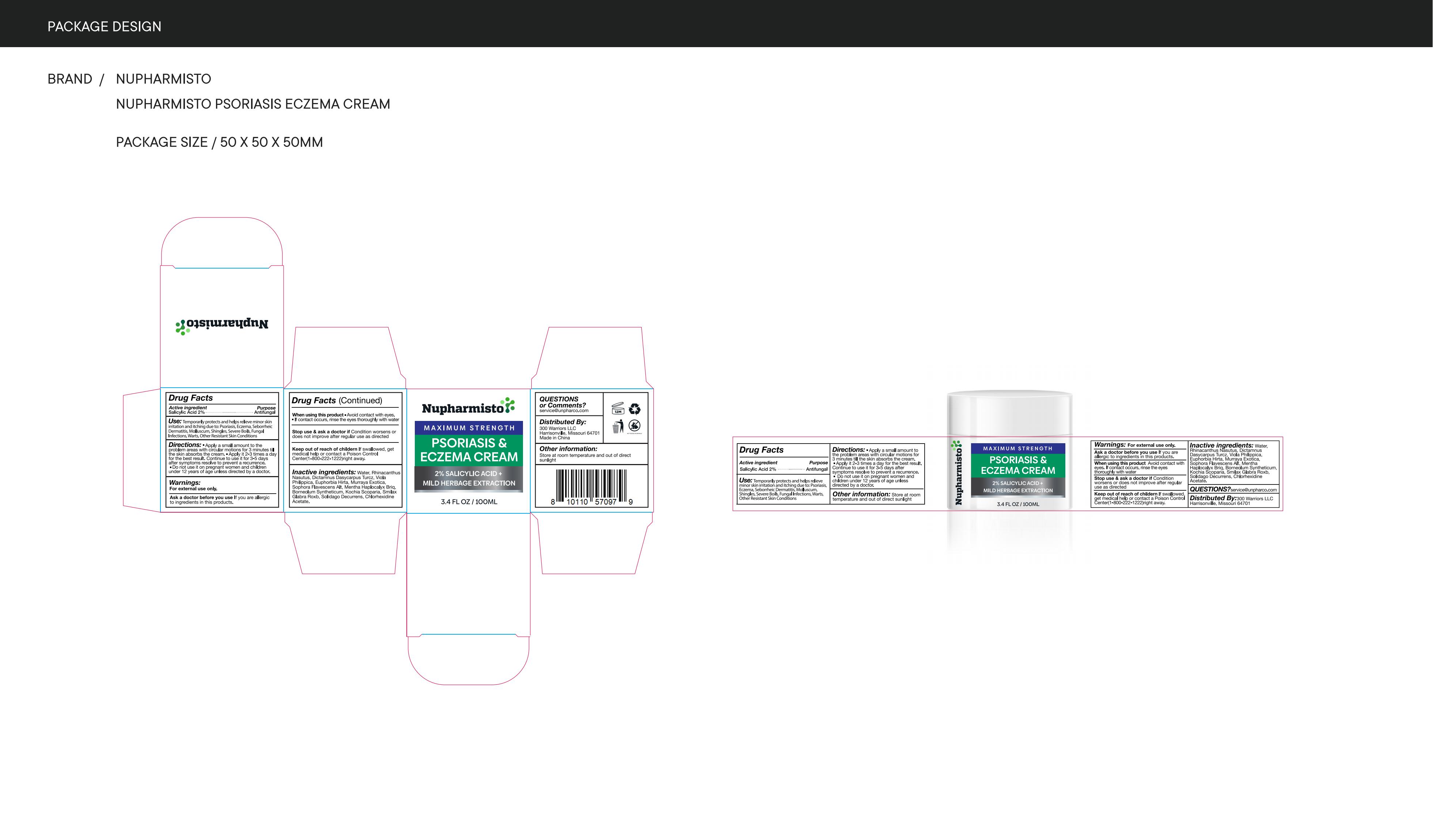
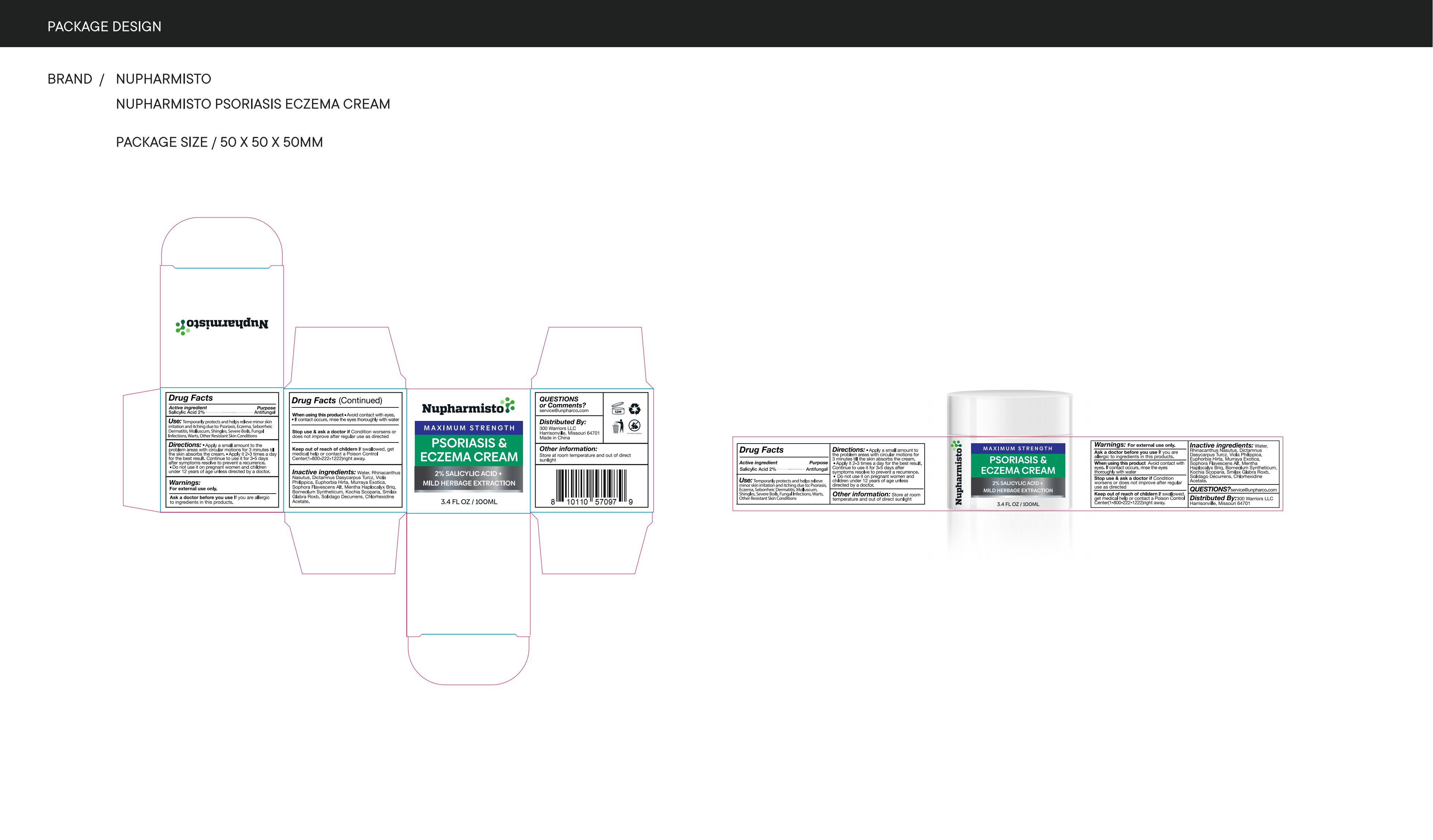
Label: NUPHARMISTO PSORIASIS ECZEMA CREAM cream
- NDC Code(s): 83771-002-01
- Packager: Inner Mongolia Green source pharmaceutical Products Co., LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 7, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
- Do not use
- When Using
- Stop Use
- Ask Doctor
- Keep Oot Of Reach Of Children
-
Directions
Apply a small amount to theproblem areas with circular motions for 3 minutes tilthe skin absorbs the cream."Apply it 2-3 times a dayfor the best result. Continue to use it for 3-5 daysafter symptoms resolve to prevent a recurrence.Do not use it on pregnant women and childrenunder 12 years of age unless directed by a doctor
- Other information
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NUPHARMISTO PSORIASIS ECZEMA CREAM
nupharmisto psoriasis eczema cream creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83771-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 0.2 g