Label: SODIUM POLYSTYRENE SULFONATE suspension
- NDC Code(s): 46287-006-01, 46287-006-04, 46287-006-60
- Packager: CMP Pharma, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated August 15, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Sodium Polystyrene Sulfonate Suspension USP (SPS® Suspension) can be administered orally or in an enema. It is a cherry-flavored suspension containing 15 grams of cation-exchange resin (Sodium Polystyrene Sulfonate USP); 21.5 mL of Sorbitol Solution USP (equivalent to approximately 20 grams of Sorbitol); 0.18 mL (0.3%) of Alcohol per 60 mL of suspension. Also contains Purified Water USP; Propylene Glycol USP; Magnesium Aluminum Silicate NF; Sodium Saccharin USP; Methylparaben NF; Propylparaben NF; and flavor.
Sodium polystyrene sulfonate is a benzene, diethenyl-, polymer with ethenylbenzene, sulfonated, sodium salt and has the following structural formula:
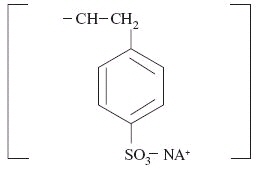
The sodium content of the suspension is 1500 mg (65 mEq) per 60 mL. It is a brown, slightly viscous suspension with an in‑vitro exchange capacity of approximately 3.1 mEq (in-vivo approximately 1 mEq) of potassium per 4 mL (1 gram) of suspension. It can be administered orally or in an enema.
-
CLINICAL PHARMACOLOGY
As the resin passes along the intestine or is retained in the colon after administration by enema, the sodium ions are partially released and are replaced by potassium ions. For the most part, this action occurs in the large intestine, which excretes potassium ions to a greater degree than does the small intestine. The efficiency of this process is limited and unpredictably variable. It commonly approximates the order of 33%, but the range is so large that definitive indices of electrolyte balance must be clearly monitored.
Metabolic data are unavailable.
- INDICATION AND USAGE
-
CONTRAINDICATIONS
SPS® Suspension is contraindicated in the following conditions: patients with hypokalemia, patients with a history of hypersensitivity to polystyrene sulfonate resins, obstructive bowel disease, oral or rectal administration in neonates (See PRECAUTIONS).
-
WARNINGS
Intestinal Necrosis
Cases of intestinal necrosis, which may be fatal, and other serious gastrointestinal adverse events (bleeding, ischemic colitis, perforation) have been reported in association with sodium polystyrene sulfonate use. The majority of these cases reported the concomitant use of sorbitol. Risk factors for gastrointestinal adverse events were present in many of the cases including prematurity, history of intestinal disease or surgery, hypovolemia, and renal insufficiency and failure. Concomitant administration of additional sorbitol is not recommended (see PRECAUTIONS, Drug Interactions).
- Use only in patients who have normal bowel function. Avoid use in patients who have not had a bowel movement post-surgery.
- Avoid use in patients who are at risk for developing constipation or impaction (including those with history of impaction, chronic constipation, inflammatory bowel disease, ischemic colitis, vascular intestinal atherosclerosis, previous bowel resection, or bowel obstruction).
- Discontinue use in patients who develop constipation.
Alternative Therapy in Severe Hyperkalemia
Since the effective lowering of serum potassium with sodium polystyrene sulfonate may take hours to days, treatment with this drug alone may be insufficient to rapidly correct severe hyperkalemia associated with states of rapid tissue breakdown (e.g., burns and renal failure) or hyperkalemia so marked as to constitute a medical emergency. Therefore, other definitive measures, including dialysis, should always be considered and may be imperative.
Hypokalemia
Serious potassium deficiency can occur from sodium polystyrene sulfonate therapy. The effect must be carefully controlled by frequent serum potassium determinations within each 24 hour period. Since intracellular potassium deficiency is not always reflected by serum potassium levels, the level at which treatment with sodium polystyrene sulfonate should be discontinued must be determined individually for each patient. Important aids in making this determination are the patient's clinical condition and electrocardiogram. Early clinical signs of severe hypokalemia include a pattern of irritable confusion and delayed thought processes.
