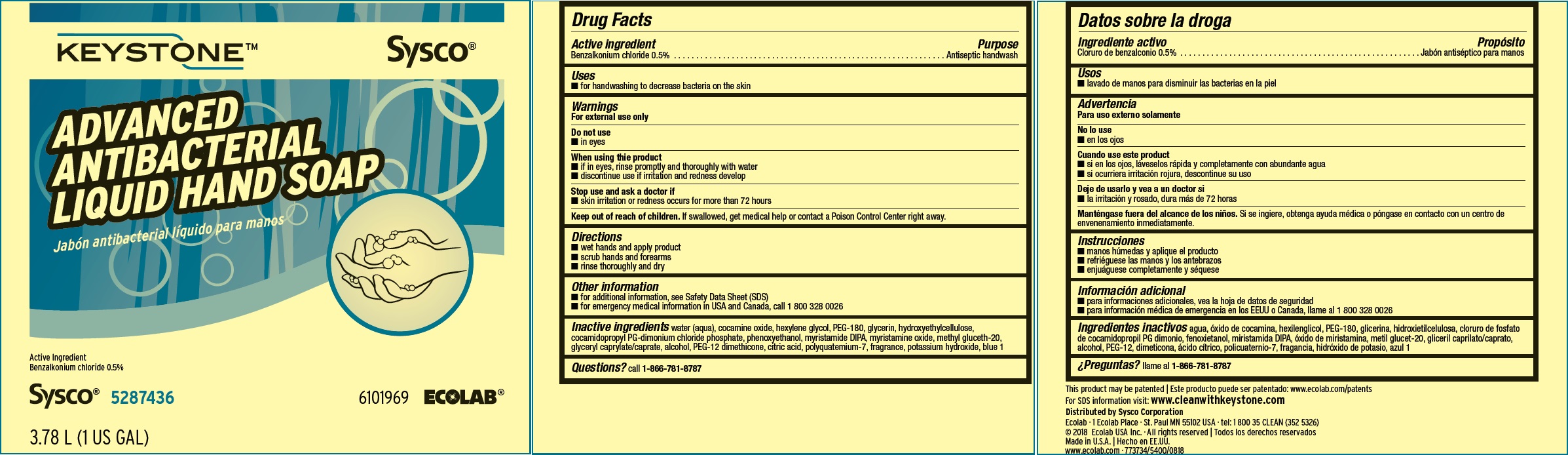
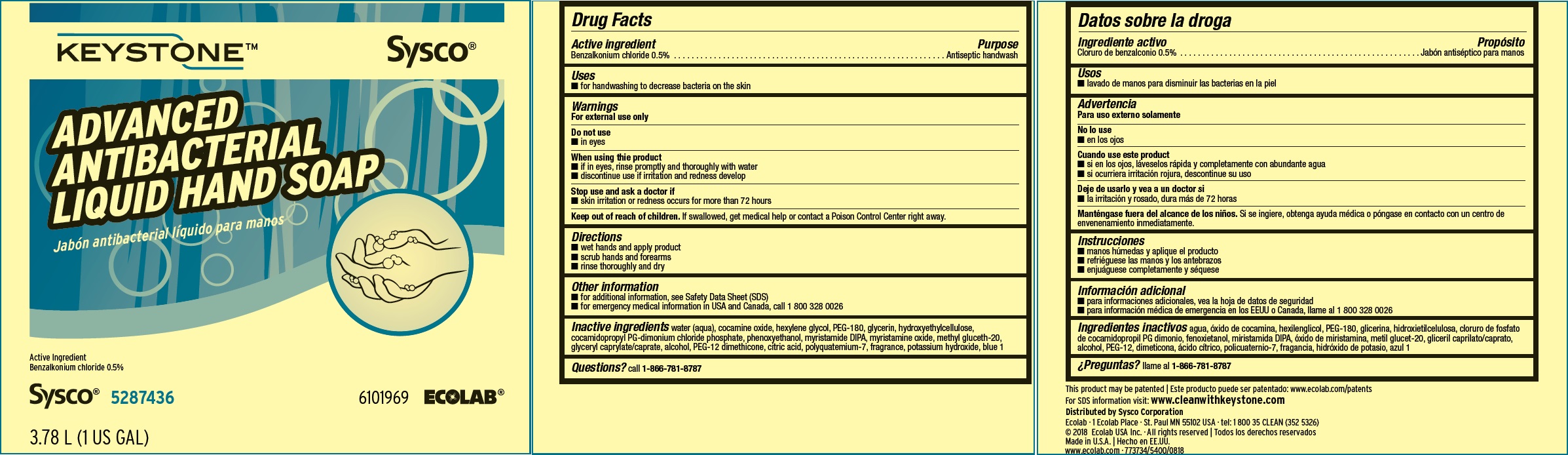
Label: KEYSTONE- benzalkonium chloride solution
- NDC Code(s): 47593-594-11
- Packager: Ecolab Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 24, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- Directions
- Other information
-
INACTIVE INGREDIENT
Inactive ingredients water (aqua), cocamine oxide, hexylene glycol, PEG-180, glycerin, hydroxyethylcellulose, cocamidopropyl PG-dimonium chloride phosphate, phenoxyethanol, myristamide DIPA, myristamine oxide, methyl gluceth-20, glyceryl caprylate/caprate, alcohol, PEG-12 dimethicone, citric acid, polyquatemium-7, fragrance, potassium hydroxide, blue 1
- QUESTIONS
-
Principal display panel and representative label
KEYSTONE Sysco
ADVANCED ANTIBACTERIAL LIQUID HAND SOAP
Active Ingredient: 0.5% Benzalkonium Chloride
Net Contents
750 mL (25 US fl oz) 6000234
Sysco 5287436 6101969 ECOLAB®
This product may be patented: www.ecolab.com/patents
For SDS information visit: www.cleanwithkeystone.com
Ecolab · 1 Ecolab Place · St. Paul MN 55102 USA · tel: 1 800 35 CLEAN (352 5326)
© 2018 Ecolab USA Inc. · All rights reserved
Made in U.S.A.
www.ecolab.com · 773734/5400/0818
-
INGREDIENTS AND APPEARANCE
KEYSTONE
benzalkonium chloride solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:47593-594 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) COCAMINE OXIDE (UNII: QWA2IZI6FI) HEXYLENE GLYCOL (UNII: KEH0A3F75J) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) GLYCERIN (UNII: PDC6A3C0OX) HYDROXYETHYL CELLULOSE (4000 MPA.S AT 1%) (UNII: ZYD53NBL45) COCAMIDOPROPYL PG-DIMONIUM CHLORIDE PHOSPHATE (UNII: H2KVQ74JM4) PHENOXYETHANOL (UNII: HIE492ZZ3T) MYRISTIC DIISOPROPANOLAMIDE (UNII: 17DN142CTK) MYRISTAMINE OXIDE (UNII: J086PM3RRT) METHYL GLUCETH-20 (UNII: J3QD0LD11P) GLYCERYL CAPRYLATE/CAPRATE (UNII: G7515SW10N) ALCOHOL (UNII: 3K9958V90M) PEG-12 DIMETHICONE (UNII: ZEL54N6W95) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) POLYQUATERNIUM-7 (70/30 ACRYLAMIDE/DADMAC; 1600000 MW) (UNII: 0L414VCS5Y) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:47593-594-11 3780 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/06/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 09/06/2018 Labeler - Ecolab Inc. (006154611)