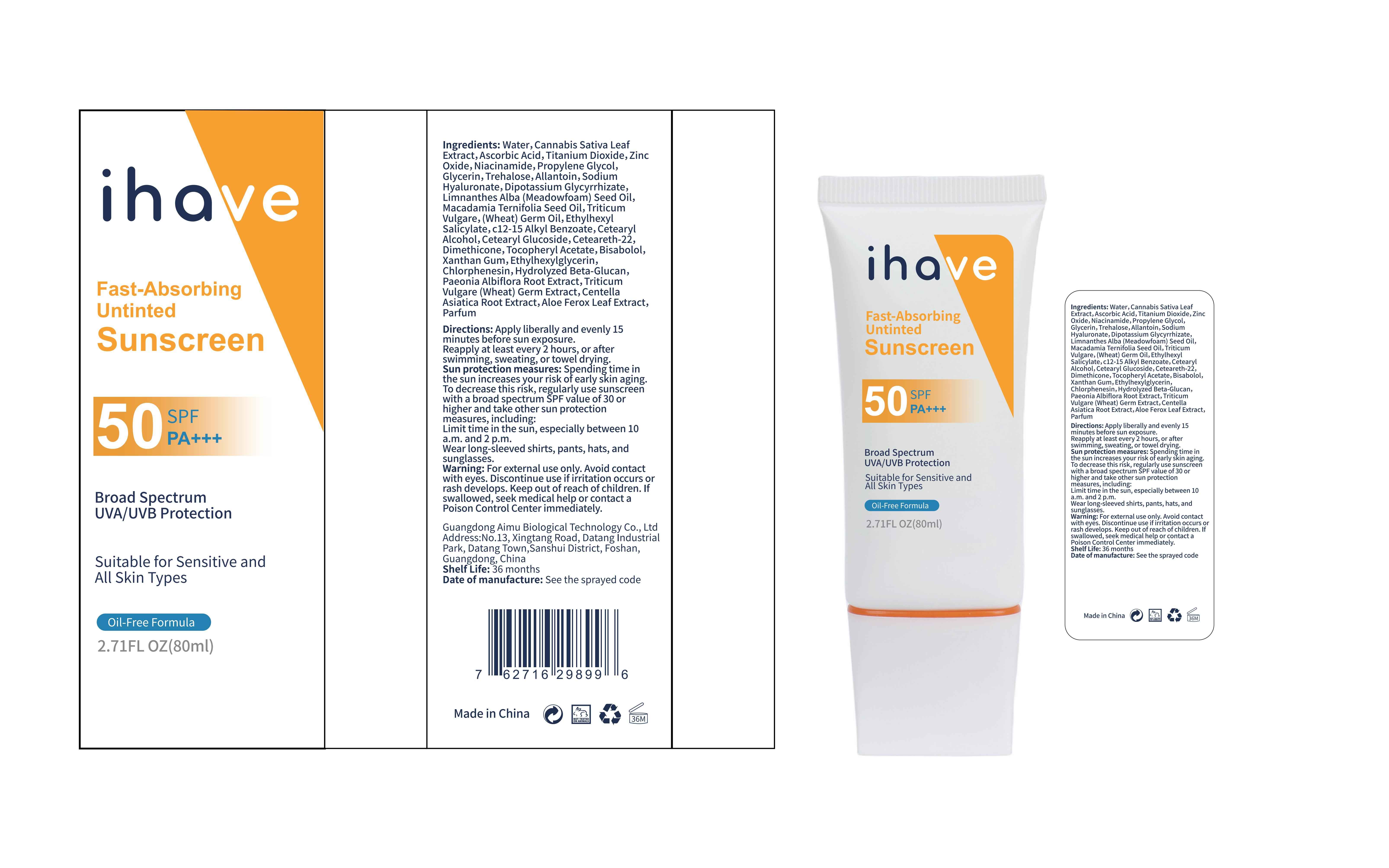
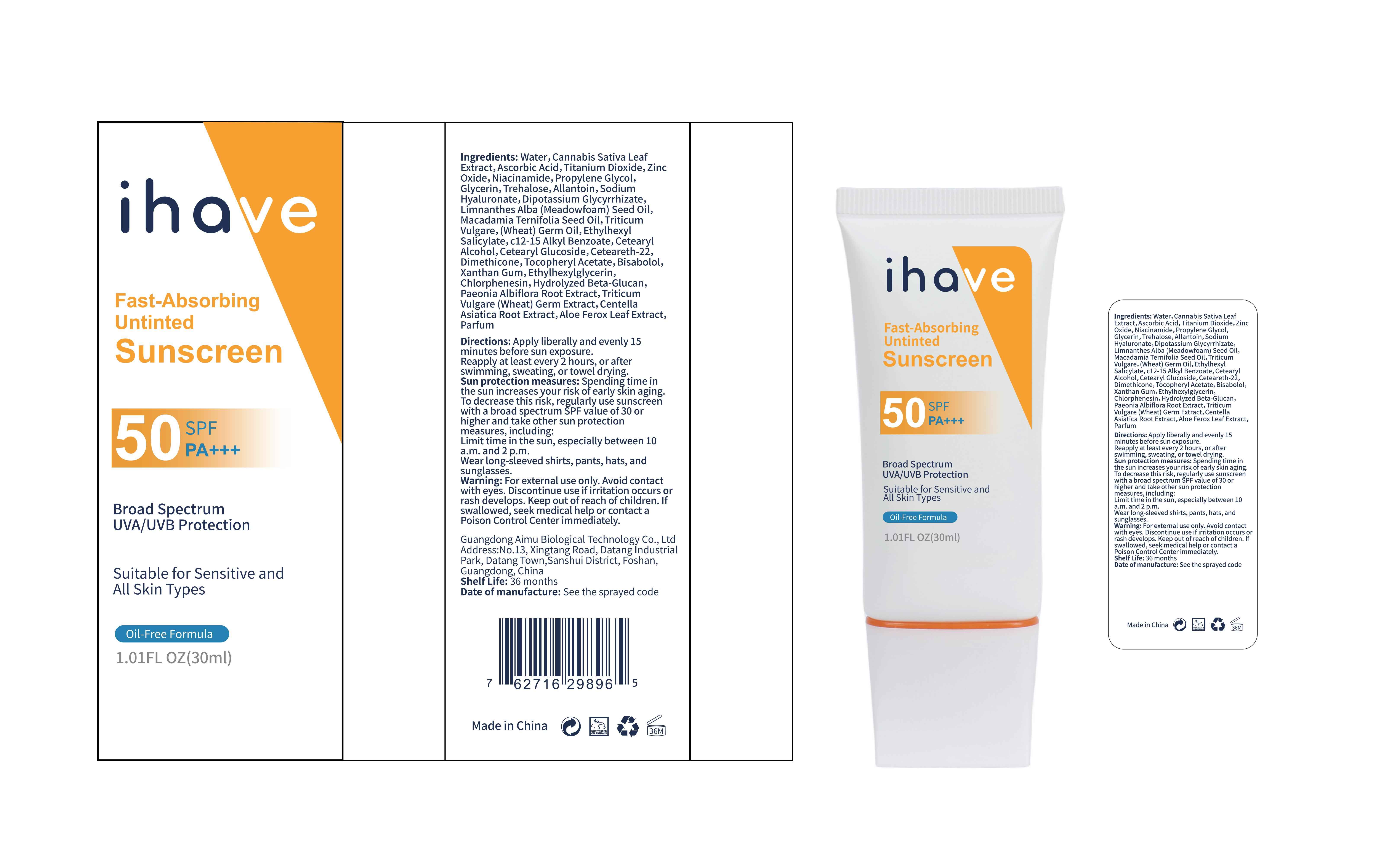
Label: IHAVE SUNSCREEN- titanium dioxide, zinc oxide, ethylhexyl salicylate cream
-
NDC Code(s):
83566-501-01,
83566-501-02,
83566-501-03,
83566-501-04, view more83566-501-05, 83566-501-06
- Packager: Guangdong Aimu Biological Technology Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 9, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
-
INACTIVE INGREDIENT
GLYCERIN
TREHALOSE
ALLANTOIN
PAEONIA LACTIFLORA ROOT
CETEARYL GLUCOSIDE
CHLORPHENESIN
MEADOWFOAM SEED OIL
HYDROGENATED MACADAMIA SEED OIL
WHEAT GERM OIL
ASCORBIC ACID
CETEARETH-22
WATER
NIACINAMIDE
PROPYLENE GLYCOL
HYALURONATE SODIUM
DIMETHICONE
CANNABIS SATIVA LEAF
CETOSTEARYL ALCOHOL
ALPHA-TOCOPHEROL ACETATE
WHEAT GERM
GLYCYRRHIZINATE DIPOTASSIUM
ALKYL (C12-15) BENZOATE
ETHYLHEXYLGLYCERIN
1,3-BETA-GLUCAN SYNTHASE COMPONENT FKS1
CENTELLA ASIATICA ROOT
ALOE FEROX LEAF
XANTHAN GUM
LEVOMENOL - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
IHAVE SUNSCREEN
titanium dioxide, zinc oxide, ethylhexyl salicylate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83566-501 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 6 g in 100 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 6 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 3 g in 100 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) TREHALOSE (UNII: B8WCK70T7I) ALLANTOIN (UNII: 344S277G0Z) PAEONIA LACTIFLORA ROOT (UNII: 3Z3866YW6P) CETEARYL GLUCOSIDE (UNII: 09FUA47KNA) CHLORPHENESIN (UNII: I670DAL4SZ) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) HYDROGENATED