Electrocardiographically, severe hypokalemia is often associated with a lengthened Q-T interval, widening, flattening, or inversion of the T wave, and prominent U waves. Also, cardiac arrhythmias may occur, such as premature atrial, nodal, and ventricular contractions, and supraventricular and ventricular tachycardias. The toxic effects of digitalis are likely to be exaggerated. Marked hypokalemia can also be manifested by severe muscle weakness, at times extending into frank paralysis.
Electrolyte Disturbances
Like all cation-exchange resins, sodium polystyrene sulfonate is not totally selective (for potassium) in its actions, and small amounts of other cations such as magnesium and calcium can also be lost during treatment. Accordingly, patients receiving sodium polystyrene sulfonate should be monitored for all applicable electrolyte disturbances.
Systemic Alkalosis
Systemic alkalosis has been reported after cation-exchange resins were administered orally in combination with nonabsorbable cation-donating antacids and laxatives such as magnesium hydroxide and aluminum carbonate. Magnesium hydroxide should not be administered with sodium polystyrene sulfonate. One case of grand mal seizure has been reported in a patient with chronic hypocalcemia of renal failure who was given sodium polystyrene sulfonate with magnesium hydroxide as a laxative (See PRECAUTIONS, Drug Interactions).
Risk of Aspiration
Cases of acute bronchitis or bronchopneumonia caused by inhalation of sodium polystyrene sulfonate particles has been reported. Patients with impaired gag reflex, altered level of consciousness, or patients prone to regurgitation may be at increased risk. Administer sodium polystyrene sulfonate suspension with the patient in an upright position.
Binding to Other Orally Administered Medications
Sodium polystyrene sulfonate suspension may bind orally administered medications, which could decrease their gastrointestinal absorption and lead to reduced efficacy. Administer other oral medications at least 3 hours before or 3 hours after sodium polystyrene sulfonate suspension. Patients with gastroparesis may require a 6 hour separation (see DOSAGE AND ADMINISTRATION and PRECAUTIONS, Drug Interactions).
-
PRECAUTIONS
Caution is advised when sodium polystyrene sulfonate is administered to patients who cannot tolerate even a small increase in sodium loads (i.e., severe congestive heart failure, severe hypertension, or marked edema). In such instances compensatory restriction of sodium intake from other sources may be indicated.
Precautions should be taken to ensure the use of adequate volumes of sodium-free cleansing enemas after rectal administration.
In the event of clinically significant constipation, treatment with SPS® Suspension should be discontinued until normal bowel motion is resumed (See WARNINGS, Intestinal Necrosis).
Drug Interactions
General Interactions
No formal drug interaction studies have been conducted in humans.
Sodium polystyrene sulfonate suspension has the potential to bind other drugs. In in-vitro binding studies, sodium polystyrene sulfonate was shown to significantly bind the oral medications (n=6) that were tested. Decreased absorption of lithium and thyroxine have also been reported with co-administration of sodium polystyrene sulfonate. Binding of sodium polystyrene sulfonate suspension to other oral medications could cause decreased gastrointestinal absorption and loss of efficacy when taken close to the time sodium polystyrene sulfonate suspension is administered. Administer sodium polystyrene sulfonate suspension at least 3 hours before or 3 hours after other oral medications. Patients with gastroparesis may require a 6 hour separation. Monitor for clinical response and/or blood levels where possible.Antacids
The simultaneous oral administration of sodium polystyrene sulfonate with nonabsorbable cation-donating antacids and laxatives may reduce the resin's potassium exchange capability.
Nonabsorbable cation-donating antacids and laxatives
Systemic alkalosis has been reported after cation exchange resins were administered orally in combination with nonabsorbable cation-donating antacids and laxatives such as magnesium hydroxide and aluminum carbonate. Magnesium hydroxide should not be administered with sodium polystyrene sulfonate. One case of grand mal seizure has been reported in a patient with chronic hypocalcemia of renal failure who was given sodium polystyrene sulfonate with magnesium hydroxide as a laxative.
