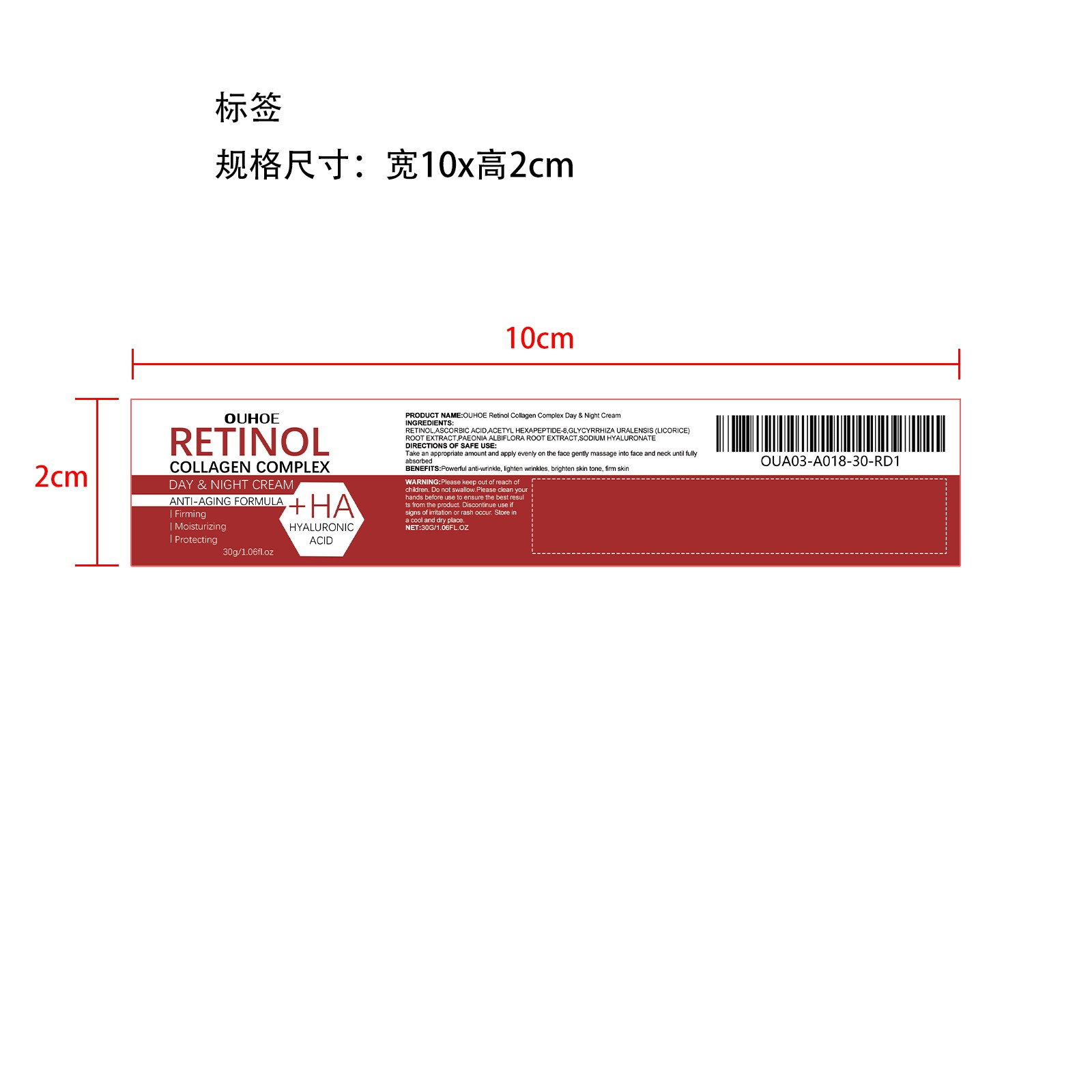
Label: CREAM cream
- NDC Code(s): 84067-076-01
- Packager: Shantou Youjia E-Commerce Co.,Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Export only
Drug Label Information
Updated March 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CREAM
cream creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84067-076 Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RETINOL (UNII: G2SH0XKK91) (RETINOL - UNII:G2SH0XKK91) RETINOL 3 mg in 30 g Inactive Ingredients Ingredient Name Strength GLYCYRRHIZA URALENSIS (UNII: 42B5YD8F0K) 4.5 mg in 30 g PAEONIA LACTIFLORA ROOT (UNII: 3Z3866YW6P) 3 mg in 30 g ASCORBIC ACID (UNII: PQ6CK8PD0R) 6 mg in 30 g ACETYL HEXAPEPTIDE-8 (UNII: L4EL31FWIL) 4.5 mg in 30 g HYALURONATE SODIUM (UNII: YSE9PPT4TH) 9 mg in 30 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84067-076-01 30 g in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2024 12/31/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Export only 02/01/2024 12/31/2024 Labeler - Shantou Youjia E-Commerce Co.,Ltd. (711173127) Establishment Name Address ID/FEI Business Operations Shantou Youjia E-Commerce Co.,Ltd. 711173127 label(84067-076)