MACADAMIA SEED OIL (UNII: W4FFC8IM2K) WHEAT GERM OIL (UNII: 14C97E680P) ASCORBIC ACID (UNII: PQ6CK8PD0R) CETEARETH-22 (UNII: 28VZG1E234) WATER (UNII: 059QF0KO0R) NIACINAMIDE (UNII: 25X51I8RD4) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) HYALURONATE SODIUM (UNII: YSE9PPT4TH) DIMETHICONE (UNII: 92RU3N3Y1O) CANNABIS SATIVA LEAF (UNII: A718Z7DA1K) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) WHEAT GERM (UNII: YR3G369F5A) GLYCYRRHIZINATE DIPOTASSIUM (UNII: CA2Y0FE3FX) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) 1,3-BETA-GLUCAN SYNTHASE COMPONENT FKS1 (UNII: 4FRY3LJG0V) CENTELLA ASIATICA ROOT (UNII: 8MXU1P42J0) ALOE FEROX LEAF (UNII: 0D145J8EME) XANTHAN GUM (UNII: TTV12P4NEE) LEVOMENOL (UNII: 24WE03BX2T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83566-501-01 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/09/2024 2 NDC:83566-501-02 40 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/09/2024 3 NDC:83566-501-03 50 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/09/2024 4 NDC:83566-501-04 60 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/09/2024 5 NDC:83566-501-05 80 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/09/2024 6 NDC:83566-501-06 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/09/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 04/09/2024 Labeler - Guangdong Aimu Biological Technology Co., Ltd (712647107) Establishment Name Address ID/FEI Business Operations Guangdong Aimu Biological Technology Co., Ltd 712647107 manufacture(83566-501)