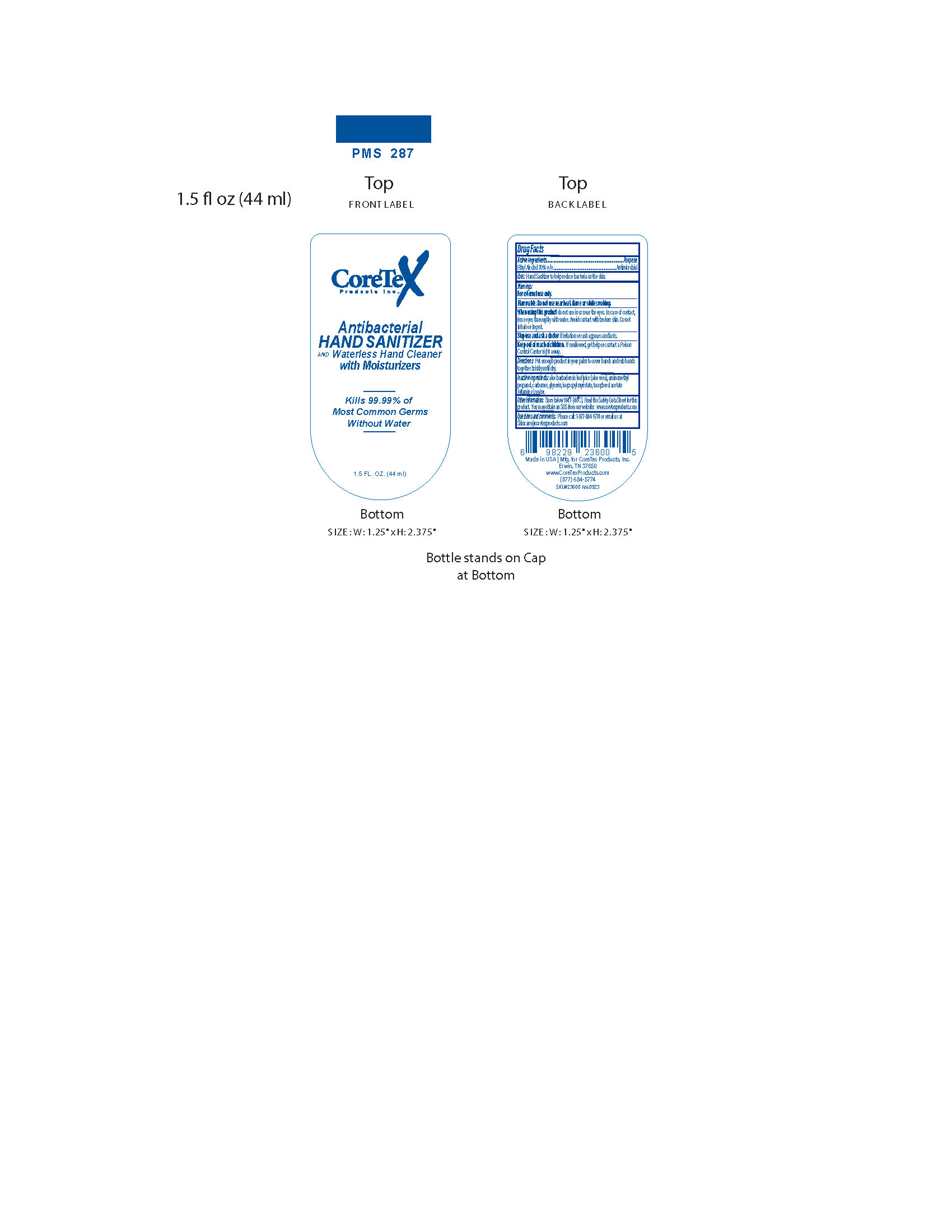
Label: CORETEX ANTIBACTERIAL HAND SANITIZER- sd alcohol liquid
-
NDC Code(s):
65753-201-01,
65753-201-02,
65753-201-03,
65753-201-05, view more65753-201-06, 65753-201-07, 65753-201-10, 65753-201-20, 65753-201-21, 65753-201-28, 65753-201-29, 65753-201-30, 65753-201-32
- Packager: CoreTex Products Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 1, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses:
- Warnings:
- Directions:
- Inactive Ingredients:
- Other Information:
- Questions and comments:
- SPL UNCLASSIFIED SECTION
- 65753-201-01
- 65753-201-02
- 65753-201-03
- 65753-201-05
- 65753-201-06
- 65753-201-07
- 65753-201-10
- 65753-201-28
- 65753-201-29
- 65753-201-30
- 65753-201-32
- 65753-201-20 and -21
-
INGREDIENTS AND APPEARANCE
CORETEX ANTIBACTERIAL HAND SANITIZER
sd alcohol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:65753-201 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 70 mL in 100 mL Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) ALOE VERA LEAF (UNII: ZY81Z83H0X) AMINOMETHYL PROPANEDIOL (UNII: CZ7BU4QZJZ) GLYCERIN (UNII: PDC6A3C0OX) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) WATER (UNII: 059QF0KO0R) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65753-201-01 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/01/2024 2 NDC:65753-201-02 59 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/01/2024 3 NDC:65753-201-03 118 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/01/2024 4 NDC:65753-201-32 44 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/01/2024 5 NDC:65753-201-28 118 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 01/01/2024 6 NDC:65753-201-05 237 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/01/2024 7 NDC:65753-201-29 237 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 01/01/2024 8 NDC:65753-201-06 355 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/01/2024 9 NDC:65753-201-07 354 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 01/01/2024 10 NDC:65753-201-30 473 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 01/01/2024 11 NDC:65753-201-10 3800 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/01/2024 12 NDC:65753-201-20 25 in 1 CONTAINER 01/01/2024 12 3.5 mL in 1 POUCH; Type 0: Not a Combination Product 13 NDC:65753-201-21 300 in 1 CONTAINER 01/01/2024 13 3.5 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 01/01/2024 Labeler - CoreTex Products Inc (061944620) Establishment Name Address ID/FEI Business Operations CoreTex Products Inc 061944620 pack(65753-201) , label(65753-201) Establishment Name Address ID/FEI Business Operations Prime Enterprises 101946028 manufacture(65753-201) , pack(65753-201) , analysis(65753-201)