Intestinal obstruction due to concretions of aluminum hydroxide when used in combination with sodium polystyrene sulfonate has been reported.
Digitalis
The toxic effects of digitalis on the heart, especially various ventricular arrhythmias and A-V nodal dissociation, are likely to be exaggerated by hypokalemia, even in the face of serum digoxin concentrations in the "normal range" (See WARNINGS).
Sorbitol
Concomitant use of sorbitol with sodium polystyrene sulfonate has been implicated in cases of intestinal necrosis, which may be fatal (See WARNINGS).
Pregnancy Category C
Animal reproduction studies have not been conducted with sodium polystyrene sulfonate. It is also not known whether sodium polystyrene sulfonate can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Sodium polystyrene sulfonate should be given to a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when sodium polystyrene sulfonate is administered to a nursing woman.
Pediatric Use
The effectiveness of SPS® Suspension in pediatric patients has not been established. The use of SPS® Suspension is contraindicated in neonates and especially in premature infants. In children and neonates, particular care should be observed with rectal administration, as excessive dosage could result in impaction of the resin. Precautions should be taken to ensure the use of adequate volumes of sodium-free cleansing enemas after rectal administration.
-
ADVERSE REACTIONS
SPS® Suspension may cause some degree of gastric irritation. Anorexia, nausea, vomiting, and constipation may occur especially if high doses are given. Also, hypokalemia, hypocalcemia, hypomagnesemia and significant sodium retention, and their related clinical manifestations, may occur (See WARNINGS). Occasionally diarrhea develops. Large doses in elderly individuals may cause fecal impaction (See PRECAUTIONS). Rare instances of intestinal necrosis have been reported. Intestinal obstruction due to concretions of aluminum hydroxide, when used in combination with sodium polystyrene sulfonate, has been reported.
The following events have been reported from worldwide post marketing experience:
- Fecal impaction following rectal administration, particularly in children;
- Gastrointestinal concretions (bezoars) following oral administration;
- Ischemic colitis, gastrointestinal tract ulceration or necrosis which could lead to intestinal perforation; and
- Rare cases of acute bronchitis and/or bronchopneumonia associated with inhalation of particles of polystyrene sulfonate (see WARNINGS).
To report suspected adverse reactions, contact CMP Pharma, Inc., toll free at 1-844-321-1443 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
OVERDOSAGE
Overdosage may result in electrolyte disturbances including hypokalemia, hypocalcemia, and hypomagnesemia. Biochemical disturbances resulting from overdosage may give rise to clinical signs and symptoms of hypokalemia, including: irritability, confusion, delayed thought processes, muscle weakness, hyporeflexia, which may progress to frank paralysis and/or apnea. Tetany may occur. Electrocardiographic changes may be consistent with hypokalemia or hypocalcemia; cardiac arrhythmias may occur. Appropriate measures should be taken to correct serum electrolytes (potassium, calcium, magnesium), and the resin should be removed from the alimentary tract by appropriate use of laxatives or enemas.
-
DOSAGE AND ADMINISTRATION
Administer sodium polystyrene sulfonate suspension at least 3 hours before or 3 hours after other oral medications. Patients with gastroparesis may require a 6 hour separation (see WARNINGS and PRECAUTIONS, Drug Interactions).
The average daily adult dose is 15 g (60 mL) to 60 g (240 mL) of suspension. This is best provided by administering 15 g (60 mL) of SPS® Suspension one to four times daily. Each 60 mL of SPS® Suspension contains 1500 mg (65 mEq) of sodium. Since the in-vivo efficiency of sodium-potassium exchange resins is approximately 33%, about one-third of the resin's actual sodium content is being delivered to the body.
In smaller children and infants, lower doses should be employed by using as a guide a rate of 1 mEq of potassium per gram of resin as the basis for calculation. Administer with patient in an upright position (see WARNINGS).
SPS® Suspension may be introduced into the stomach through a plastic tube and, if desired, given with a diet appropriate for a patient in renal failure.
