Label: UNSEEN SUNSCREEN LIP PROTECTOR SPF 30- avobenzone, homosalate, octisalate, octocrylene stick
- NDC Code(s): 75936-638-01, 75936-638-02
- Packager: Supergoop, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 27, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Inactive Ingredients: Octyldodecanol, C12-15 Alkyl Benzoate, Ricinus Communis (Castor) Seed Oil, Diisostearyl Malate, Flavor, Dibutyl Lauroyl Glutamide, Phenethyl Benzoate, Dibutyl Ethylhexanoyl Glutamide, Silica Silylate, Brassica Campestris/Aleurites Fordi Oil Crosspolymer, Octyldodecyl Neopentanoate, Saccharin, Tocopherol, Euterpe Oleracea Fruit Oil
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- WARNINGS
- INDICATIONS & USAGE
-
DOSAGE & ADMINISTRATION
Directions
Apply generously and evenly 15 minutes before sun exposure
Reapply at least every 2 hours
Use a water resistant sunscreen if swimming or sweating
Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
Limit time in the sun, especially from 10 a.m.- 2 p.m.
Wear long-sleeved shirts, pants, hats, and sunglasses
Children under 6 months: Ask a doctor
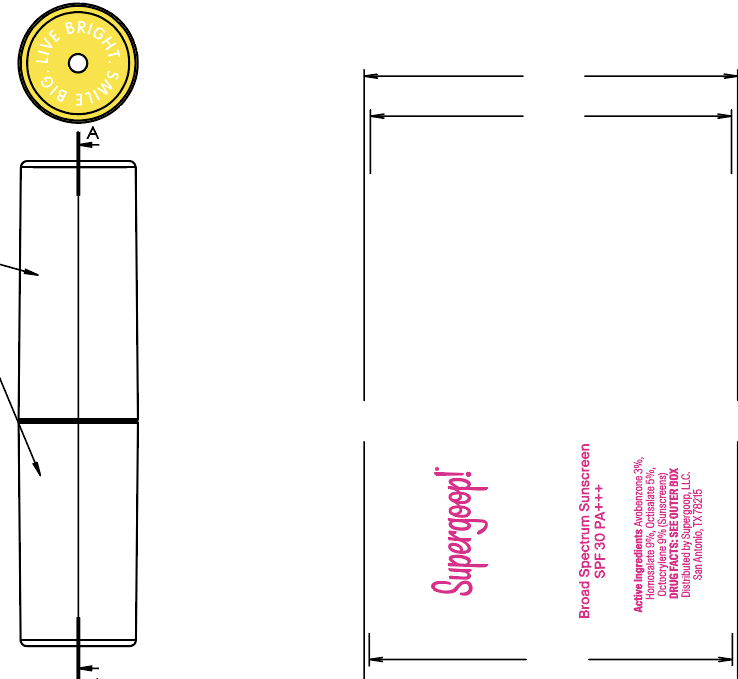
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
UNSEEN SUNSCREEN LIP PROTECTOR SPF 30
avobenzone, homosalate, octisalate, octocrylene stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:75936-638 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 100 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 9 g in 100 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 9 g in 100 mL Inactive Ingredients Ingredient Name Strength SILICA DIMETHYL SILYLATE (UNII: EU2PSP0G0W) OCTYLDODECYL NEOPENTANOATE (UNII: X8725R883T) OCTYLDODECANOL (UNII: 461N1O614Y) BRASSICA RAPA VAR. RAPA OIL (UNII: N4G8379626) DIBUTYL ETHYLHEXANOYL GLUTAMIDE (UNII: 0IAF2L30VS) DIISOSTEARYL MALATE (UNII: QBS8A3XZGQ) RICINUS COMMUNIS SEED (UNII: 7EK4SFN1TX) EUTERPE OLERACEA WHOLE (UNII: Y57H6218HP) DIBUTYL LAUROYL GLUTAMIDE (UNII: 3V7K3IA58X) PHENETHYL BENZOATE (UNII: 0C143929GK) SACCHARIN (UNII: FST467XS7D) TOCOPHEROL (UNII: R0ZB2556P8) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) DRYOBALANOPS AROMATICA WHOLE (UNII: 34303GFW6A) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:75936-638-02 1 in 1 CARTON 02/29/2024 1 NDC:75936-638-01 3.5 mL in 1 CYLINDER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 02/29/2024 Labeler - Supergoop, LLC (117061743) Registrant - Supergoop, LLC (117061743)