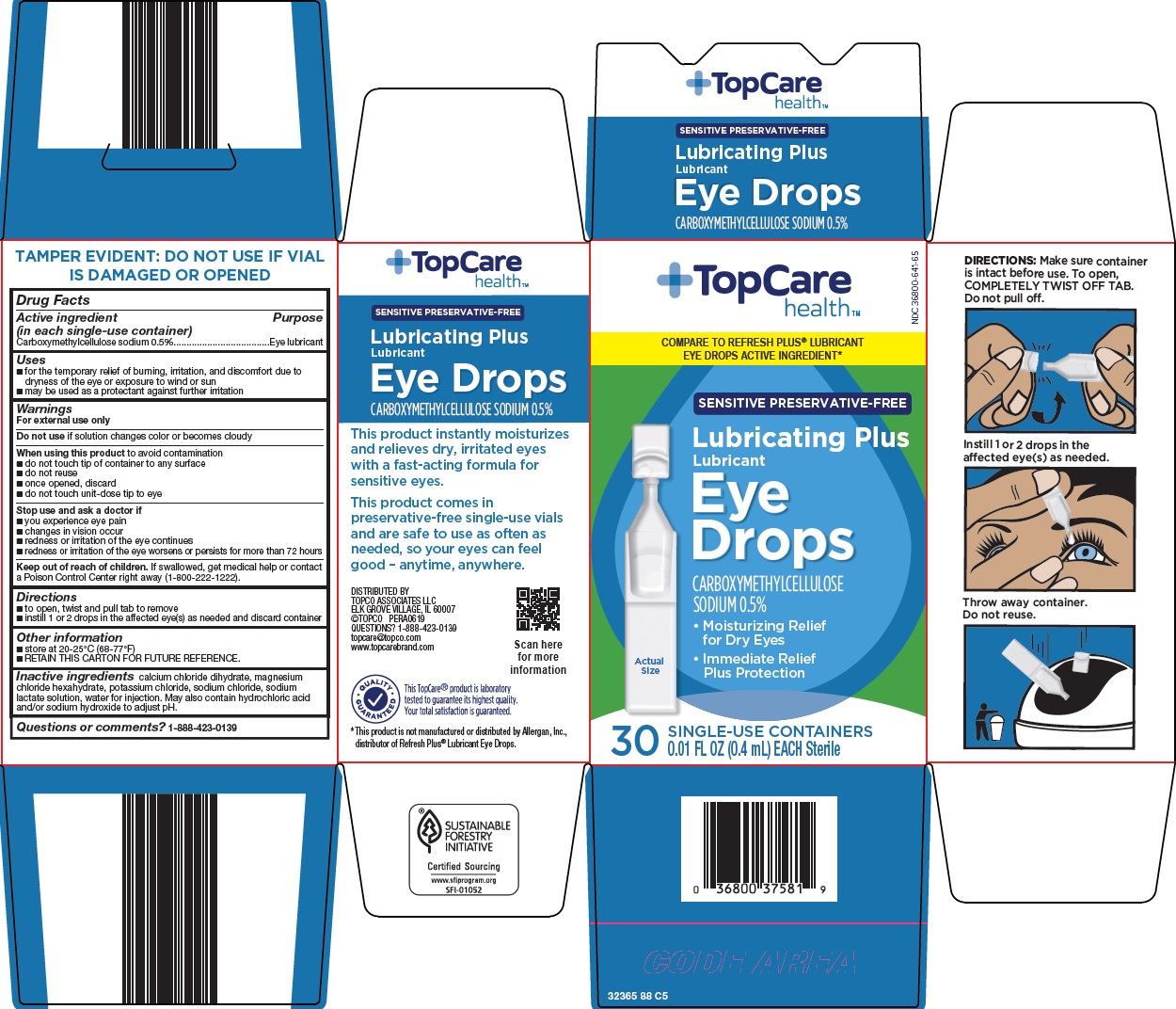
Label: TOPCARE EYE DROPS- carboxymethylcellulose sodium solution/ drops
- NDC Code(s): 36800-641-65
- Packager: Topco Associates LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 1, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each single-use container)
- Purpose
- Uses
-
Warnings
For external use only
When using this product
to avoid contamination
- •
- do not touch tip of container to any surface
- •
- do not reuse
- •
- once opened, discard
- •
- do not touch unit-dose tip to eye
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Package/Label Principal Display Panel
TopCare health™
COMPARE TO REFRESH PLUS® LUBRICANT EYE DROPS ACTIVE INGREDIENT
SENSITIVE PRESERVATIVE-FREE
Lubricating Plus
Lubricant
Eye Drops
CARBOXYMETHYLCELLULOSE SODIUM 0.5%
Moisturizing Relief for Dry Eyes
Immediate Relief Plus Protection
Actual Size
30 SINGLE-USE CONTAINERS
0.01 FL OZ (0.4 mL) EACH Sterile
-
INGREDIENTS AND APPEARANCE
TOPCARE EYE DROPS
carboxymethylcellulose sodium solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:36800-641 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) (CARBOXYMETHYLCELLULOSE - UNII:05JZI7B19X) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM 0.5 g in 100 mL Inactive Ingredients Ingredient Name Strength CALCIUM CHLORIDE (UNII: M4I0D6VV5M) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:36800-641-65 6 in 1 CARTON 05/31/2019 1 5 in 1 POUCH 1 0.4 mL in 1 VIAL, SINGLE-USE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 05/31/2019 Labeler - Topco Associates LLC (006935977)