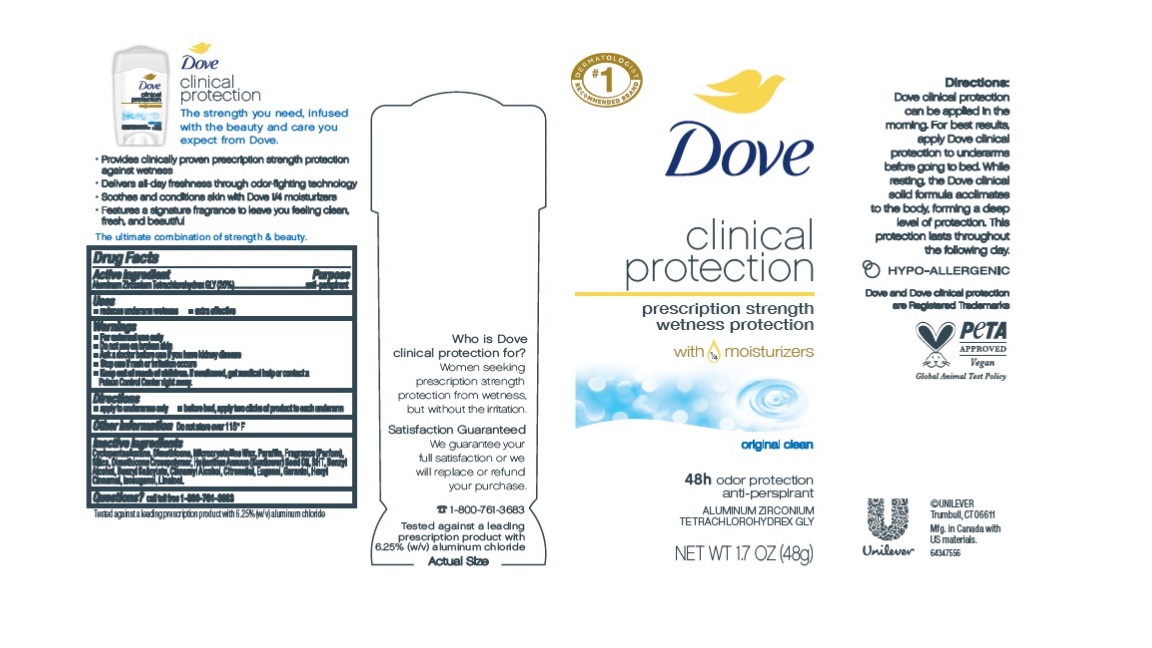
Label: DOVE- clinical protection original clean antiperspirant and deodorant stick
- NDC Code(s): 64942-2269-1, 64942-2269-2
- Packager: Conopco Inc. d/b/a/ Unilever
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DOVE CLINICAL PROTECTION ORIGINAL CLEAN ANTIPERSPIRANT & DEODORANT - aluminum zirconium tetrachlorohydrex gly stick
- Drug Facts
- Purpose
- Uses
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- Questions?
- Packaging
-
INGREDIENTS AND APPEARANCE
DOVE
clinical protection original clean antiperspirant and deodorant stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:64942-2269 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY (UNII: 8O386558JE) (ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY - UNII:8O386558JE) ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY 20 g in 100 g Inactive Ingredients Ingredient Name Strength .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) EUGENOL (UNII: 3T8H1794QW) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) PARAFFIN (UNII: I9O0E3H2ZE) SUNFLOWER OIL (UNII: 3W1JG795YI) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) DIMETHICONE (UNII: 92RU3N3Y1O) DIMETHICONE CROSSPOLYMER (450000 MPA.S AT 12% IN CYCLOPENTASILOXANE) (UNII: UF7620L1W6) CINNAMYL ALCOHOL (UNII: SS8YOP444F) GERANIOL (UNII: L837108USY) BENZYL SALICYLATE (UNII: WAO5MNK9TU) BENZYL ALCOHOL (UNII: LKG8494WBH) .ALPHA.-HEXYLCINNAMALDEHYDE (UNII: 7X6O37OK2I) LINALOOL, (+/-)- (UNII: D81QY6I88E) ISOEUGENOL (UNII: 5M0MWY797U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64942-2269-2 1 in 1 CARTON 04/14/2023 1 NDC:64942-2269-1 48 g in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M019 04/14/2023 Labeler - Conopco Inc. d/b/a/ Unilever (001375088)