in 100 mL Inactive Ingredients Ingredient Name Strength CHLORHEXIDINE ACETATE (UNII: 5908ZUF22Y) DICTAMNUS DASYCARPUS WHOLE (UNII: IM7ZCU30YH) SOPHORA FLAVESCENS WHOLE (UNII: X8KX602M5L) WATER (UNII: 059QF0KO0R) BASSIA SCOPARIA WHOLE (UNII: 240G38P85Z) RHINACANTHUS NASUTUS WHOLE (UNII: QW70YFP724) EUPHORBIA HIRTA (UNII: L13YF113GN) MURRAYA PANICULATA FRUIT (UNII: PE014Z7R7C) SOLIDAGO DECURRENS WHOLE (UNII: Y026RJV99T) VIOLA PHILIPPICA WHOLE (UNII: O176VC3N54) MENTHA CANADENSIS FLOWERING TOP (UNII: 2492Y6JJNA) BORNEOL (UNII: M89NIB437X) SMILAX GLABRA WHOLE (UNII: H51N91QNEB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83771-002-01 100 mL in 1 CANISTER; Type 0: Not a Combination Product 04/06/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M005 04/06/2024 Labeler - Inner Mongolia Green source pharmaceutical Products Co., LTD (699508923) Establishment Name Address ID/FEI Business Operations Inner Mongolia Green source pharmaceutical Products Co., LTD 699508923 label(83771-002) , manufacture(83771-002)