SPS® Suspension may also be given, although with less effective results, as an enema consisting (for adults) of 30 g (120 mL) to 50 g (200 mL) every six hours. The enema should be retained as long as possible and followed by a cleansing enema.
After an initial cleansing enema, a soft, large size (French 28) rubber tube is inserted into the rectum for a distance of about 20 cm, with the tip well into the sigmoid colon, and taped into place. The suspension is introduced at body temperature by gravity. The suspension is flushed with 50 or 100 mL of fluid, following which the tube is clamped and left in place. If back leakage occurs, the hips are elevated on pillows or a knee-chest position is taken temporarily. The suspension is kept in the sigmoid colon for several hours, if possible. Then the colon is irrigated with a sodium-free cleansing enema at body temperature in order to remove the resin. Two quarts of flushing solution may be necessary. The returns are drained constantly through a Y tube connection. Particular attention should be paid to this cleansing enema, because sorbitol is present in the vehicle.
The intensity and duration of therapy depend upon the severity and resistance of hyperkalemia.
SPS® Suspension should not be heated for to do so may alter the exchange properties of the resin.
-
HOW SUPPLIED
SPS® Suspension is a light brown, cherry-flavored suspension supplied in pint (473 mL) bottles (NDC 46287-006-01), 120 mL bottles (NDC 46287-006-04), and 60 mL unit dose bottles, 10 bottles per carton (NDC 46287-006-60).
Dispense in a tight container, as defined in the USP. If repackaging into other containers, store in refrigerator and use within 14 days of packaging.
SHAKE WELL BEFORE USING.
Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F). [See USP Controlled Room Temperature].
CMP Pharma, Inc.
P.O. Box 147
Farmville, North Carolina 27828Revised August 2021
3081 R0821
-
PRINCIPAL DISPLAY PANEL - 473 mL Bottle Label
NDC 46287–006–01
473 mLSPS® Suspension
SODIUM POLYSTYRENE SULFONATE SUSPENSION, USP
For Oral or Rectal Use
Sodium Polystyrene Sulfonate USP 15 g/60 mL
Also contains: Sorbitol Solution USP (equivalent to
approximately 20 g of Sorbitol), Alcohol 0.3%, Purified
Water USP, Propylene Glycol USP, Magnesium Aluminum
Silicate NF, Sodium Saccharin USP, Methylparaben NF,
Propylparaben NF, & FlavorSodium content 1.5 g (65 mEq) in 60 mL
USUAL DOSE: See accompanying package insert for full
information.Store at 20°-25°C (68°-77°F); excursions permitted
to 15°-30°C (59°-86°F). [See USP Controlled Room
Temperature].Dispense in a tight container, as defined in the USP.
GTIN: 00346287006015
SHAKE WELL
Rx Onlycmp
PHARMA
Farmville, NC 278283064
R1219
-
INGREDIENTS AND APPEARANCE
SODIUM POLYSTYRENE SULFONATE
sodium polystyrene sulfonate suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:46287-006 Route of Administration ORAL, RECTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM POLYSTYRENE SULFONATE (UNII: 1699G8679Z) (POLYSTYRENE SULFONIC ACID - UNII:70KO0R01RY) SODIUM POLYSTYRENE SULFONATE 15 g in 60 mL Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) 21.5 mL in 60 mL ALCOHOL (UNII: 3K9958V90M) 0.18 mL in 60 mL WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) SACCHARIN SODIUM (UNII: SB8ZUX40TY) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) Product Characteristics Color BROWN (Light) Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46287-006-01 473 mL in 1 BOTTLE; Type 0: Not a Combination Product 12/08/1982 2 NDC:46287-006-04 120 mL in 1 BOTTLE; Type 0: Not a Combination Product 07/16/1987 3 NDC:46287-006-60 10 in 1 CARTON 12/12/1985 3 60 mL in 1 BOTTLE, UNIT-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA087859 12/08/1982 Labeler - CMP Pharma, Inc. (005224175) Establishment Name Address ID/FEI Business Operations CMP Pharma, Inc. 005224175 MANUFACTURE(46287-006)

